Abstract
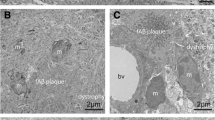
Subjects with Down syndrome (DS) inevitably develop histopathological features pathognomonic of Alzheimer’s disease (AD), and DS can therefore be considered a human model of AD. Similar to AD, microglial activation has been reported in DS and the idea that detrimental neuroinflammation plays a key role in the pathogenesis of neurodegeneration is firmly embedded. However, recent work from this laboratory has offered evidence for an alternative view regarding the role of microglial cells in AD pathogenesis by showing presence of dystrophic (senescent) rather than activated microglia in both the AD and DS brain. In this report, we build on previously published observations in human brain and offer a detailed analysis of microglial senescent pathology in the temporal cortices of 6 DS cases in their 40s, a critical age bracket where virtually all DS subjects acquire neurofibrillary degeneration characteristic of AD. Our findings using both Iba1 and anti-ferritin immunostaining of microglial cells show that coincident with the appearance of tau pathology in DS subjects there is consistent presence of dystrophic microglial cells and conspicuous absence of activated microglia using both markers. The extent of microglial pathology varied among the individual DS cases, but they all revealed decreased numbers of normal microglia ranging from 19 to 85% of the controls. Nearly all of the ferritin-positive microglia, which constitute a subset of the total Iba1-reactive microglial population, exhibited dystrophic morphology. In its most severe form dystrophy was evident as total fragmentation of the cells’ cytoplasm (cytorrhexis), which likely reflects terminal degeneration of microglia. Severely dystrophic, ferritin-positive cells were often found to be colocalized with tau-positive senile plaques. Our findings help to consolidate the idea that microglial degeneration and neurofibrillary degeneration are closely linked events in a human model of AD. They suggest that microglial degeneration follows a gradually progressive course that increases in its severity in parallel with the progression of AD neurodegenerative changes.







Similar content being viewed by others
References
Akiyama H, McGeer PL (1990) Brain microglia constitutively express beta-2 integrins. J Neuroimmunol 30:81–93
Boje KM, Arora PK (1992) Microglial-produced nitric oxide and reactive nitrogen oxides mediate neuronal cell death. Brain Res 587:250–256
Colton CA, Gilbert DL (1987) Production of superoxide anions by a CNS macrophage, the microglia. FEBS Lett 223:284–288
Connor JR, Menzies SL, St Martin SM, Mufson EJ (1990) Cellular distribution of transferrin, ferritin, and iron in normal and aged human brains. J Neurosci Res 27:595–611
Croisier E, Moran LB, Dexter DT, Pearce RK, Graeber MB (2005) Microglial inflammation in the parkinsonian substantia nigra: relationship to alpha-synuclein deposition. J Neuroinflammation 2:14
Dringen R (2005) Oxidative and antioxidative potential of brain microglial cells. Antioxid Redox Signal 7:1223–1233
Fendrick SE, Xue QS, Streit WJ (2007) Formation of multinucleated giant cells and microglial degeneration in rats expressing a mutant Cu/Zn superoxide dismutase gene. J Neuroinflammation 4:9
Fiala M, Lin J, Ringman J et al (2005) Ineffective phagocytosis of amyloid-beta by macrophages of Alzheimer’s disease patients. J Alzheimers Dis 7:221–232
Goos JD, Kester MI, Barkhof F et al (2009) Patients with Alzheimer disease with multiple microbleeds: relation with cerebrospinal fluid biomarkers and cognition. Stroke 40:3455–3460
Graeber MB (2010) Changing face of microglia. Science 330:783–788
Graeber MB, Streit WJ, Kreutzberg GW (1988) Axotomy of the rat facial nerve leads to increased CR3 complement receptor expression by activated microglial cells. J Neurosci Res 21:18–24
Griffin WS, Stanley LC, Ling C et al (1989) Brain interleukin 1 and S-100 immunoreactivity are elevated in Down syndrome and Alzheimer disease. Proc Natl Acad Sci USA 86:7611–7615
Grundke-Iqbal I, Fleming J, Tung YC et al (1990) Ferritin is a component of the neuritic (senile) plaque in Alzheimer dementia. Acta Neuropathol 81:105–110
Hayes A, Thaker U, Iwatsubo T, Pickering-Brown SM, Mann DM (2002) Pathological relationships between microglial cell activity and tau and amyloid beta protein in patients with Alzheimer’s disease. Neurosci Lett 331:171–174
Hayes GM, Woodroofe MN, Cuzner ML (1987) Microglia are the major cell type expressing MHC class II in human white matter. J Neurol Sci 80:25–37
Head E, Garzon-Rodriguez W, Johnson JK et al (2001) Oxidation of Abeta and plaque biogenesis in Alzheimer’s disease and Down syndrome. Neurobiol Dis 8:792–806
Hirrlinger J, Gutterer JM, Kussmaul L, Hamprecht B, Dringen R (2000) Microglial cells in culture express a prominent glutathione system for the defense against reactive oxygen species. Dev Neurosci 22:384–392
Hof PR, Bouras C, Perl DP et al (1995) Age-related distribution of neuropathologic changes in the cerebral cortex of patients with Down’s syndrome. Quantitative regional analysis and comparison with Alzheimer’s disease. Arch Neurol 52:379–391
