Abstract
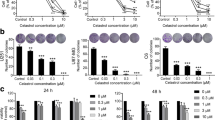
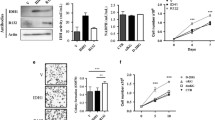
IDH1 mutations in gliomas associate with longer survival. Prooxidant and antiproliferative effects of IDH1 mutations and its d-2-hydroxyglutarate (2-HG) product have been described in vitro, but inconsistently observed. It is also unclear whether overexpression of mutant IDH1 in wild-type cells accurately phenocopies the effects of endogenous IDH1-mutations on tumor apoptosis and autophagy. Herein we investigated the effects of 2-HG and mutant IDH1 overexpression on proliferation, apoptosis, oxidative stress, and autophagy in IDH1 wild-type glioma cells, and compared those results with patient-derived tumors. 2-HG reduced viability and proliferation of U87MG and LN18 cells, triggered apoptosis in LN18 cells, and autophagy in U87MG cells. In vitro studies and flank xenografts of U87MG cells overexpressing R132H IDH1 exhibited increased oxidative stress, including increases of both manganese superoxide dismutase (MnSOD) and p62. Patient-derived IDH1-mutant tumors showed no significant differences in apoptosis or autophagy, but showed p62 accumulation and actually trended toward reduced MnSOD expression. These data indicate that mutant IDH1 and 2-HG can induce oxidative stress, autophagy, and apoptosis, but these effects vary greatly according to cell type.





Similar content being viewed by others
References
Bleeker FE, Atai NA, Lamba S et al (2010) The prognostic IDH1(R132) mutation is associated with reduced NADP+-dependent IDH activity in glioblastoma. Acta Neuropathol 119(4):487–494
Bralten LB, Kloosterhof NK, Balvers R et al (2011) IDH1 R132H decreases proliferation of glioma cell lines in vitro and in vivo. Ann Neurol 69(3):455–463
Brauburger K, Burckhardt G, Burckhardt BC (2011) The sodium-dependent di- and tricarboxylate transporter, NaCT, is not responsible for the uptake of d-, L-2-hydroxyglutarate and 3-hydroxyglutarate into neurons. J Inherit Metab Dis 34(2):477–482
Cherra SJ 3rd, Dagda RK, Chu CT (2010) Review: autophagy and neurodegeneration: survival at a cost? Neuropathol Appl Neurobiol 36(2):125–132
Choi AM, Ryter SW, Levine B (2013) Autophagy in human health and disease. N Engl J Med 368(7):651–662
Chowdhury R, Yeoh KK, Tian YM et al (2011) The oncometabolite 2-hydroxyglutarate inhibits histone lysine demethylases. EMBO Rep 12(5):463–469
Dang L, White DW, Gross S et al (2009) Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 462(7274):739–744
Dhar SK, St Clair DK (2012) Manganese superoxide dismutase regulation and cancer. Free Radic Biol Med. 52(11–12):2209–2222
Haapasalo H, Kylaniemi M, Paunul N, Kinnula VL, Soini Y (2003) Expression of antioxidant enzymes in astrocytic brain tumors. Brain Pathol 13(2):155–164
Horbinski C (2013) What do we know about IDH1/2 mutations so far, and how do we use it? Acta Neuropathol 125(5):621–636
Ishii N, Maier D, Merlo A et al (1999) Frequent co-alterations of TP53, p16/CDKN2A, p14ARF, PTEN tumor suppressor genes in human glioma cell lines. Brain Pathol 9(3):469–479
Jane EP, Premkumar DR, Pollack IF (2011) Bortezomib sensitizes malignant human glioma cells to TRAIL, mediated by inhibition of the NF-κB signaling pathway. Mol Cancer Therap 10(1):198–208
Jin G, Reitman ZJ, Duncan CG et al (2013) Disruption of wild-type IDH1 suppresses d-2-hydroxyglutarate production in IDH1-mutated gliomas. Cancer Res 73(2):496–501
