Abstract
Purpose
A high-fat diet (HFD) induces gut microbiota (GM) disorders, leading to intestinal barrier dysfunction and inflammation. Ferulic acid (FA) has shown anti-obesity effects, e.g., reducing body weight and food intake. However, the mechanism linking the anti-obesity effects of FA and GM modulation remains obscure. The present study aimed to clarify the mechanism underlying the anti-obesity effects of FA and modulation of the GM.
Methods
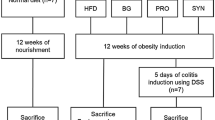
C57BL/6 J mice were fed by a low-fat diet (LFD) and HFD with or without FA at a dose of 100 mg/kg of body weight by oral gavage for 12 weeks. Using high-throughput sequencing, gas chromatography, real-time fluorescence quantitative PCR and immunohistochemical staining, the attenuation of obesity by FA were assessed via intestinal barrier integrity, inflammation, and the GM.
Results
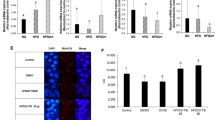
FA reduced weight gain, improved HFD-induced GM imbalance, significantly enhanced intestinal short-chain fatty acid (SCFA)-producing bacteria (e.g., Olsenella, Eisenbergiella, Dubosiella, Clostridiales_unclassified, and Faecalibaculum) along with SCFA accumulation and its receptors’ expression, decreased endotoxin-producing bacteria or obesity-related bacterial genera, and serum endotoxin (lipopolysaccharides), and inhibited the colonic TLR4/NF-κB pathway. Thus, FA can mitigate colonic barrier dysfunction and intestinal inflammation, induce the production of SCFAs and inhibit endotoxins by modulating the GM.
Conclusion
These results indicate that enhancement of intestinal barrier by altering the GM may be an anti-obesity target of FA and that FA can be used as a functional compound with great developmental values.
Graphical Abstract








Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- FA:
-
Ferulic acid
- GM:
-
Gut microbiota
- HFD:
-
High-fat diet
- IECs:
-
Intestinal epithelial cells
- LEfSe:
-
Linear discriminant analysis effect size analysis
- LPS:
-
Lipopolysaccharide
- NF-κB:
-
Nuclear factor Kappa-B
- OTUs:
-
Operational taxonomic units
- SCFA:
-
Short-chain fatty acid
- TJs:
-
Tight junctions
References
Gao X, Chang S, Liu S, Peng L, Xie J, Dong W, Tian Y, Sheng J (2020) Correlations between α-linolenic acid-improved multitissue homeostasis and gut microbiota in mice fed a high-fat diet. mSystems 5:e00391-e1320. https://doi.org/10.1128/mSystems.00391-20
Sun L, Ma L, Ma Y, Zhang F, Zhao C, Nie Y (2018) Insights into the role of gut microbiota in obesity: pathogenesis, mechanisms, and therapeutic perspectives. Protein Cell 9:397–403. https://doi.org/10.1007/s13238-018-0546-3
Miyamoto J, Igarashi M, Watanabe K, Karaki S-i, Mukouyama H, Kishino S, Li X, Ichimura A, Irie J, Sugimoto Y, Mizutani T, Sugawara T, Miki T, Ogawa J, Drucker DJ, Arita M, Itoh H, Kimura I (2019) Gut microbiota confers host resistance to obesity by metabolizing dietary polyunsaturated fatty acids. Nat Commun 10:4007. https://doi.org/10.1038/s41467-019-11978-0
Dalby MJ, Ross AW, Walker AW, Morgan PJ (2017) Dietary uncoupling of gut microbiota and energy harvesting from obesity and glucose tolerance in mice. Cell Rep 21:1521–1533. https://doi.org/10.1016/j.celrep.2017.10.056
Rohr MW, Narasimhulu CA, Rudeski-Rohr TA, Parthasarathy S (2020) Negative effects of a high-fat diet on intestinal permeability: a review. Adv Nutr 11:77–91. https://doi.org/10.1093/advances/nmz061
Dao MC, Clément K (2018) Gut microbiota and obesity: concepts relevant to clinical care. Eur J Intern Med 48:18–24. https://doi.org/10.1016/j.ejim.2017.10.005
