Abstract
Pelagic eggs and larvae of many coral reef fishes will encounter a dynamic and risky environment as they disperse between the reef and offshore habitats. Life-history theory predicts that spawning adults should synchronize their reproductive effort with specific environmental conditions that facilitate offspring survival. Favourable conditions for reproduction may be determined by local environmental conditions at the spawning site, or signalled by larger-scale environmental cues, such as the lunar cycle. Multiple cues may interact in complex ways to cause additional variation in spawning intensity. We evaluated a set of environmental variables that potentially determine temporal and spatial variation in spawning patterns of a highly iteroparous fish, the sixbar wrasse (Thalassoma hardwicke). Specifically, we monitored focal territories of terminal-phase males over a 5-month period, quantified spawning activities, and evaluated a hierarchical set of predictive models using a model selection approach (AICc). Temporal variation in spawning (and population densities at the spawning site) was most strongly associated with the lunar cycle and maximal around the new moon. Local hydrodynamic conditions and other environmental variables observable at the spawning site were less strongly correlated with temporal variation in spawning. Territory proximity to the reef edge was a strong predictor of spatial variation in spawning intensity; territories closest to the reef edge experienced more spawning. These observations suggest that females make predictable decisions about where and when they spawn. Females appear to recognize strong, persistent spatial gradients in spawning habitat quality, and primarily vary their spawning effort in accordance with a large-scale environmental cue (the lunar cycle).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reproductive timing and investment decisions exhibited by individuals of both sexes can be a strong determinant of fitness, and some reproductive strategies may outperform others (Lott 1991; Shapiro 1991). Reproductive success may be enhanced when individuals derive fitness benefits from specific environmental cues that indicate favourable conditions for reproduction (Gross 1996; Warner 1997; Brockman and Taborsky 2008). In marine systems, reproducing individuals commonly experience heterogeneous and dynamic environments that may vary greatly in their risks and rewards. Such species often respond to specific environmental cues that enable them to optimize their reproductive success. For example, many temperate fishes initiate reproduction in response to changes in water temperature, and this may lead to reproductive phenologies that improve food availability for their offspring (i.e. the ‘Match-Mismatch hypothesis’ of Cushing 1975). However, in tropical marine environments with less seasonal variation, many coral reef fishes spawn continuously throughout the year (exhibiting ‘extreme iteroparity’, Philippi and Seger 1989; Warner 1998; Wilbur and Rudolf 2006; Shima et al. 2018). Some researchers have equated the existence of extreme iteroparity in tropical reef fish species as an evolutionary response to an extremely unpredictable marine environment for offspring survival (e.g. ‘bet hedging strategy’, see Goodman, 1984; Einum and Fleming 2004; Hughes 2017). Others have hypothesized that adults may try to reduce the effect of density-dependent selection pressures on post-settlement offspring mortality, by spreading their reproductive effort through time (Shima et al. 2018). Alternatively, extreme iteroparity may simply be the best strategy when there is no clear advantage to spawning at one time or another.
Species that exhibit extreme iteroparity may be less reliant on external cues for reproductive investment decisions (Seger and Brockman 1987). Indeed, reproductive investment by many iteroparous, pelagic spawners appears to be relatively invariant in time and space (Warner 1995; Sancho et al. 2000; Claydon et al. 2014). Spawning aggregations of many of these species form on a daily basis and often during a particular time of day (Robertson 1983; Domeier and Colin 1997; Sancho et al. 2000; Claydon et al. 2014). Such spawning activities also commonly occur in particular locations, typically associated with geomorphological features such as the down-current edges of reef slopes (Randall and Randall 1963; Colin and Bell 1991; Warner 1995; Heyman and Kjerfve 2008; Claydon et al. 2014).
Many protogynous reef fishes have haremic social structures and may spawn in pairs and/or smaller groups (Warner and Hoffman 1980a, 1980b; Warner 1984; Kuwamura et al. 2009). Although reproduction is spread along a temporal continuum, spawning intensity may still vary through space and time (Johannes 1978; Robertson 1983; Petersen et al. 1992; Gust 2004; Claydon et al. 2014). For such species, the number of individuals at a spawning site can vary from several spawning pairs to hundreds of individuals over the course of a few days (Warner and Hoffman 1980a, 1980b; Warner 1995; Domeier and Colin 1997; Claydon et al. 2014). Spawning frequencies can also vary substantially between sites that are separated by only a few metres (Warner and Hoffman 1980a, 1980b; Warner 1984; Petersen et al. 1992). Consequently, even extremely iteroparous species may use environmental cues to make informed decisions about reproductive allocation in order to maximize their fitness.
A variety of hypotheses have been proposed to explain spatio-temporal variation in reproductive allocation of highly iteroparous reef fish. For example, fluctuations in spawning might be a plastic response to specific oceanographic conditions that promote survival of eggs and larvae through to settlement (Johannes 1978; Lobel 1978; Robertson et al. 1990; Claydon 2004). Such species may choose to spawn at times or places where local currents facilitate fertilization success, and/or rapid transport fertilized eggs away from reef-associated benthic and pelagic predators (Johannes 1978; Lobel 1978; Sancho et al. 2000). In apparent support of this hypothesis, studies have documented a positive correlation between spawning intensity and tidal-driven, outflowing currents, and/or increased current speeds that may facilitate a rapid dispersal of propagules into oceanic waters (Sancho et al. 2000; Claydon et al. 2014; Donahue et al. 2015).
