Abstract
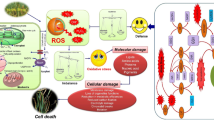
Hyperhydric disorders occur frequently in plant tissues cultured in vitro and cause several morphological and physiological abnormalities. However, a systematic defense response is triggered by hyperhydric conditions. The accumulation of reactive oxygen species (ROS), activities of antioxidant enzymes and their immunoblots, and the proteome-level changes in normal versus hyperhydric shoots of carnation (Dianthus caryophyllus) cultured in vitro were investigated. Total proteins were also extracted from the shoot and analyzed by two-dimensional electrophoresis. Among a total of 700 spots detected, only 40 had significant changes in abundance in the hyperhydric compared to the normal shoots, which were further identified by a mass spectrometer (MALDI-TOF MS). Most of them were involved in photosynthesis, RNA processing, and general metabolisms, while the rest were involved in secondary metabolic processes. These identified proteins in carnation shoots may provide novel evidences for stress tolerance against hyperhydricity.






Similar content being viewed by others
Abbreviations
- 2-DE:
-
Second-dimensional gel electrophoresis
- IEF:
-
Isoelectric focusing
- IPG:
-
Immobilized pressure gradient
- MALDI-TOF MS:
-
Matrix-assisted laser desorption/ionization time of flight mass spectrometer
- SDS-PAGE:
-
Sodium dodecyl sulfate polyacrylamide gel electrophoresis
- ACN:
-
Acetonitrile
- BSA:
-
Bovine serum albumin
- CHAPS:
-
3-[(3-Cholamidopropyl) dimethylammonio]-1-propanesulfonate
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Ahsan N, Renault J, Komatsu S (2009) Recent developments in the application of proteomics to the analysis of plant responses to heavy metals. Proteomics 9:2602–2621
Balen B, Tkalec M, Pavokovic´ D, Pevalek-Kozlina B, Krsnik-Rasol M (2009) Growth conditions in in vitro culture can induce oxidative stress in Mammillaria gracilis tissues. J Plant Growth Regul 28:36–45
Bardford MM (1976) A rapid and sensitive method for quantification of microgram quantities of proteins utilizing the principal of protein-dye binding. Anal Biochem 72:248–254
Cao S, Jiang L, Song S, Jing R, Xu G (2006) AtGRP7 is involved in the regulation of abscisic acid and stress responses in Arabidopsis. Cell Mol Biol Lett 11:526–535
Cassells AC, Curry RF (2001) Oxidative stress and physiological, epigenetic and genetic variability in plant tissue culture: implications for micropropagators and genetic engineers. Plant Cell Tissue Organ Cult 64:145–157
Chakrabarty D, Park SY, Ali MB, Shin KS, Paek KY (2005) Hyperhydricity in apple: ultrastructural and physiological aspects. Tree Physiol 26:377–388
Chen J, Ziv M (2001) The effect of ancymidol on hyperhydricity, regeneration, starch and antioxidant enzymatic activities in liquid-cultured Narcissus. Plant Cell Rep 20:22–27
Correl MJ, Wu Y, Weathers PJ (2001) Controlling hyperhydratation of carnations (Dianthus caryophyllus L.) grown in a mist reactor. Biotech Bioeng 71:307–314
Dewir YH, Chakrabarty D, Ali MB, Hahn EJ, Paek KY (2006) Lipid peroxidation and antioxidant enzyme activities of Euphorbia millii hyperhydric shoots. Environ Exp Bot 58:93–99
Dhindsa RH, Plumb-Dhindsa P, Thorpe TA (1981) Leaf senescence correlated with increased level of membrane permeability, lipid peroxidation and decreased level of SOD and CAT. J Exp Bot 32:93–101
Eat Picoli, Otoni WC, Figueira ML, Carolino SMSB, Almeida RS, Silva EAM, Carvalho CR, Fontes EPB (2001) Hyperhydricity in in vitro eggplant regenerated shoots: structural characteristics and involvement if BiP (Binding protein). Plant Sci 160:857–868
Fernandez-Garcı´a N, Piqueras A, Olmos E (2008) Sub-cellular location of H2O2, peroxidases and pectin epitopes in control and hyperhydric shoots of carnation. Environ Exp Bot 62:168–175
Fontes MA, Otoni WC, Carolino SMB, Brommonschenkel SH, Fontes EPB, Fári M, Louro RP (1999) Hyperhydricity in pepper plants regenerated in vitro: involvement of BiP (Binding protein) and ultrastructural aspects Plant Cell. Report 19:81–87