Kumar N, Pruthi V (2014) Potential applications of ferulic acid from natural sources. Biotechnol Rep 4:86–93. https://doi.org/10.1016/j.btre.2014.09.002
Byrd AL, Liu M, Fujimura KE, Lyalina S, Nagarkar DR, Charbit B, Bergstedt J, Patin E, Harrison OJ, Quintana-Murci L, Mellman I, Duffy D, Albert ML, Consortium tMI (2020) Gut microbiome stability and dynamics in healthy donors and patients with non-gastrointestinal cancers. J Exp Med. https://doi.org/10.1084/jem.20200606
Wang W, Pan Y, Wang L, Zhou H, Song G, Wang Y, Liu J, Li A (2019) Optimal dietary ferulic acid for suppressing the obesity-related disorders in leptin-deficient obese C57BL/6J-ob/ob mice. J Agric Food Chem 67:4250–4258. https://doi.org/10.1021/acs.jafc.8b06760
Zhao Z, Moghadasian MH (2008) Chemistry, natural sources, dietary intake and pharmacokinetic properties of ferulic acid: a review. Food Chem 109:691–702. https://doi.org/10.1016/j.foodchem.2008.02.039
Mancuso C, Santangelo R (2014) Ferulic acid: pharmacological and toxicological aspects. Food Chem Toxicol 65:185–195. https://doi.org/10.1016/j.fct.2013.12.024
Luna-Vital D, Luzardo-Ocampo I, Cuellar-Nuñez ML, Loarca-Piña G, Gonzalez de Mejia E (2020) Maize extract rich in ferulic acid and anthocyanins prevents high-fat-induced obesity in mice by modulating SIRT1, AMPK and IL-6 associated metabolic and inflammatory pathways. J Nutr Biochem 79:108343. https://doi.org/10.1016/j.jnutbio.2020.108343
Naowaboot J, Piyabhan P, Munkong N, Parklak W, Pannangpetch P (2016) Ferulic acid improves lipid and glucose homeostasis in high-fat diet-induced obese mice. Clin Exp Pharmacol Physiol 43:242–250. https://doi.org/10.1111/1440-1681.12514
Salazar-López NJ, Astiazarán-García H, González-Aguilar GA, Loarca-Piña G, Ezquerra-Brauer J-M, Domínguez Avila JA, Robles-Sánchez M (2017) Ferulic acid on glucose dysregulation, dyslipidemia, and inflammation in diet-induced obese rats: an integrated study. Nutrients 9:675. https://doi.org/10.3390/nu9070675
Song Y, Wu M-s, Tao G, Lu M-w, Lin J, Huang J-q (2020) Feruloylated oligosaccharides and ferulic acid alter gut microbiome to alleviate diabetic syndrome. Food Res Int 137:109410. https://doi.org/10.1016/j.foodres.2020.109410
Tian B, Zhao J, Zhang M, Chen Z, Ma Q, Liu H, Nie C, Zhang Z, An W, Li J (2021) Lycium ruthenicum anthocyanins attenuate high-fat diet-induced colonic barrier dysfunction and inflammation in mice by modulating the gut microbiota. Mol Nutr Food Res 65:2000745. https://doi.org/10.1002/mnfr.202000745
Tian B, Geng Y, Xu T, Zou X, Mao R, Pi X, Wu W, Huang L, Yang K, Zeng X, Sun P (2022) Digestive characteristics of Hericium erinaceus polysaccharides and their positive effects on fecal microbiota of male and female volunteers during in vitro fermentation. Front Nutr. https://doi.org/10.3389/fnut.2022.858585
Zhao B, Xia B, Li X, Zhang L, Liu X, Shi R, Kou R, Liu Z, Liu X (2020) Sesamol supplementation attenuates DSS-induced colitis via mediating gut barrier integrity, inflammatory responses, and reshaping gut microbiome. J Agric Food Chem 68:10697–10708. https://doi.org/10.1021/acs.jafc.0c04370
Tian B, Liu M, An W, Yu L, Zhang J, Liu Y, Zhao J, Li J (2020) Lycium barbarum relieves gut microbiota dysbiosis and improves colonic barrier function in mice following antibiotic perturbation. J Funct Foods 71:103973. https://doi.org/10.1016/j.jff.2020.103973
Poquet L, Clifford MN, Williamson G (2008) Transport and metabolism of ferulic acid through the colonic epithelium. Drug Metab Dispos 36:190–197. https://doi.org/10.1124/dmd.107.017558