Additionally, reproductive decisions may be influenced by risks associated with spawning (Warner 1998). Highly iteroparous individuals should normally prioritize their own survival over the slight fitness increases that may be gained from any individual reproductive bout, because these benefits may be greatly outweighed by the fitness gains from future reproduction. Consequently, highly iteroparous species may be more risk averse and should only intensify their spawning activities when conditions enhance their own survival, and conversely, they should avoid spawning in times or places of high predation risk. Environmental conditions that increase predation risk (e.g. low light levels, reduced visibility, or extremely exposed spawning territories) should be avoided (Robertson and Hoffman 1977; Shapiro et al. 1988; Warner 1998; Claydon et al. 2014).
However, because the consequences of local conditions on fitness may be difficult to predict (as they are too variable in time and space), spawning periodicity in iteroparous species may reflect a generalized adaptive response to an easily detectable, larger-scale environmental cue (Warner 1997). Environmental cues that prevail over a broad geographic range may not necessarily always maximize fitness; however, they may guarantee the highest average fitness for a population over a reproductive season. Many studies suggest that periodic spawning activities (across a wide range of taxa) are correlated with the lunar cycle—potentially because the lunar cycle represents such an easily observable, large-scale cue (Robertson 1983; Robertson et al. 1990; Zeller 1998; Claydon et al. 2014; Shima et al. 2020). The lunar state could simply serve as a convenient cue to facilitate synchronized reproduction (Lobel 1978; Colin and Clavijo 1988; Colin & Bell 1991; Claydon 2004) or to couple reproduction with the optimal timing of settlement (Robertson et al. 1988; Doherty et al. 1994; Shima et al. 2018). Additionally, spawning at particular portions of the lunar cycle may enhance dispersal of fertilized eggs (e.g. via tidal effects: Robertson et al. 1990; Claydon 2004; Claydon et al. 2014), and/or growth and survival of eggs and larvae in the offshore habitat (e.g. via effects related to moonlight: Shima and Swearer 2019; Shima et al 2020).
Here, we evaluate temporal and spatial variation in spawning patterns of the sixbar wrasse, Thalassoma hardwicke, in relation to broad and local scale environmental conditions. The sixbar wrasse is a small-bodied, protogynous reef fish. Some level of spawning is observable most days throughout the year and primarily concentrated during a 2-h period in the afternoon (Mitterwallner 2020). Socially dominant males establish mating territories at particular locations on a daily basis. We took advantage of the predictability of this system to address the following questions: (1) How does spawning intensity vary among days? (2) What are the potential environmental drivers of reproductive investment decisions? (3) To what extent are reproductive patterns associated with local cues (and their interactions), and to what extent are they influenced by larger-scale environmental cues like the lunar phase?
Methods
Study site and species
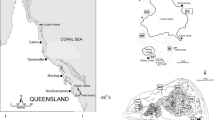
We surveyed spawning patterns of the sixbar wrasse (Thalassoma hardwicke; henceforth, sixbars) from February to June 2017, at two focal sites on the north shore of Moorea, French Polynesia. Sixbar wrasse are widely distributed throughout the Indo-Pacific region, and they are one of the most conspicuous and abundant reef fishes within inshore reef habitats of French Polynesia (Galzin 1987). Sixbar wrasse are protogynous hermaphrodites (Warner 1984a). Local populations contain a mix of initial phase (IP) females and males (which are morphologically indistinguishable from one another) and terminal-phase (TP) males (which are distinguishable by their brighter colour patterns and blunter heads). Sixbars are highly iteroparous, and they spawn throughout the lunar cycle and across much of the year (Claydon et al. 2014). Like other members of the genus (e.g. Thalassoma bifasciatum; Warner 1984a; Warner 1995), sixbars migrate to particular areas (e.g. reef edges) to spawn, with the largest females travelling as far as 400 m to reach spawning areas (Mitterwallner 2020). At these locations, TP males defend mating territories and actively court females. Spawners may engage in both pair spawning (i.e. one TP male and one female) and group spawning. IP males frequently attempt to ‘sneak’ mating opportunities during bouts of pair spawning, and they appear to participate openly in group spawning. On Moorea, most sixbar spawning activity is concentrated from 1300 to 1600 h daily, and consistently maximal from ~ 1400–1500 (P. Mitterwallner unpublished data).
Moorea is surrounded by a barrier reef crest that delineates the outermost extent of the lagoon system (spanning a width of ~ 1 km from reef crest to shore). Much of the lagoon is dominated by wide, flat areas (1–5 m depth) of sand and rubble, interspersed with patches of coral, algal turf, and extensive stands of macroalgae (Galzin and Pointier 1985). Water is forced into the lagoon by wave action and generally flows shoreward from the reef crest and then back out to sea through breaks (i.e. ‘passes’) in the reef crest (Galzin and Pointier 1985). Many species including sixbars spawn at the reef edge near these passes (P. Mitterwallner unpublished data).
Surveys of spawning activity
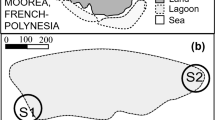
We conducted preliminary surveys to determine focal locations, durations, and times of day for subsequent spawning observations. Based on these initial surveys, we selected two focal sites (hereafter, S1 and S2) situated at the reef edge, near passes, on the north-eastern side of the island (see Fig. 1). Within each site, we identified and mapped three focal territories (T1, T2, T3) of terminal-phase males. These territories were distributed along a gradient from exposed (i.e. T1 closest to the reef edge) to sheltered conditions (i.e. T3 furthest from the reef edge; Fig. 1).