Franck T, Kevers C, Gaspar T (1995) Protective enzymatic systems against activated oxygen species compared in normal and vitrified shoots of Prunus avium L. raised in vitro. Plant Growth Regul 16:253–256
Franck T, Kevers C, Gaspar T, Dommes J, Deby C, Greimers R, Serteyn D, Deby-Dupont G (2004) Hyperhydricity of Prunus avium shoots cultured on gelrite: a controlled stress response. Plant Physiol Biochem 42:519–527
Galbally J, Galbally E (1997) Carnations and Pinks for Garden and Greenhouse. Timber Press, Portland
George EF (1996) Plant propagation by Tissue Culture, part 2. In: practice. Exegetics Ltd, Edington
Gribble K, Sarafis V, Conroy J (2003) Vitrified plants: towards an understanding of their nature. Phytomorphology 53:1–10
Harris EH, Boynton JE, Gillham NW (1994) Chloroplast ribosomes and protein synthesis. Microbiol Rev 58:700–754
Hassannejad S, Bernard F, Mirzajani F, Gholami M (2012) SA improvement of hyperhydricity reversion in Thymus daenensis shoots culture may be associated with polyamines changes. Plant Physiol Biochem 51:40–46
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplast I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Huang YC, Chiang CH, Li CM, Yu TA (2010) Transgenic watermelon lines expressing the nucleocapsid gene of Watermelon silver mottle virus and the role of thiamine in reducing hyperhydricity in regenerated shoots. Plant Cell Tissue Organ Cult 106:21–29
Jaspers P, Kangasjärvi J (2010) Reactive oxygen species in abiotic stress signaling. Physiol Plant 138:405–413
Kevers C, Coumans M, Coumans-Gilles F, Gaspar T (1984) Physiological and biochemical events leading to vitrification or shoots cultured in vitro. Physiol Plant 61:69–74
Lai CC, Lin HM, Nalawade SM, Fang W, Tsay HS (2005) Hyperhydricity in shoot cultures of Scrophularia yoshimurae can be effectively reduced by ventilation of culture vessels. J Plant Physiol 162:355–361
Leshem B (1983) Growth of carnation meristems in vitro: anatomical structure of abnormal plantlets and the effect of agar concentration in the medium on their formation. Ann Bot 52:413–415
Lorkovic ZJ (2009) Role of plant RNA-binding proteins in development, stress response and genome organization. Trends Plant Sci 14:229–236
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Mittler R, Vanderauwera S, Gollery M, Van Breusegem F (2004) The reactive oxygen gene network in plants. Trends Plant Sci 9:490–498
Mittler R, Vanderauwera S, Suzuki N, Miller G, Tognetti VB, Vandepoele K, Gollery M, Shulaev V, Breusegem FV (2011) ROS signaling: the new wave? Trends Plant Sci 16:300–309
Molassiotis AN, Diamantidis GC, Therios IN, Tsirakoglou V, Dimassi KN (2005) Oxidative stress, antioxidant activity and Fe(III)-chelate reductase activity of five Prunus rootstocks explants in response to Fe deficiency. Plant Growth Regul 46:69–78
Muneer S, Jeong BR (2015a) Proteomic analysis of salt-stress responsive proteins in roots of tomato (Lycopersicon esculentum L.) plants towards silicon efficiency. Plant Growth Regul 77(2):133–146
Muneer S, Jeong BR (2015b) Proteomic analysis provides new insights in phosphorus homeostasis subjected to Pi (inorganic phosphate) starvation in tomato Plants (Solanum lycopersicum L.). PLoS ONE 10(7):e0134103
Muneer S, Kim TH, Qureshi MI (2012) Fe modulates Cd-induced oxidative stress and the expression of stress responsive proteins in the nodules of Vigna radiata. Plant Grow Regul 68:421–433
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Nakano M, Nobuta K, Vemaraju K, Tej SS, Skogen JW, Meyers BC (2006) Plant MPSS databases: signature-based transcriptional resources for analyses of mRNA and small RNA. Nucleic Acids Res 34:731–735
Olmos E, HellÍn E (1998) Ultrastructural differences of hyperhydric and normal leaves from regenerated carnation plants. Sci Hort 75:91–101
Olmos S, Piqueras A, Martinez-Solano JR, Hellin E (1997) The subcellular localization of peroxidase and the implication of oxidative stress in hyperhydrated leaves of regenerated carnation shoots. Plant Sci 130:97–105
Pandey A, Choudhary MK, Bhushan D, Chattopadhyay A, Chakraborty S, Datta A, Chakroborty N (2006) The Nuclear proteome of chick pea (Cicer arietinum L.) reveals predicted and un-expected proteins. J Proteome Res 5(12):3301–3311