Zhao Z, Egashira Y, Sanada H (2003) Digestion and absorption of ferulic acid sugar esters in rat gastrointestinal tract. J Agric Food Chem 51:5534–5539. https://doi.org/10.1021/jf034455u
Zhang Z, Yang P, Zhao J (2022) Ferulic acid mediates prebiotic responses of cereal-derived arabinoxylans on host health. Anim Nutr 9:31–38. https://doi.org/10.1016/j.aninu.2021.08.004
Vitaglione P, Mennella I, Ferracane R, Rivellese AA, Giacco R, Ercolini D, Gibbons SM, La Storia A, Gilbert JA, Jonnalagadda S, Thielecke F, Gallo MA, Scalfi L, Fogliano V (2015) Whole-grain wheat consumption reduces inflammation in a randomized controlled trial on overweight and obese subjects with unhealthy dietary and lifestyle behaviors: role of polyphenols bound to cereal dietary fiber. Am J Clin Nutr 101:251–261. https://doi.org/10.3945/ajcn.114.088120
Ma Y, Chen K, Lv L, Wu S, Guo Z (2019) Ferulic acid ameliorates nonalcoholic fatty liver disease and modulates the gut microbiota composition in high-fat diet fed ApoE−/− mice. Biomed pharmacother 113:108753–108753. https://doi.org/10.1016/j.biopha.2019.108753
Ohwada K (1992) Body surface area of the golden Syrian hamster. Exp Anim 41:221–224. https://doi.org/10.1538/expanim1978.41.2_221
Reid G, Abrahamsson T, Bailey M, Bindels LB, Bubnov R, Ganguli K, Martoni C, O’Neill C, Savignac HM, Stanton C, Ship N, Surette M, Tuohy K, van Hemert S (2017) How do probiotics and prebiotics function at distant sites? Benefic Microbes 8:521–533. https://doi.org/10.3920/bm2016.0222
Bubnov RV, Babenko LP, Lazarenko LM, Mokrozub VV, Spivak MY (2018) Specific properties of probiotic strains: relevance and benefits for the host. EPMA J 9:205–223. https://doi.org/10.1007/s13167-018-0132-z
Le Chatelier E, Nielsen T, Qin J, Prifti E, Hildebrand F, Falony G, Almeida M, Arumugam M, Batto J-M, Kennedy S (2013) Richness of human gut microbiome correlates with metabolic markers. Nature 500:541. https://doi.org/10.1038/nature12506
Yan S, Shi R, Li L, Ma S, Zhang H, Ye J, Wang J, Pan J, Wang Q, Jin X, Liu X, Liu Z (2019) Mannan oligosaccharide suppresses lipid accumulation and appetite in western-diet-induced obese mice via reshaping gut microbiome and enhancing short-chain fatty acids production. Mol Nutr Food Res 63:1900521. https://doi.org/10.1002/mnfr.201900521
Bagarolli RA, Tobar N, Oliveira AG, Araújo TG, Carvalho BM, Rocha GZ, Vecina JF, Calisto K, Guadagnini D, Prada PO (2017) Probiotics modulate gut microbiota and improve insulin sensitivity in DIO mice. J Nutr Biochem 50:16–25. https://doi.org/10.1016/j.jnutbio.2017.08.006
Magne F, Gotteland M, Gauthier L, Zazueta A, Pesoa S, Navarrete P, Balamurugan R (2020) The firmicutes/bacteroidetes ratio: a relevant marker of gut dysbiosis in obese patients? Nutrients. https://doi.org/10.3390/nu12051474
Mao B, Li D, Zhao J, Liu X, Gu Z, Chen YQ, Zhang H, Chen W (2015) Metagenomic insights into the effects of fructo-oligosaccharides (FOS) on the composition of fecal microbiota in mice. J Agric Food Chem 63:856–863. https://doi.org/10.1021/jf505156h
Amir I, Bouvet P, Legeay C, Gophna U, Weinberger A (2014) Eisenbergiella tayi gen. nov., sp. nov., isolated from human blood. Int J Syst Evol Microbiol 64:907–914. https://doi.org/10.1099/ijs.0.057331-0
Guo W-L, Guo J-B, Liu B-Y, Lu J-Q, Chen M, Liu B, Bai W-D, Rao P-F, Ni L, Lv X-C (2020) Ganoderic acid A from Ganoderma lucidum ameliorates lipid metabolism and alters gut microbiota composition in hyperlipidemic mice fed a high-fat diet. Food Funct 11:6818–6833. https://doi.org/10.1039/D0FO00436G