The two surveyed spawning sites (S1) and (S2) within a lagoon at the north shore of Moorea, French Polynesia (a, b). Within each site, we monitored focal territories (T1, T2, T3) of terminal-phase males (c). These territories were distributed along a gradient from exposed (i.e. T1 closest to the reef edge) to sheltered conditions (i.e. T3 furthest from the reef edge)
We surveyed focal territories in each site on alternate days. Each site was surveyed between 2 and 4 pm, ~ 3–5 times per week over 5 lunar cycles (i.e. 20 weeks). During each survey, a single observer (PM) recorded spawning activities within each focal TP territory (n = 3) over a 20-min duration. At each survey, focal territories were observed sequentially (i.e. over ~ 1 h), in randomized order, and during peak spawning activity. ‘Spawning’ was evidenced by female(s) and focal male(s) rising towards the surface and releasing eggs and sperm simultaneously. We estimated spawning intensity as the cumulative number of spawning events (i.e. pooling all pair spawns, group spawns, sneaking and streaking events). At the start of each survey, we estimated densities of fish at the spawning site by quantifying IP and TP fish along a single, permanent transect (100 m × 5 m width) centred on the spawning site. We surveyed this transect three times on each day of observation. We also recorded a set of 7 environmental variables during each observation period (detailed in Table 1).
Data analysis
We used a periodic regression (linear-circular regression) approach to model the cyclical nature of the lunar cycle and to evaluate primary sources of variation in sixbar spawning activity over time. Following methods of deBruyn and Meeuwig (2001), the calendar date of each observation was converted to a ‘lunar day’ (0–29.5; day 0 corresponds to the new moon, day 15 to the full moon, etc.). Lunar days were then divided into 360° (or 2π radians) to allocate each day an angular equivalent, theta (θ). The cyclical pattern of the lunar calendar was then expressed by sine and cosine transformations of theta. The cosine term describes a phase shift near 0° or 180° (full moon and new moon), and the sine term describes a phase shift between 90 and 270° (first and last quarter). A positive cosθ coefficient represents a peak at new moon, and a negative cosθ coefficient a peak at full moon. A significant positive sinθ coefficient corresponds to a peak during the first quarter, and a significant negative sin coefficient corresponds to a peak during the last quarter. The transformed data can then be analysed by using simple linear or linear mixed effect regression.
We used general linear mixed effect models (glmmTMB package, Brooks et al. 2017; R Core Team 2019) with a Poisson error distribution (because our observations consisted of counts) to evaluate the main sources of variation in (1) sixbar spawning activity and (2) population densities at the spawning site. Initially, we fitted a fully saturated model with all explanatory variables. The fully saturated model included the explanatory variables as fixed effects: ‘site’ (e.g. S1 or S2), ‘territory location (T1, T2, or T3), ‘sinθ, cosθ, ‘temperature’, ‘daytime cloud cover’, ‘night-time cloud cover’ (previous night), ‘visibility’, ‘current speed’, and ‘current direction’ (see Table 1 and Table 1A for details). The full model also included ‘Date’ (coded numerically) as a random effect. For spawning activities, we also included all possible two-way interactions between the local environmental variables ‘current speed’, ‘current direction’, ‘visibility’, and ‘cloud cover’ to evaluate whether co-occurrence of specific conditions might disproportionally influence spawning activities (e.g. do spawning frequencies significantly increase if outflowing currents co-occur with elevated current speeds?).
To account for potential temporal autocorrelation, we incorporated an AR(1) covariance structure in the model, as described in the glmmTMB vignette (Brooks et al. 2017). The covariance structure in the models was represented by ‘territory id’ as the random intercept and ‘date’ as grouping parameter to account for temporal autocorrelation between territories. This significantly improved the fit of the initial model based on p-value, AICc comparisons, and DHARMA (Hartig 2018) residual plots, suggesting that results were serially autocorrelated. To validate model suitability, we checked for uniformity of the residual quantiles (observed vs expected), as well as for overdispersion and outliers in the diagnostic plots, using the DHARMA package in R (Hartig 2018). In all cases the fits of the global models appeared satisfactory. We then used Akaike’s information criterion corrected for small sample sizes (AICc) to identify an appropriately reduced model (i.e. the candidate model with the lowest AICc; obtained via multimodel selection methods developed by the MuMIn package in R, see Bartón 2015). If the top set of models only varied slightly in their data fit, we performed a model averaging approach across all models with < 2 ΔAICc (the difference between a given model and the model with the lowest AICc), to generate more robust parameter estimates (Burnham and Anderson 2002). Model averaging was employed with the model.avg function of the MuMIn package in R (Bartón 2015). This function calculates a weighted average of each predictor variable appearing in the top candidate model set (i.e. all candidate models with ΔAICc < 2). Additionally, we calculated 95% confidence intervals for each predictor in the final averaged model, to determine whether an explanatory variable had a significant effect on the response variable (predictors with confidence intervals that overlap zero are usually not significant). The relative importance of a given predictor was then quantified as the sum of Akaike weights of each model (ΔAICc < 2) in which a particular predictor is represented (RVI > 0.9 = high importance; RVI > 0.6 = moderate importance; RVI < 0.6 = low importance; RVI < 0.5 = no importance, see Burnham and Anderson 2002).
To visualize the predicted effects of significant factors on the dependent variables, we additionally calculated estimated marginal means for each predictor, with covariates held at their mean value (function emmeans in the R package emmeans, see Lenth 2018).
Results
In total, we observed 649 bouts of spawning over a period of 150 days. On average, we recorded 3.2 spawning events per 20-min observation period, and spawning frequency varied substantially through time (Fig. 2).
Sources of variation in overall spawning frequency
Model selection yielded 15 models with a delta AICc < 2 for overall spawning frequencies as response variable (i.e. the total number of spawning events observed per 20-min observation period). The averaged model (i.e. the average over all models AICc < 2) retained moon phase (sinθ as well as the cosθ term), territory position, an interaction between cloud cover and current speed, current direction, visibility, and location as explanatory variables (see Table 2 and attachment Table A1).