Perez-Tornero O, Egea J, Olmos E, Burgos L (2001) Control of hyperhydricity in micropropagated apricot cultivars. In vitro Cell Dev Biol Plant 37:250–254
Piqueras A, Han BH, Van Huylenbroeck JM, Debergh PC (1998) Effect of different environmental conditions in vitro on sucrose metabolism and antioxidant enzymatic activities in cultured shoots of Nicotiana tabacum L. Plant Growth Regul 25:5–10
Rakwal R, Agrawal GK (2003) Rice proteomics: current status and future prospectives. Electrophoresis 24:3378–3389
Ranieri A, Castagna A, Baldan B, Soldatini GF (2001) Iron deficiency differently affects peroxidase isoforms in sunflower. J Exp Bot 52:25–35
Rojas-Martinez L, Visser RG, de Klerk GJ (2010) The hyperhydricity syndrome: waterlogging of plant tissues as a major cause. Propag Ornam Plants 10:169–175
Saher S, Piqueras A, Hellin E, Olmos E (2004) Hyperhydricity in micropropagated carnation shoots: the role of oxidative stress. Physiol Plant 120:152–161
Saher S, Piqueras A, Hellin E, Olmos E (2005) Prevention of hyperhydricity in micropropagated carnation shoots by bottom cooling: implications of oxidative stress. Plant Cell Tissue Organ Cult 81:149–158
Shigeoka S, Ishikawa T, Tamoi M, Miyagawa Y, Takeda T, Yabuta Y, Yoshimura K (2002) Regulation and function of ascorbate peroxidase isoenzymes. J Exp Bot 53:1305–1319
Song XS, Tiao CL, Shi K, Mao WH, Ogweno JO, Zhou YH, Yu JQ (2006) The response of antioxidant enzymes in cellular organelles in cucumber (Cucumis sativus L.) leaves to methyl viologen-induced photo-oxidative stress. Plant Growth Regul 49:85–93
Sreedhar RV, Venkatachalam L, Neelwarne B (2009) Hyperhydricityrelated morphologic and biochemical changes in Vanilla (Vanilla planifolia). J Plant Growth Regul 28:46–57
Subramanian AR (1993) Molecular genetics of chloroplast ribosomal proteins. Trends Biochem Sci 18:177–180
Tian J, Jiang F, Wu Z (2015) The apoplastic oxidative burst as a key factor of hyperhydricity in garlic plantlet in vitro. Plant Cell Tissue Org Cult 120:571–584
van den Dries N, GiannìS Czerednik A, Krens FA, de Klerk GJ (2013) Flooding of the apoplast is a key factor in the development of hyperhydricity. J Exp Bot 64:5221–5230
Wang YL, Wang XD, Zhao B, Wang YC (2007) Reduction of hyperhydricity in the culture of Lepidium meyenii shoots by the addition of rare earth elements. Plant Growth Regul 52:151–159
Wang CQ, Zhang YF, Zhang YB (2008) Scavenger enzyme activities in subcellular fractions of white clover (Trifolium repens L.) under PEG-induced water stress. J Plant Growth Regul 27:387–393
Wu Z, Chen LJ, Long YJ (2009) Analysis of ultrastructure and reactive oxygen species of hyperhydric garlic (Allium sativum L.) shoot. In vitro Cell Dev Biol-Plant 45:483–490
Yamaguchi K, Subramanian AR (2000) The plastid ribosomal proteins. Identification of all the proteins in the 50 S subunit of an organelle ribosome (chloroplast). J Biol Chem 275:28466–28482
Yamaguchi K, Subramanian AR (2003) Proteomic identification of all plastid-specific ribosomal proteins in higher plant chloroplast 30S ribosomal subunit. Eur J Biochem 270:190–205
Ziv M (1991) Vitrification: morphological and physiological disorders of in vitro shoots. In: Debergh PC, Zimmerman RH (eds) Micropropagation. Kluwer Academic Publishers, Dordrecht, pp 45–69
Acknowledgments
This work was supported from “Onsite Cooperative Agriculture Research” (Project no. PJ01090805) RDA, Republic of Korea. SM and PS were supported by BK21 Plus program (Brain Korea 21), Ministry of Education, Republic of Korea.
Author Contributions
SM and BRJ designed the experiments, SM did experiments and analyzed the data, PS cultured and took care of plants, SM and BRJ wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Muneer, S., Soundararajan, P. & Jeong, B.R. Proteomic and Antioxidant Analysis Elucidates the Underlying Mechanism of Tolerance to Hyperhydricity Stress in In Vitro Shoot Cultures of Dianthus caryophyllus . J Plant Growth Regul 35, 667–679 (2016). https://doi.org/10.1007/s00344-015-9569-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-015-9569-7