Li L-L, Wang Y-T, Zhu L-M, Liu Z-Y, Ye C-Q, Qin S (2020) Inulin with different degrees of polymerization protects against diet-induced endotoxemia and inflammation in association with gut microbiota regulation in mice. Sci Rep 10:978. https://doi.org/10.1038/s41598-020-58048-w
Kameyama K, Itoh K (2014) Intestinal colonization by a Lachnospiraceae Bacterium contributes to the development of diabetes in obese mice. Microbes Environ 29:427–430. https://doi.org/10.1264/jsme2.ME14054
Wang P, Gao J, Ke W, Wang J, Li D, Liu R, Jia Y, Wang X, Chen X, Chen F, Hu X (2020) Resveratrol reduces obesity in high-fat diet-fed mice via modulating the composition and metabolic function of the gut microbiota. Free Radic Biol Med 156:83–98. https://doi.org/10.1016/j.freeradbiomed.2020.04.013
Hou D, Zhao Q, Yousaf L, Khan J, Xue Y, Shen Q (2020) Consumption of mung bean (Vigna radiata L.) attenuates obesity, ameliorates lipid metabolic disorders and modifies the gut microbiota composition in mice fed a high-fat diet. J Funct Foods 64:103687. https://doi.org/10.1016/j.jff.2019.103687
Wang K, Jin X, Li Q, Sawaya ACHF, Le Leu RK, Conlon MA, Wu L, Hu F (2018) Propolis from different geographic origins decreases intestinal inflammation and Bacteroides spp populations in a model of DSS-induced colitis. Mol Nutr Food Res. https://doi.org/10.1002/mnfr.201800080
Wan Y, Wang F, Yuan J, Li J, Jiang D, Zhang J, Li H, Wang R, Tang J, Huang T, Zheng J, Sinclair AJ, Mann J, Li D (2019) Effects of dietary fat on gut microbiota and faecal metabolites, and their relationship with cardiometabolic risk factors: a 6-month randomised controlled-feeding trial. Gut 68:1417. https://doi.org/10.1136/gutjnl-2018-317609
Friedrich V, Forné I, Matzek D, Ring D, Popper B, Jochum L, Spriewald S, Straub T, Imhof A, Krug A, Stecher B, Brocker T (2021) Helicobacter hepaticus is required for immune targeting of bacterial heat shock protein 60 and fatal colitis in mice. Gut Microbes 13:1882928. https://doi.org/10.1080/19490976.2021.1882928
Leonard W, Zhang P, Ying D, Fang Z (2021) Hydroxycinnamic acids on gut microbiota and health. Compr Rev Food Sci Food Saf 20:710–737. https://doi.org/10.1111/1541-4337.12663
Arnolds K, Lozupone C (2016) Striking a balance with help from our little friends—how the gut microbiota contributes to immune homeostasis. Yale J Biol Med 89:389–395. www.ncbi.nlm.nih.gov/pmc/journals/504/
Sanna S, van Zuydam NR, Mahajan A, Kurilshikov A, Vila AV, Võsa U, Mujagic Z, Masclee AA, Jonkers DM, Oosting M (2019) Causal relationships among the gut microbiome, short-chain fatty acids and metabolic diseases. Nat Genet 51:600. https://doi.org/10.1038/s41588-019-0350-x
Gribble FM, Reimann F (2019) Function and mechanisms of enteroendocrine cells and gut hormones in metabolism. Nat Rev Endocrinol 15:226–237. https://doi.org/10.1038/s41574-019-0168-8
Zhang X-S, Li J, Krautkramer KA, Badri M, Battaglia T, Borbet TC, Koh H, Ng S, Sibley RA, Li Y (2018) Antibiotic-induced acceleration of type 1 diabetes alters maturation of innate intestinal immunity. Elife 7:e37816. https://doi.org/10.7554/eLife.37816
Lagkouvardos I, Lesker TR, Hitch TC, Gálvez EJ, Smit N, Neuhaus K, Wang J, Baines JF, Abt B, Stecher B (2019) Sequence and cultivation study of Muribaculaceae reveals novel species, host preference, and functional potential of this yet undescribed family. Microbiome 7:28. https://doi.org/10.1186/s40168-019-0637-2