However, confidence intervals as well as relative variable importance suggested that only moon phase and territory location (i.e. proximity to the reef edge) had a significant effect on overall spawning frequency (Fig. 3a, b and Table 2). A positive cosθ regression coefficient in the final model indicated significantly higher spawning activities around the new moon in comparison with full moon periods (0.867 ± 0.168 SE; 95% CI: 0.535, 1.198, RVI = 1.0). A marginally non-significant negative sinθ regression coefficient with an RVI of 0.8 suggested elevated activities of spawning during the third quarter (-0.266 ± 0.203 SE; 95% CI: -0.666, 0.134, RVI = 0.8). Together, these results suggest a steady increase of spawning activities from first quarter/full moon towards third quarter/new moon, with greatest activity peaks around the new moon period (roughly 6 times higher at new moon relative to full moon; Fig. 3a). Spawning intensity was also strongly influenced by territory position: exposed territories close to the reef edge exhibited significantly higher spawning frequencies relative to more sheltered territories (territory 3: -2.049 ± 0.231 SE; 95% CI: -2.505, -1.592, RVI = 1.0, 30–50% decrease in T3 and T2 relative to T1, Fig. 3b).
Sources of variation in overall sixbar spawning frequency through time. a Smoothed curve (± 95% confidence interval) of the lunar effect on predicted spawning frequency during the 29.5-day lunar cycle). b Variation in spawning among territories (T1 is closest to reef edge, T3 furthest from reef edge). Panels b gives marginal means back-transformed to the original scale (± SE) estimated with other fixed effects held at their mean values
Model averaging revealed that the local variables that were retained in the final model (i.e. interaction between cloud cover and current speed, visibility, current direction, and spawning location) had 95% confidence intervals that included zero, and a relative variable importance (RVI) of < 0.5 (see Table 2). This strongly suggests that local physical variables did not have a significant effect on variation of spawning frequencies at the spawning sites.
Population densities at the spawning site
Densities of sixbars at the spawning site ranged from 7.5 to 66 individuals/500 m2 per observation period. Linear regression analysis revealed that spawning activities were significantly positively correlated with population densities at the spawning site (F1,38 = 5.87, p = 0.02, Fig. 4a). Model averaging suggested that densities of sixbars that aggregated at the spawning sites were most strongly associated with the cosθ term of the final model, with highest population densities at new moon (0.317 ± 0.099 SE; 95% CI: 0.117, 0.517, RVI = 1.0; see Table 3 and attachment Table A2, Fig. 4b). Additionally, ‘spawning location’ was found to be an important term after model averaging, with higher population densities at spawning site S2 (0.467 ± 0.146 SE; 95% CI: 0.170, 0.763, RVI = 1.0; see Table 3). Apart from the ‘cosθ’ term and ‘spawning site’, ‘current direction’ and ‘sinθ’ remained in the final model. However, confidence intervals as well as RVI indicated that neither current direction nor sinθ proved to be important predictors of changes in population densities at the spawning sites (see Table 3).
a Smoothed curve (± 95% confidence interval) of the lunar effect on predicted spawning frequency during the 29.5-day lunar cycle. b Linear regression between population density at the local spawning site and number of spawns per 20 min. P-values and regression coefficient indicate a significant positive relationship
Discussion
Sixbar wrasse spawned on nearly every day of our 150d observation period, but the intensity of spawning varied strongly with the lunar cycle. Spawning was maximal around the new moon and greatest at male territories closest to the reef edge. Other abiotic variables were uncorrelated or weakly correlated with spawning intensity.
Lunar periodicity
Potential drivers of lunar periodicity: offspring survival
Many reef fishes exhibit lunar periodicity in spawning activities, with spawning particularly common around the new and/or full moon (Johannes 1978; Takemura et al. 2004; Claydon et al. 2014). In situations where the environments of offspring may be rapidly changing or difficult to predict, a generalized response to an easily detectable large-scale cue like the lunar state may provide several selective advantages for spawners (Warner 1997). For sixbars, spawning close to the new moon may maximize fitness by enhancing survival of adults and/or growth and survival of offspring.
Temporal patterns of spawning are frequently linked to hydrodynamic regimes that facilitate offshore transport of eggs and larvae (Johannes 1978; Sancho et al. 2000; Claydon et al. 2014). However, local current dynamics in shallow lagoon habitats may be a poor predictor for dispersal processes because local wind conditions and shear-flow generated eddies can generate substantial variability in current patterns around the spawning site (see Warner 1997; Ezer et al. 2011). This may explain why our analyses failed to detect any strong relationships between spawning activity and local current velocity/direction. Maximum tidal amplitudes during new and full moon phases that promote a persistent, large-scale offshore transport despite the local variability, may provide spawners with a more useful (generalized) cue for transport-related processes (Robertson et al. 1990; Warner 1997; Claydon 2004; Claydon et al. 2014). However, if sixbars are only attempting to spawn during peak tidal amplitudes, then they might be expected to exhibit semi-lunar spawning patterns (i.e. spawning disproportionately on both the new moon and full moon, when tidal amplitudes are similarly maximal). So, at best, lunar-driven offshore dispersal provides only a partial explanation for the patterns of spawning that we observed. Alternatively, we note that our sampling of reproductive activities may have been insufficient to detect more subtle patterns of variation, and/or correlations with some environmental variables.
