Abstract
Objective
To determine the diagnostic performance of neuromelanin-sensitive magnetic resonance imaging discriminating between patients with Parkinson’s disease and normal healthy controls and to identify factors causing heterogeneity influencing the diagnostic performance.
Methods
A systematic literature search in the Ovid-MEDLINE and EMBASE databases was performed for studies reporting the relevant topic before February 17, 2020. The pooled sensitivity and specificity values with their 95% confidence intervals were calculated using bivariate random-effects modeling. Subgroup and meta-regression analyses were also performed to determine factors influencing heterogeneity.
Results
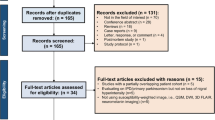
Twelve articles including 403 patients with Parkinson’s disease and 298 control participants were included in this systematic review and meta-analysis. Neuromelanin-sensitive magnetic resonance imaging showed a pooled sensitivity of 89% (95% confidence interval, 86–92%) and a pooled specificity of 83% (95% confidence interval, 76–88%). In the subgroup and meta-regression analysis, a disease duration longer than 5 and 10 years, comparisons using measured volumes instead of signal intensities, a slice thickness in terms of magnetic resonance imaging parameters of more than 2 mm, and semi-/automated segmentation methods instead of manual segmentation improved the diagnostic performance.
Conclusion
Neuromelanin-sensitive magnetic resonance imaging had a favorable diagnostic performance in discriminating patients with Parkinson’s disease from healthy controls. To improve diagnostic accuracy, further investigations directly comparing these heterogeneity-affecting factors and optimizing these parameters are necessary.
Key Points
• Neuromelanin-sensitive MRI favorably discriminates patients with Parkinson’s disease from healthy controls.
• Disease duration, parameters used for comparison, magnetic resonance imaging slice thickness, and segmentation methods affected heterogeneity across the studies.





Similar content being viewed by others
Abbreviations
- HC:
-
Normal healthy control
- HSROC:
-
Hierarchical summary receiver operating characteristic
- LC:
-
Locus coeruleus
- NM-MRI:
-
Neuromelanin-sensitive MRI
- PD:
-
Parkinson’s disease
- PRISMA:
-
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
- QUADAS-2:
-
Quality Assessment of Diagnostic Accuracy Studies-2
- SN:
-
Substantia nigra
References
Fedorow H, Tribl F, Halliday G, Gerlach M, Riederer P, Double KL (2005) Neuromelanin in human dopamine neurons: comparison with peripheral melanins and relevance to Parkinson’s disease. Prog Neurobiol 75:109–124
Sulzer D, Zecca L (2000) Intraneuronal dopamine-quinone synthesis: a review. Neurotox Res 1:181–195
Mann DM, Yates PO (1983) Pathological basis for neurotransmitter changes in Parkinson’s disease. Neuropathol Appl Neurobiol 9:3–19
Nakamura K, Sugaya K (2014) Neuromelanin-sensitive magnetic resonance imaging: a promising technique for depicting tissue characteristics containing neuromelanin. Neural Regen Res 9:759–760
Sasaki M, Shibata E, Tohyama K et al (2006) Neuromelanin magnetic resonance imaging of locus ceruleus and substantia nigra in Parkinson’s disease. Neuroreport 17:1215–1218
Wang X, Zhang Y, Zhu C et al (2019) The diagnostic value of SNpc using NM-MRI in Parkinson’s disease: meta-analysis. Neurol Sci. https://doi.org/10.1007/s10072-019-04014-y
Castellanos G, Fernandez-Seara MA, Lorenzo-Betancor O et al (2015) Automated neuromelanin imaging as a diagnostic biomarker for Parkinson’s disease. Mov Disord 30:945–952
Fabbri M, Reimao S, Carvalho M et al (2017) Substantia nigra neuromelanin as an imaging biomarker of disease progression in Parkinson’s disease. J Parkinsons Dis 7:491–501
Isaias IU, Trujillo P, Summers P et al (2016) Neuromelanin imaging and dopaminergic loss in Parkinson’s disease. Front Aging Neurosci 8:196
Kawaguchi H, Shimada H, Kodaka F et al (2016) Principal component analysis of multimodal neuromelanin MRI and dopamine transporter PET data provides a specific metric for the nigral dopaminergic neuronal density. PLoS One 11:e0151191
Le Berre A, Kamagata K, Otsuka Y et al (2019) Convolutional neural network-based segmentation can help in assessing the substantia nigra in neuromelanin MRI. Neuroradiology. https://doi.org/10.1007/s00234-019-02279-w