Tian B, Pan Y, Wang J, Cai M, Ye B, Yang K, Sun P (2022) Insoluble dietary fibers from by-products of edible fungi industry: basic structure, physicochemical properties, and their effects on energy intake. Front Nutr. https://doi.org/10.3389/fnut.2022.851228
Hernández MAG, Canfora EE, Jocken JWE, Blaak EE (2019) The short-chain fatty acid acetate in body weight control and insulin sensitivity. Nutrients 11:1943. https://doi.org/10.3390/nu11081943
Kimura I, Ozawa K, Inoue D, Imamura T, Kimura K, Maeda T, Terasawa K, Kashihara D, Hirano K, Tani T (2013) The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nat commun 4:1829. https://doi.org/10.1038/ncomms2852
Bernardi S, Del Bo’ C, Marino M, Gargari G, Cherubini A, Andrés-Lacueva C, Hidalgo-Liberona N, Peron G, González-Dominguez R, Kroon P, Kirkup B, Porrini M, Guglielmetti S, Riso P (2020) Polyphenols and Intestinal Permeability: Rationale and Future Perspectives. J Agric Food Chem 68:1816–1829. https://doi.org/10.1021/acs.jafc.9b02283
Lin MY, de Zoete MR, van Putten JP, Strijbis K (2015) Redirection of epithelial immune responses by short-chain fatty acids through inhibition of histone deacetylases. Front Immunol 6:554. https://doi.org/10.3389/fimmu.2015.00554
Wang D, Sun L, Liu X, Niu Z, Chen S, Tang L, Zheng H, Chen X, Li H, Lu L, Malik V, Lin X (2020) Replacing white rice bars with peanuts as snacks in the habitual diet improves metabolic syndrome risk among Chinese adults: a randomized controlled trial. Am J Clin Nutr 113:28–35. https://doi.org/10.1093/ajcn/nqaa307
Guo S, Nighot M, Al-Sadi R, Alhmoud T, Nighot P, Ma TY (2015) Lipopolysaccharide regulation of intestinal tight junction permeability is mediated by TLR4 signal transduction pathway activation of FAK and MyD88. J Immunol 195:4999–5010. https://doi.org/10.4049/jimmunol.1402598
Gong J, Hu M, Huang Z, Fang K, Wang D, Chen Q, Li J, Yang D, Zou X, Xu L (2017) Berberine attenuates intestinal mucosal barrier dysfunction in type 2 diabetic rats. Front Pharmacol 8:42. https://doi.org/10.3389/fphar.2017.00042
Acknowledgements
This work was sponsored by Zhejiang Provincial Key Research and Development Program (2022C04036, 2019C02100, 2019C02074 and 2019C02040) and the Cooperative Project Fund of the Zhejiang University of Technology and Zhejiang Institute of Modern TCM and Natural Medicine Co., Ltd. (KYY-HX-20211132). We sincerely thank Zhixin Luo (Northwest A&F University) for animal feeding and management.
Author information
Authors and Affiliations
Contributions
Conceptualization, BT and YG; methodology, BT and PW; software, MC and JH; validation and formal analysis, BT and DX; data curation, BT and WC; writing-original draft preparation, BT; writing-review and editing, JN and KY; project administration, BT and PS; and funding acquisition, KY.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
394_2022_2927_MOESM1_ESM.docx
Supplementary Information The e-component of this article contains supplementary materials in several parts: 1) Primer sequences used for real-time quantitative PCR; 2)The average body weight changes of mice; 3)The feed intake curve of mice; 4) Alpha diversity analysis of intestinal microbiota in mice; 5) The gut microbial taxonomic profiling at the phylum and genus levels (DOCX 735 KB)
Rights and permissions
About this article
Cite this article
Tian, B., Geng, Y., Wang, P. et al. Ferulic acid improves intestinal barrier function through altering gut microbiota composition in high-fat diet-induced mice. Eur J Nutr 61, 3767–3783 (2022). https://doi.org/10.1007/s00394-022-02927-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-022-02927-7