Lunar illumination might influence the fitness of dispersing eggs and larvae. Many juvenile reef fish settle back to the reef at night (Dufour and Galzin 1993) and during new moon, presumably to reduce their risk to predators (Sponaugle and Cowen 1994; Robertson et al. 1999; Shima and Swearer 2019). Similarly, the degree of lunar illumination at night might drive reproductive decisions of adults. Since a majority of iteroparous, protogynous species spawn in the afternoon (Colin and Clavijo 1988; Sancho et al. 2000; Claydon et al. 2014), dark nights during the new moon might reduce predation pressure on recently fertilized eggs that are still in close proximity to the reef.
If the developmental duration of pelagic larvae is invariant, the precise timing of spawning might dictate the timing of larval settlement to the benthos. Reef fish might use the lunar phase as an environmental cue to spawn at times of the month that facilitate their offspring’s settlement at a favourable time (e.g. the new moon; Robertson et al. 1988; Shima et al. 2018). Coral trout (Plectropomus leopardus) appear to exemplify this: spawning occurs around the new moon (Samoilys 1997), larvae develop for ~ 25 days in the pelagic (Doherty et al. 1994), and then settle back to the reef close to the following new moon (when risk from nocturnally active predators is presumed to be lowest). Temporal coupling of production and settlement has also been observed for the damselfish Stegastes partitus in the Caribbean (Robertson et al., 1988). Sixbars also spawn and settle disproportionately on the new moon (Shima et al. 2018). However, they have an average pelagic larval duration (PLD) of 47 days (Victor 1986; Shima et al. 2020). Given the preponderance of spawning close to the new moon, a PLD of 47 days would seem to indicate that settlement should commonly occur close to the full moon (a pattern that is not observed; Shima et al. 2018). This apparent conundrum is reconciled, at least in part, by plasticity in larval development time: PLD is variable in this species (range: 37–61 days), and larvae (that survive to successfully settle) appear to adjust their PLD to settle closer to the new moon irrespective of their date of spawning (Shima et al., 2020). If adaptive plasticity in PLD effectively decouples spawning dates from settlement dates for sixbars, this suggests that their lunar periodicity in spawning might not be related to selection pressures associated with settlement. So why then do they spawn disproportionately around the new moon? One possible explanation is that spawning on a new moon results in a growth advantage to larvae that survive to settlement (Shima et al 2021). The mechanisms underlying this pattern remain untested, but may be related to interactions with a community of organisms that migrate vertically in the water column (see Shima et al 2021, 2022 for the full hypothesis). The growth advantage associated with spawning on the new moon may improve fitness of subsequent life stages, because fish that settle at a larger size may have a future competitive advantage (Forrester 1991; Sogard 1997; Shima and Findlay 2002). Newly settled sixbars appear to have a size-based dominance hierarchy (personal observations), and individuals that settle larger may have better access to limited food and shelter resources (Forrester 1991; Shima and Findlay 2002; McCormick & Hoey 2004). If these size-related advantages persist and compound through time, then offspring that are born around the new moon (and survive to settle) may ultimately be more likely survive to sex change (and transform into territorial males) due to their size-related competitive abilities. In a protogynous breeding system, territorial males achieve the highest levels of fitness because they can monopolize mating opportunities with many females (Warner and Hoffman 1980a, 1980b; Warner 1984a; Walker et al. 2007). Consequently, parents might periodically increase their reproductive effort around the new moon as a strategy to produce high-quality phenotypes (e.g. fast growers) that can maximize offspring production.
Potential drivers of lunar periodicity: adult survival
Lunar-cyclic reproduction may be an adaptive response to specific predator–prey dynamics that periodically increase mortality risk for sixbars adults at the spawning sites. We regularly observed spawning aggregations of common prey species like Chlorurus sordidus and Acanthurus nigrofuscus during full moon phases at the spawning sites. Since these small-bodied species usually reproduce in large, conspicuous aggregations near the reef edge, they might attract predators that hunt along the reef edges. For sixbar wrasse, concentrating reproductive activities around the third quarter/new moon may be a strategy to evade predators (i.e. reduce ‘apparent competition’) around the full moon (Shima et al., 2020).
Alternatively, particular phases of the moon might not provide any selective advantage, and the lunar cycle may simply be a timing cue to synchronize reproduction across individuals (Colin and Bell 1991; Domeier and Colin 1997; Claydon et al. 2014). We regularly observed smaller initial phase males and females forming group spawning aggregations within male territories during new moon. Higher densities of initial phase males (and corresponding group spawning activities) might simply be a response to an increased number of females at the mating site. However, synchrony of reproductive activities around the new moon might also represent a targeted strategy to improve survival chances of smaller individuals (that are particularly vulnerable to predators): group spawning may reduce per capita predation risk and/or improve predator detection due to an increased collective vigilance (see ‘many eyes hypothesis’, Roberts 1996). Additionally, constraining the portion of the month over which spawning occurs may enable smaller individuals to allocate more time to feeding (and growth) instead of to costly (and sometimes life-threatening) reproductive behaviours like sneaking, courtship, and migration (Hoffman 1983; Colin and Clavijo 1988; Molloy et al. 2012
Local environmental cues
Although our findings suggest that spawning intensity is most strongly shaped by a large-scale environmental cue (i.e. the lunar cycle), model predictions also indicate an important (albeit weaker) effect of local environmental conditions. Spawning intensity varied strongly with proximity to the reef edge. Male territories closest to the reef edge in our study had ~ sixfold more spawning events than those furthest from the reef edge. This effect size is similar to the magnitude of increase in spawning intensity from full moon to new moon. Despite intense courtship efforts of territorial males in sheltered territories (i.e. furthest from the reef edge), most females (particularly the largest ones) travelled through those territories to mate with males that occupied the more exposed territories (i.e. closest to the reef edge). Potentially, exposed territories might provide an advantage immediately following the release of eggs (Hensley et al. 1994): shear flows along outer reef edges produce hydrological turbulences that may rapidly entrain the fertilized eggs into larger current systems, transporting them beyond the reach of potential egg predators (Colin and Clavijo 1988; Claydon 2004; Hamner and Largier 2011). When spawning did occur in sheltered territories, it tended to involve smaller individuals (Mitterwallner 2020). This suggests some risk-based decision making by spawners. Hence, territory selection might be mediated by a site-specific trade-off between predation pressure on adults, and the initial survival potential of released eggs (Robertson and Hoffman 1977; Sancho et al. 2000; Claydon 2004).