Ogisu K, Kudo K, Sasaki M et al (2013) 3D neuromelanin-sensitive magnetic resonance imaging with semi-automated volume measurement of the substantia nigra pars compacta for diagnosis of Parkinson’s disease. Neuroradiology 55:719–724
Ohtsuka C, Sasaki M, Konno K et al (2013) Changes in substantia nigra and locus coeruleus in patients with early-stage Parkinson’s disease using neuromelanin-sensitive MR imaging. Neurosci Lett 541:93–98
Prasad S, Stezin A, Lenka A et al (2018) Three-dimensional neuromelanin-sensitive magnetic resonance imaging of the substantia nigra in Parkinson’s disease. Eur J Neurol 25:680–686
Pyatigorskaya N, Magnin B, Mongin M et al (2018) Comparative study of MRI biomarkers in the substantia nigra to discriminate idiopathic Parkinson disease. AJNR Am J Neuroradiol 39:1460–1467
Schwarz ST, Xing Y, Tomar P, Bajaj N, Auer DP (2017) In vivo assessment of brainstem depigmentation in Parkinson disease: potential as a severity marker for multicenter studies. Radiology 283:789–798
Takahashi H, Watanabe Y, Tanaka H et al (2018) Comprehensive MRI quantification of the substantia nigra pars compacta in Parkinson’s disease. Eur J Radiol 109:48–56
Zupan G, Suput D, Pirtosek Z, Vovk A (2019) Semi-automatic signature-based segmentation method for quantification of neuromelanin in substantia nigra. Brain Sci:9
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Ann Intern Med 151:W65–W94
Whiting PF, Rutjes AW, Westwood ME et al (2011) QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med 155:529–536
Suh CH, Park SH (2016) Successful publication of systematic review and meta-analysis of studies evaluating diagnostic test accuracy. Korean J Radiol 17:5–6
Kim KW, Lee J, Choi SH, Huh J, Park SH (2015) Systematic review and meta-analysis of studies evaluating diagnostic test accuracy: a practical review for clinical researchers-part I. general guidance and tips. Korean J Radiol 16:1175–1187
Lee J, Kim KW, Choi SH, Huh J, Park SH (2015) Systematic review and meta-analysis of studies evaluating diagnostic test accuracy: a practical review for clinical researchers-part II. Statistical methods of meta-analysis. Korean J Radiol 16:1188–1196
Reitsma JB, Glas AS, Rutjes AW, Scholten RJ, Bossuyt PM, Zwinderman AH (2005) Bivariate analysis of sensitivity and specificity produces informative summary measures in diagnostic reviews. J Clin Epidemiol 58:982–990
Rutter CM, Gatsonis CA (2001) A hierarchical regression approach to meta-analysis of diagnostic test accuracy evaluations. Stat Med 20:2865–2884
Deeks JJ, Macaskill P, Irwig L (2005) The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed. J Clin Epidemiol 58:882–893
Hoaglin DC (2016) Misunderstandings about Q and ‘Cochran's Q test’ in meta-analysis. Stat Med 35:485–495
Deville WL, Buntinx F, Bouter LM et al (2002) Conducting systematic reviews of diagnostic studies: didactic guidelines. BMC Med Res Methodol 2:9
Cassidy CM, Zucca FA, Girgis RR et al (2019) Neuromelanin-sensitive MRI as a noninvasive proxy measure of dopamine function in the human brain. Proc Natl Acad Sci U S A 116:5108–5117
Kashihara K, Shinya T, Higaki F (2011) Neuromelanin magnetic resonance imaging of nigral volume loss in patients with Parkinson’s disease. J Clin Neurosci 18:1093–1096
Kuya K, Ogawa T, Shinohara Y et al (2018) Evaluation of Parkinson’s disease by neuromelanin-sensitive magnetic resonance imaging and (123)I-FP-CIT SPECT. Acta Radiol 59:593–598
Kuya K, Shinohara Y, Miyoshi F, Fujii S, Tanabe Y, Ogawa T (2016) Correlation between neuromelanin-sensitive MR imaging and (123)I-FP-CIT SPECT in patients with parkinsonism. Neuroradiology 58:351–356
Langley J, Huddleston DE, Sedlacik J, Boelmans K, Hu XP (2017) Parkinson’s disease-related increase of T2*-weighted hypointensity in substantia nigra pars compacta. Mov Disord 32:441–449
Matsuura K, Maeda M, Yata K et al (2013) Neuromelanin magnetic resonance imaging in Parkinson’s disease and multiple system atrophy. Eur Neurol 70:70–77
Moon WJ, Park JY, Yun WS et al (2016) A comparison of substantia nigra T1 hyperintensity in Parkinson’s disease dementia, Alzheimer’s disease and age-matched controls: volumetric analysis of neuromelanin imaging. Korean J Radiol 17:633–640
Hatano T, Okuzumi A, Kamagata K et al (2017) Neuromelanin MRI is useful for monitoring motor complications in Parkinson’s and PARK2 disease. J Neural Transm (Vienna) 124:407–415