Itagaki S, McGeer PL, Akiyama H, Zhu S, Selkoe D (1989) Relationship of microglia and astrocytes to amyloid deposits of Alzheimer disease. J Neuroimmunol 24:173–182
Ito D, Imai Y, Ohsawa K et al (1998) Microglia-specific localisation of a novel calcium binding protein, Iba1. Brain Res Mol Brain Res 57:1–9
Jellinger K, Paulus W, Grundke-Iqbal I, Riederer P, Youdim MB (1990) Brain iron and ferritin in Parkinson’s and Alzheimer’s diseases. J Neural Transm Park Dis Dement Sect 2:327–340
Kaneko Y, Kitamoto T, Tateishi J, Yamaguchi K (1989) Ferritin immunohistochemistry as a marker for microglia. Acta Neuropathol 79:129–136
Koeppen AH, Dentinger MP (1988) Brain hemosiderin and superficial siderosis of the central nervous system. J Neuropathol Exp Neurol 47:249–270
Lemstra AW, Groen in’t Woud JC, Hoozemans JJ et al (2007) Microglia activation in sepsis: a case-control study. J Neuroinflammation 4:4
Lopes KO, Sparks DL, Streit WJ (2008) Microglial dystrophy in the aged and Alzheimer’s disease brain is associated with ferritin immunoreactivity. Glia 56:1048–1060
Lott IT, Head E (2005) Alzheimer disease and Down syndrome: factors in pathogenesis. Neurobiol Aging 26:383–389
Lynch T, Cherny RA, Bush AI (2000) Oxidative processes in Alzheimer’s disease: the role of abeta-metal interactions. Exp Gerontol 35:445–451
Mann DM, Esiri MM (1989) The pattern of acquisition of plaques and tangles in the brains of patients under 50 years of age with Down’s syndrome. J Neurol Sci 89:169–179
Mann DM, Iwatsubo T, Fukumoto H et al (1995) Microglial cells and amyloid beta protein (A beta) deposition; association with A beta 40-containing plaques. Acta Neuropathol 90:472–477
Masliah E, Mallory M, Hansen L et al (1991) Immunoreactivity of CD45, a protein phosphotyrosine phosphatase, in Alzheimer’s disease. Acta Neuropathol 83:12–20
Mattiace LA, Davies P, Dickson DW (1990) Detection of HLA-DR on microglia in the human brain is a function of both clinical and technical factors. Am J Pathol 136:1101–1114
McGeer PL, Itagaki S, Tago H, McGeer EG (1987) Reactive microglia in patients with senile dementia of the Alzheimer type are positive for the histocompatibility glycoprotein HLA-DR. Neurosci Lett 79:195–200
Meda L, Cassatella MA, Szendrei GI et al (1995) Activation of microglial cells by beta-amyloid protein and interferon-gamma. Nature 374:647–650
Njie EG, Boelen E, Stassen FR et al (2010) Ex vivo cultures of microglia from young and aged rodent brain reveal age-related changes in microglial function. Neurobiol Aging [Epub ahead of print]
Perlmutter LS, Barron E, Chui HC (1990) Morphologic association between microglia and senile plaque amyloid in Alzheimer’s disease. Neurosci Lett 119:32–36
Power JH, Blumbergs PC (2009) Cellular glutathione peroxidase in human brain: cellular distribution, and its potential role in the degradation of Lewy bodies in Parkinson’s disease and dementia with Lewy bodies. Acta Neuropathol 117:63–73
Rozemuller AJ, van Gool WA, Eikelenboom P (2005) The neuroinflammatory response in plaques and amyloid angiopathy in Alzheimer’s disease: therapeutic implications. Curr Drug Targets CNS Neurol Disord 4:223–233
Simmons DA, Casale M, Alcon B et al (2007) Ferritin accumulation in dystrophic microglia is an early event in the development of Huntington’s disease. Glia 55:1074–1084
Smith MA, Zhu X, Tabaton M et al (2010) Increased iron and free radical generation in preclinical Alzheimer disease and mild cognitive impairment. J Alzheimers Dis 19:363–372
Stoltzner SE, Grenfell TJ, Mori C et al (2000) Temporal accrual of complement proteins in amyloid plaques in Down’s syndrome with Alzheimer’s disease. Am J Pathol 156:489–499
Streit WJ, Braak H, Xue QS, Bechmann I (2009) Dystrophic (senescent) rather than activated microglial cells are associated with tau pathology and likely precede neurodegeneration in Alzheimer’s disease. Acta Neuropathol 118:475–485
Streit WJ, Sammons NW, Kuhns AJ, Sparks DL (2004) Dystrophic microglia in the aging human brain. Glia 45:208–212
Streit WJ, Xue QS (2009) Life and death of microglia. J Neuroimmune Pharmacol 4:371–379
Tchaikovskaya T, Fraifeld V, Urphanishvili T et al (2005) Glutathione S-transferase hGSTM3 and ageing-associated neurodegeneration: relationship to Alzheimer’s disease. Mech Ageing Dev 126:309–315
Verina T, Kiihl SF, Schneider JS, Guilarte TR (2011) Manganese exposure induces microglia activation and dystrophy in the substantia nigra of non-human primates. Neurotoxicology 32:215–226
Wisniewski KE, Wisniewski HM, Wen GY (1985) Occurrence of neuropathological changes and dementia of Alzheimer’s disease in Down’s syndrome. Ann Neurol 17:278–282
Xue QS, Yang C, Hoffman PM, Streit WJ (2010) Microglial response to murine leukemia virus-induced encephalopathy is a good indicator of neuronal perturbations. Brain Res 1319:131–141
Acknowledgments
Supported by NIH grant AG023665.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xue, QS., Streit, W.J. Microglial pathology in Down syndrome. Acta Neuropathol 122, 455–466 (2011). https://doi.org/10.1007/s00401-011-0864-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-011-0864-5