Our study incorporated a wide range of environmental variables. This enabled us to evaluate potential spawning cues (and their interactions) that operate across a range of scales. Our findings suggest that temporal variation in spawning intensity of the sixbar wrasse is overwhelmingly shaped by a large-scale, global cue (the lunar cycle). In a temporarily variable environment, purely conditional adjustments may be evolutionary detrimental, since individuals are not able to predict long-term consequences on offspring dispersal (Lloyd 1984; Moran 1992; Warner 1997). Consequently, selection may favour fixed responses to a global cue (the lunar cycle), as a mechanism that guarantees intermediate offspring fitness across a range of environmental states (Moran 1992; Warner 1997). Additionally, we hypothesize that lunar-cyclic reproduction might represent a strategy to maximize adult survival potential. Our study also suggests that parents seem to utilize a detectable local cue for reproduction: territory location appears to convey sufficient information in terms of initial offspring survival to elicit behavioural responses over short timescales.
Change history
17 October 2022
Missing Open Access funding information has been added in the Funding Note.
References
Barlow GW (1981) Patterns of parental investment, dispersal and size among coral-reef fishes. Environ Biol Fishes 6:65–85
Barton K (2015) MuMIn: Multi-model inference. R package version 1.13.4. See https://CRAN.R- project.org/package=MuMIn
Bates D, Maechler M, Bolker B (2012) lme4: Linear mixed-effects models using S4 classes. R package version 0.999999–0. Available: http://cran.r-project.org/web/packages/lme4/lme4
Brockman HJ, Taborsky M (2008) Alternative reproductive tactics and the evolution of alternative allocation phenotypes. In: Oliveira RF, Taborsky M, Brockman HJ (eds) Alternative reproductive tactics: an integrative approach. Cambridge University Press, Cambridge, pp 2–51
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Machler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modelling. The R Journal, pp. 378–400
Burnham KP, Anderson DR (2002) Model selection and inference. A practical information-theoretic approach. Springer, Berlin Heidelberg New York
Claydon J (2004) Spawning aggregations of coral reef fishes: characteristics, hypotheses, threats and management. Oceanogr Mar Biol 42:265–302
Claydon J, McCormick MI, Jones GP (2012) Patterns of migration between feeding and spawning sites in a coral reef surgeonfish. Coral Reefs 31:77–87
Claydon J, McCormick MI, Jones GP (2014) Multispecies spawning sites for fishes on a low-latitude coral reef: spatial and temporal patterns. J Fish Biol 84:1136–1163
Colin PL, Bell LJ (1991) Aspects of the spawning of labrid and scarid fishes (Pisces: Labroidei) at Enewetak Atoll, Marshall Islands with notes on other families. Environ Biol Fishes 31:229–260
Colin PL, Clavijo IE (1988) Spawning activity of fishes producing pelagic eggs on a shelf edge coral reef, southwestern Puerto Rico. Bull Mar Sci 43:149–179
Cushing DH (1975) Marine ecology and fisheries. Cambridge University Press, Cambridge
Danilowicz BS, Sale PF (1999) Relative intensity of predation on the French grunt, Haemulon flavolineatum, during diurnal, dusk, and nocturnal periods on a coral reef. Mar Biol 133:337–343
deBruyn AMH, Meeuwig JJ (2001) Detecting lunar cycles in marine ecology: periodic regression versus categorical ANOVA. Mar Ecol Prog Ser 214:307–310
Doherty PJ, Fowler AJ, Samoilys MA, Harris DA (1994) Monitoring the replenishment of coral trout (Pisces: Serranidae) populations. Bull Mar Sci 54:343–355
Doherty PJ, Dufour V, Galzin R, Hixon MA, Planes S (2004) High mortality during settlement is a population bottleneck for a tropical surgeonfish. Ecology 85:2422–2428
Domeier ML, Colin PL (1997) Tropical reef fish spawning aggregations defined and reviewed. Bull Mar Sci 60:698–726
Donahue MJ, Karnauskas M, Toews C, Paris CB (2015) Location isn’t everything: timing of spawning aggregations optimizes larval replenishment. PLoS ONE 10:1–14
Dufour V, Galzin R (1993) Colonization patterns of reef fish larvae to the lagoon at Moorea Island, French Polynesia. Mar Ecol Prog Ser 102:143–152
Einum S, Fleming IA (2004) Environmental unpredictability and offspring size: Conservative versus diversified bet-hedging. Evol Ecol Res 6:443–455
Ezer T, Heyman WD, Houser C, Kjerfve B (2011) Modelling and observations of high-frequency flow variability and internal waves at a Caribbean reef spawning aggregation site. Ocean Dyn 61:581–598
Forrester GE (1991) Social rank, individual size and group composition as determinants of food consumption by humbug damselfish. Dascyllus Aruanus Animal Behaviour 42(5):701–711
Galzin R (1987) Structure of fish communities of French Polynesian coral reefs. II. Temporal Scales Marine Ecol Prog Series 41:137–145
Galzin R, Pointier JP (1985) Moorea Island, Society Archipelago. In Delesalle B, Galzin R, Salvat B (eds). Fifth International Coral Reef Congress, Tahiti, French Polynesia, 1:75–101