Jin G, Reitman ZJ, Spasojevic I et al (2011) 2-Hydroxyglutarate production, but not dominant negative function, is conferred by glioma-derived NADP-dependent isocitrate dehydrogenase mutations. PloS One 6(2):e16812
Klink B, Miletic H, Stieber D et al (2013) A novel, diffusely infiltrative xenograft model of human anaplastic oligodendroglioma with mutations in FUBP1, CIC, and IDH1. PLoS ONE 8(3):e59773
Klionsky DJ, Abeliovich H, Agostinis P et al (2008) Guidelines for the use and interpretation of assays for monitoring autophagy in higher eukaryotes. Autophagy. 4(2):151–175
Ladoire S, Chaba K, Martins I et al (2012) Immunohistochemical detection of cytoplasmic LC3 puncta in human cancer specimens. Autophagy. 8(8):1175–1184
Latini A, da Silva CG, Ferreira GC et al (2005) Mitochondrial energy metabolism is markedly impaired by d-2-hydroxyglutaric acid in rat tissues. Mol Genet Metab 86(1–2):188–199
Latini A, Scussiato K, Rosa RB et al (2003) d-2-hydroxyglutaric acid induces oxidative stress in cerebral cortex of young rats. Eur J Neurosci 17(10):2017–2022
Lazovic J, Soto H, Piccioni D et al (2012) Detection of 2-hydroxyglutaric acid in vivo by proton magnetic resonance spectroscopy in U87 glioma cells overexpressing isocitrate dehydrogenase-1 mutation. Neuro Oncol. 14(12):1465–1472
Leonardi R, Subramanian C, Jackowski S, Rock CO (2012) Cancer-associated isocitrate dehydrogenase mutations inactivate NADPH-dependent reductive carboxylation. The J Biol Chem 287(18):14615–14620
Li S, Chou AP, Chen W et al (2013) Overexpression of isocitrate dehydrogenase mutant proteins renders glioma cells more sensitive to radiation. Neuro Oncol. 15(1):57–68
Li X, Fang P, Mai J, Choi ET, Wang H, Yang XF (2013) Targeting mitochondrial reactive oxygen species as novel therapy for inflammatory diseases and cancers. J Hematol Oncol. 6:19
Losman JA, Looper R, Koivunen P et al (2013) (R)-2-Hydroxyglutarate Is sufficient to promote leukemogenesis and its effects are reversible. Science 339(6127):1621–1625
Luchman HA, Stechishin OD, Dang NH et al (2012) An in vivo patient-derived model of endogenous IDH1-mutant glioma. Neuro-oncology 14(2):184–191
Metallo CM, Gameiro PA, Bell EL et al (2011) Reductive glutamine metabolism by IDH1 mediates lipogenesis under hypoxia. Nature 481(7381):380–384
Mohrenz IV, Antonietti P, Pusch S et al (2013) Isocitrate dehydrogenase 1 mutant R132H sensitizes glioma cells to BCNU-induced oxidative stress and cell death. Apoptosis 18(11):1416–1425
Mullen AR, Wheaton WW, Jin ES et al (2011) Reductive carboxylation supports growth in tumour cells with defective mitochondria. Nature 481(7381):385–388
Nezis IP, Stenmark H (2012) p62 at the interface of autophagy, oxidative stress signaling, and cancer. Antioxid Redox Signal 17(5):786–793
Piaskowski S, Bienkowski M, Stoczynska-Fidelus E et al (2011) Glioma cells showing IDH1 mutation cannot be propagated in standard cell culture conditions. Br J Cancer 104(6):968–970
Poon HF, Castegna A, Farr SA et al (2004) Quantitative proteomics analysis of specific protein expression and oxidative modification in aged senescence-accelerated-prone 8 mice brain. Neuroscience 126(4):915–926
Reitman ZJ, Jin G, Karoly ED et al (2011) Profiling the effects of isocitrate dehydrogenase 1 and 2 mutations on the cellular metabolome. Proc Natl Acad Sci USA 108(8):3270–3275
Shibata T, Kokubu A, Miyamoto M, Sasajima Y, Yamazaki N (2011) Mutant IDH1 confers an in vivo growth in a melanoma cell line with BRAF mutation. Am J Pathol 178(3):1395–1402