Matsusue E, Fujihara Y, Tanaka K et al (2019) The utility of the combined use of (123)I-FP-CIT SPECT and neuromelanin MRI in differentiating Parkinson’s disease from other parkinsonian syndromes. Acta Radiol 60:230–238
Ohtsuka C, Sasaki M, Konno K et al (2014) Differentiation of early-stage parkinsonisms using neuromelanin-sensitive magnetic resonance imaging. Parkinsonism Relat Disord 20:755–760
Shinde S, Prasad S, Saboo Y et al (2019) Predictive markers for Parkinson’s disease using deep neural nets on neuromelanin sensitive MRI. Neuroimage Clin 22:101748
Wang J, Huang Z, Li Y et al (2019) Neuromelanin-sensitive MRI of the substantia nigra: an imaging biomarker to differentiate essential tremor from tremor-dominant Parkinson’s disease. Parkinsonism Relat Disord 58:3–8
Xiang Y, Gong T, Wu J et al (2017) Subtypes evaluation of motor dysfunction in Parkinson’s disease using neuromelanin-sensitive magnetic resonance imaging. Neurosci Lett 638:145–150
Reimao S, Pita Lobo P, Neutel D et al (2015) Substantia nigra neuromelanin magnetic resonance imaging in de novo Parkinson’s disease patients. Eur J Neurol 22:540–546
Reimao S, Pita Lobo P, Neutel D et al (2015) Substantia nigra neuromelanin-MR imaging differentiates essential tremor from Parkinson’s disease. Mov Disord 30:953–959
Reimao S, Pita Lobo P, Neutel D et al (2015) Quantitative analysis versus visual assessment of neuromelanin MR imaging for the diagnosis of Parkinson’s disease. J Parkinsons Dis 5:561–567
Takahashi H, Watanabe Y, Tanaka H et al (2018) Quantifying changes in nigrosomes using quantitative susceptibility mapping and neuromelanin imaging for the diagnosis of early-stage Parkinson’s disease. Br J Radiol 91:20180037
Taniguchi D, Hatano T, Kamagata K et al (2018) Neuromelanin imaging and midbrain volumetry in progressive supranuclear palsy and Parkinson’s disease. Mov Disord 33:1488–1492
Jin L, Wang J, Wang C et al (2019) Combined visualization of nigrosome-1 and neuromelanin in the substantia nigra using 3T MRI for the differential diagnosis of essential tremor and de novo Parkinson’s disease. Front Neurol 10:100
Sommerauer M, Fedorova TD, Hansen AK et al (2018) Evaluation of the noradrenergic system in Parkinson’s disease: an 11C-MeNER PET and neuromelanin MRI study. Brain 141:496–504
Tanaka M, Aihara Y, Ikeda S, Aihara Y (2011) Neuromelanin-related contrast in the substantia nigra semiquantitatively evaluated by magnetic resonance imaging at 3T: comparison between normal aging and Parkinson disease. Rinsho Shinkeigaku 51:14–20
Takahashi H, Watanabe Y, Tanaka H et al (2019) Quantifying the severity of Parkinson disease by use of dopaminergic neuroimaging. AJR Am J Roentgenol. https://doi.org/10.2214/ajr.18.20655:1-6
Ehrminger M, Latimier A, Pyatigorskaya N et al (2016) The coeruleus/subcoeruleus complex in idiopathic rapid eye movement sleep behaviour disorder. Brain 139:1180–1188
García-Lorenzo D, Longo-Dos Santos C, Ewenczyk C et al (2013) The coeruleus/subcoeruleus complex in rapid eye movement sleep behaviour disorders in Parkinson's disease. Brain 136:2120–2129
Ogura A, Maeda F, Miyai A, Kikumoto R (2005) Effects of slice thickness and matrix size on MRI for signal detection. Nihon Hoshasen Gijutsu Gakkai Zasshi 61:1140–1143
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT; No. 2019R1F1A1063771) and by the SNUBH Research Fund (Grant No. 09-2019-003).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Yun Jung Bae.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors (Se Jin Cho) has significant statistical expertise.
Informed consent
Written informed consent was not required for this study because this article is systematic review and meta-analysis.
Ethical approval
Institutional Review Board approval was not required because this article is systematic review and meta-analysis.
Methodology
• Systematic review
• Meta-analysis
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 3341 kb)
Rights and permissions
About this article
Cite this article
Cho, S.J., Bae, Y.J., Kim, JM. et al. Diagnostic performance of neuromelanin-sensitive magnetic resonance imaging for patients with Parkinson’s disease and factor analysis for its heterogeneity: a systematic review and meta-analysis. Eur Radiol 31, 1268–1280 (2021). https://doi.org/10.1007/s00330-020-07240-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-07240-7