Goodman D (1984) Risk spreading as an adaptive strategy in iteroparous life histories. Theor Popul Biol 25:1–20
Gross MR (1996) Alternative reproductive strategies and tactics: diversity within sexes. TREE 2:92–98
Gust N (2004) Variation in the population biology of protogynous coral reef fishes over tens of kilometres. Can J Fish Aquat Sci 61:205–218
Hamner WH, Largier JL (2011) Oceanography of the planktonic stages of aggregation spawning reef fishes. In: de Mitcheson YS, Colin PL (eds) Reef fish spawning aggregations: biology, research and management. Springer, Heidelberg, pp 159–190
Hartig F (2017) DHARMa: Residual diagnostics for hierarchical (multi-level mixed) regression models. r package version 0.1.5
Hartig F (2018) DHARMa: Residual diagnostics for hierarchical (multi-level/mixed) regression models. R package version 0.1.6. Available from: https://cran.r-project.org/web/packages/DHARMa/index.html
Hensley DA, Appeldoorn RS, Shapiro DY, Ray M, Turingan RG (1994) Egg dispersal in a Caribbean coral reef fish, Thalassoma bifasciatum, I- Dispersal over the reef platform. Bull Mar Sci 54:256–270
Heyman WD, Kjerfve B (2008) Characterization of multi-species reef fish spawning aggregations at Gladden Spit, Belize. Bull Mar Sci 83:531–551
Hobson ES (1974) Feeding relationships of teleostean fishes on coral reefs in Kona. Hawaii Fish Bulletin NOAA 72:915–1031
Hoffman SG (1983) Sex-related foraging behaviour in sequentially hermaphroditic hogfishes (Bodianus spp.). Ecology 64:798–808
Hughes PW (2017) Between semelparity and iteroparity: Empirical evidence for a continuum of modes of parity. Ecol Evol 7:8232–8261
Johannes RE (1978) Reproductive strategies of coastal marine fishes in the tropics. Environ Biol Fishes 3:65–84
Karnauskas M, Cherubin LM, Paris CB (2011) Adaptive significance of the formation of multi-species fish spawning aggregations near submerged Capes. PLoS ONE 6:e22067
Kuwamura T, Suzuki S, Kadota T (2009) Interspecific variation in the spawning time of labrid fish on a fringing reef at Iriomote Island, Okinawa. Ichthyol Res 63:460–469
Lenth RV (2018) emmeans: Estimates Marginal Means, aka Least-Squares Means. Retrieved from https://CRAN.R-project.org/package=emmeans
Lloyd DG (1984) Variation strategies of plants in heterogeneous environments. Biol J Lin Soc 21:357–385
Lobel PS (1978) Diel, lunar, and seasonal periodicity in the reproductive behavior of the pomacanthid fish, Centropyge potteri, and some other reef fishes in Hawaii. Pac Sci 32:193–207
Lott DF (1991) Intraspecific variation in the social systems of wild vertebrates. Cambridge University Press, Cambridge, p 238
McCormick MI, Hoey AS (2004) Larval growth history determines juvenile growth and survival in a tropical marine fish. Oikos 106(2):225–242
Meyer CG, Papastamatiou YP, Clark TB (2010) Differential movement patterns and site fidelity among trophic groups of reef fishes in a Hawaiian marine protected area. Mar Biol 157:1499–1511
Mitterwallner P (2020) Reproductive timing and investment decisions of a protogynous, hermaphroditic coral reef fish species. Ph.D. thesis, Victoria University of Wellington
Molloy PP, Cote IM, Reynolds JD (2012) Why spawn in aggregations? In: de Mitcheson YS, Colin PL (eds) Reef fish spawning aggregations: Biology, Research and Management. Springer, New York, pp 57–83
Moran NA (1992) The evolutionary maintenance of alternative phenotypes. Am Nat 139:971–989
Moyer JT (1989) Reef channels as spawning sites for fishes on the Shiraho coral reef, Ishigaki Island, Japan. Japanese J Ichthyol 36:371–375
Nakagawa S, Schielzeth H (2013) A general and simple method for obtaining R 2 from generalized linear mixed-effects models. Methods Ecol Evol 2:133–142
Nemeth RS, Blondeau J, Herzlieb S, Kadison E (2007) Spatial and temporal patterns of movement and migration at spawning aggregations of red hind, Epinephelus guttatus, in the U.S. Virgin Islands Environ Biol Fish 78:365–381
Petersen CW, Warner RR, Cohen S, Hess HC, Sewell AT (1992) Variable pelagic fertilization success: implications for mate choice and spatial patterns of mating. Ecology 73:391–401
Philippi T, Seger J (1989) Hedging one’s evolutionary bets, revisited. Trends Ecol Evol 4:41–44
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Randall JE, Randall HA (1963) The spawning and early development of the Atlantic parrotfish, Sparisoma rubripinne, with notes on other scarid and labrid fishes. Zoologica 48:49–60
Roberts G (1996) Why individual vigilance declines as group size increases. Anim Behav 51:1077–1086
Robertson DR (1983) On the spawning behavior and spawning cycles of eight surgeonfishes (Acanthuridae) from the Indo-Pacific. Environ Biol Fishes 9:193–223
Robertson DR, Hoffman SG (1977) The roles of female mate choice and predation in the mating systems of some tropical labroid fishes. Z Tierpsychol 45:298–320
Robertson DR, Green DG, Victor BC (1988) Temporal coupling of production and recruitment. Ecology 69:370–381
Robertson DR, Petersen CW, Brawn JD (1990) Lunar reproductive cycles of benthic brooding reef fishes: reflections of larval biology or adult biology. Ecol Monogr 60:311–329