Struys EA, Jansen EE, Verhoeven NM, Jakobs C (2004) Measurement of urinary d- and L-2-hydroxyglutarate enantiomers by stable-isotope-dilution liquid chromatography-tandem mass spectrometry after derivatization with diacetyl-L-tartaric anhydride. Clinical Chem 50(8):1391–1395
Sultana R, Butterfield DA (2008) Slot-blot analysis of 3-nitrotyrosine-modified brain proteins. Methods Enzymol 440:309–316
Valadez JG, Grover VK, Carter MD et al. (2012) Identification of Hedgehog pathway responsive glioblastomas by isocitrate dehydrogenase mutation. Cancer Lett 328(2):297–306
Wise DR, Ward PS, Shay JE et al (2011) Hypoxia promotes isocitrate dehydrogenase-dependent carboxylation of alpha-ketoglutarate to citrate to support cell growth and viability. Proc Natl Acad Sci USA 108(49):19611–19616
Xu W, Yang H, Liu Y et al (2010) Oncometabolite 2-hydroxyglutarate is a competitive inhibitor of alpha-ketoglutarate-dependent dioxygenases. cancer cell 19(1):17–30
Zhu J, Cui G, Chen M et al (2012) Expression of R132H mutational IDH1 in human U87 glioblastoma cells affects the SREBP1a pathway and induces cellular proliferation. J Mol Neurosci 50(1):165–171
Acknowledgments
C.H. was supported by K08 CA155764 (National Cancer Institute), 2P20 RR020171 COBRE pilot grant (National Institute of General Medical Sciences), The Peter and Carmen Lucia Buck Training Program in Translational Clinical Oncology, and the University of Kentucky College of Medicine Physician Scientist Program. The Markey Biospecimen and Tissue Procurement (BSTP) Shared Resource Facility facilitated the construction of tissue microarrays and immunohistochemical studies. Special thanks to Dana Napier for her excellent histologic expertise. Study data were collected and managed using REDCap electronic data capture tools hosted at the University of Kentucky. This research was also supported by the Markey Cancer Center Free Radical Biology in Cancer (FRBC) Shared Resource Facility. Flow cytometry and cell sorting was carried out at the University of Kentucky Flow Cytometry and Cell Sorting (FCCS) Core Facility, which is supported in part by the Office of the Vice President for Research, the Markey Cancer Center and a grant from the NIH Shared Instrument Program (S10 RR026827-01A1). The BSTP, FRBC, and FCCS Shared Resource Facilities are all supported by the University of Kentucky Markey Cancer Center (P30CA177558). We thank Dr. Hai Yan of Duke University Medical Center for supplying us with pEGFP-N1-IDH1 and pEGFP-N1-IDH1R132H plasmids. We thank Dr. Haining Zhu and Dr. Jozsef Gal, Department of Molecular and Cellular Biochemistry of the University of Kentucky, for providing the GFP-LC3 plasmid. We thank Dr. Daret St. Clair, Department of Toxicology, for her excellent suggestions involving MnSOD experiments. We also thank Drs. Jeremy Rich and Monica Venere from the Cleveland Clinic Foundation for their generous help and training with the patient-derived glioma cultures.
Conflict of interest
None of the authors have any conflicts of interest pertaining to the data in this study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gilbert, M.R., Liu, Y., Neltner, J. et al. Autophagy and oxidative stress in gliomas with IDH1 mutations. Acta Neuropathol 127, 221–233 (2014). https://doi.org/10.1007/s00401-013-1194-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-013-1194-6