Robertson DR, Swearer SE, Kaufmann K, Brothers EB (1999) Settlement vs. environmental dynamics in a pelagic-spawning reef fish at Caribbean Panama. Ecol Monogr 69:195–218
Samoilys MA (1997) Periodicity of spawning aggregations of coral trout Plectropomus leopardus (Pisces: Serranidae) on the northern Great Barrier Reef. Mar Ecol Prog Ser 160:149–159
Sancho G, Solow AR, Lobel PS (2000) Environmental influences on the diel timing of spawning in coral reef fishes. Mar Ecol Prog Ser 206:193–212
Schultz ET, Warner RR (1991) Phenotypic plasticity in life-history traits of female Thalassoma bifasciatum (Pisces: Labridae): 2. Correlation of fecundity and growth rate in comparative studies. Environ Biol Fishes 30:333–344
Seger J, Brockman HJ (1987) What is bet-hedging? Oxf Surv Evol Biol 4:181–211
Shapiro DY (1991) Intraspecific variability in social systems of coral reef fishes. In: Sale P (ed) The ecology of fishes on coral reefs. Academic Press, San Diego, pp 331–355
Shapiro D, Hensley D, Appeldoorn R (1988) Pelagic spawning and egg transport in coral-reef fishes: a skeptical overview. Environ Biol Fishes 22:3–14
Shima JS, Findlay AM (2002) Pelagic larval growth rate impacts benthic settlement and survival of temperate reef fish. Mar Ecol Prog Ser 235:303–309
Shima JS, Noonburg EG, Swearer SE, Alonzo SH, Osenberg CW (2018) Born at the right time? A conceptual framework linking reproduction, development, and settlement in reef fish. Ecology 99:116–126
Shima JS, Osenberg CW, Noonburg EG, Alonzo SH, Swearer SE (2021) Lunar rhythms in growth of larval fish. Proc R Soc B 288:20202609. https://doi.org/10.1098/rspb.2020.2609
Shima JS, Osenberg CW, Alonzo SH, Noonburg EG, Swearer SE (2022) How moonlight shapes environments, life histories, and ecological interactions on coral reefs. Emerg Topics in Life Sci. https://doi.org/10.1042/ETLS20210237
Shima JS, Swearer SE (2019) Moonlight enhances growth in larval fish. Ecology, e02563
Shima JS, Osenberg CW, Alonzo SH, Noonburg EG, Mitterwallner P, Swearer SE (2020) Reproductive phenology across the lunar cycle: Parental decisions, offspring responses, and consequences for reef fish. Ecology 101:e03086
Sogard SM (1997) Size-selective mortality in the juvenile stage of teleost fishes: a review. Bull Mar Sci 60:1129–1157
Sponaugle S, Cowen RK (1994) Larval durations and recruitment patterns of two Caribbean gobies (Gobiidae): contrasting early life histories in demersal spawners. Mar Biol 120:133–143
Takemura A, Rahman MS, Nakamura S, Young JP, Takano K (2004) Lunar cycles and reproductive activity in reef fishes with particular attention to rabbitfishes. Fish Fish 5:317–328
Victor BC (1986) Duration of the planktonic larval stage of one hundred species of Pacific and Atlantic wrasses (family Labridae). Mar Biol 90:317–326
Walker SPW, Ryen CA, McCormick MI (2007) Rapid larval growth predisposes sex change and sexual size dimorphism in a protogynous hermaphrodite, Parapercis snyderi Jordan & Starks 1905. J Fish Biol 71:1347–1357
Warner RR (1984) Mating behavior and hermaphroditism in coral reef fishes: the diverse forms of sexuality. Am Sci 72:128–136
Warner RR (1995) Large mating aggregations and daily long-distance spawning migrations in the bluehead wrasse, Thalassoma bifasciatum. Environ Biol Fishes 44:337–345
Warner RR (1997) Evolutionary ecology: How to reconcile pelagic dispersal with local adaptation. Coral Reefs 16:115–120
Warner RR (1998) The role of extreme iteroparity and risk avoidance in the evolution of mating systems. J Fish Biol 53:82–93
Warner RR, Hoffman SG (1980a) Population density and the economics of territoriality in a coral reef fish. Ecology 61:772–780
Warner RR, Hoffman SG (1980b) Local population size as a determinant of mating system and sexual composition in two tropical marine fishes (Thalassoma spp.). Evolution 34:508–518
Warner RR, Shapiro DY, Marcanato A, Petersen CW (1995) Sexual conflict: males with highest mating success convey the lowest fertilization benefits to females. Proceed Royal Soc B: Biol Sci 262:135–139
Wilbur HM, Rudolf VHW (2006) Life-history evolution in uncertain environments: bet hedging in time. Am Nat 168:398–411
Zeller DC (1998) Spawning aggregations: Patterns of movement of the coral trout Plectropomus leopardus (Serranidae) as determined by ultrasonic telemetry. Mar Ecol Prog Ser 162:253–263
Acknowledgements
P. Caie, K. Hillyer assisted with data collection. Research grants from Marsden Fund (VUW1503, 2016–2020) and Victoria University of Wellington provided funding. Victoria University Coastal Ecology Lab and the UC Gump Research Station provided essential logistic support.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Topical editor Alastair Harborne.
Appendix
Appendix
See Tables
4 and
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mitterwallner, P., Shima, J.S. The relative influence of environmental cues on reproductive allocation of a highly iteroparous coral reef fish. Coral Reefs 41, 1323–1335 (2022). https://doi.org/10.1007/s00338-022-02239-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-022-02239-6