Abstract
Key message
HT-induced ROS burst in developing anther is closely related to the lowered CAT activity as the result of the markedly suppressed OsCATB transcript, thereby causing severe fertility injury for rice plants exposed to HT at meiosis stage.
Abstract
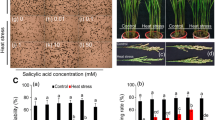
The reproductive stage of rice plants is highly sensitive to heat stress. In this paper, different rice cultivars were used to investigate the relationship of HT-induced floret sterility with reactive oxygen species (ROS) detoxification in rice anthers under well-controlled climatic conditions. Results showed that high temperature (HT) exposure significantly enhanced the ROS level and malondialdehyde (MDA) content in developing anther, and the increase in ROS amount in rice anther under HT exposure was closely associated with HT-induced decline in the activities of several antioxidant enzymes. For various antioxidant enzymes, SOD and CAT were more susceptible to the ROS burst in rice anther induced by HT exposure than APX and POD, in which SOD and CAT activity in developing anther decreased significantly by HT exposure, whereas APX activity was relatively stable among different temperature regimes. HT-induced decrease in CAT activity was attributable to the suppressed transcript of OsCATB. This occurrence was strongly responsible for HT-induced increase in ROS level and oxidative-damage in rice anther, thereby it finally caused significant reduction in pollen viability and floret fertility for the rice plants exposed to HT during meiosis. Exogenous application of 1000 µM salicylic acid (SA) may alleviate HT-induced reduction in pollen viability and floret fertility, concomitantly with the increased CAT activity and reduced ROS level in rice anther.







Similar content being viewed by others
Abbreviations
- APX:
-
Ascorbate peroxidase
- CAT:
-
Catalase
- CMS:
-
Cytoplasmic male sterility
- GR:
-
Glutathione reductase
- HT:
-
High temperature
- MDA:
-
Malondialdehyde
- NT:
-
Normal temperature
- PCD:
-
Programmed cell death
- POD:
-
Peroxidase
- ROS:
-
Reactive oxygen species
- SA:
-
Salicylic acid
- SOD:
-
Superoxide dismutase
References
Asada K (1997) The role of ascorbate peroxidase and monodehydro ascorbte reductase in H2O2 scavenging in plants. Cold Spring Harb Monogr Arch 34:715–735
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cao Z, Zhao Q, Huang F, Wei K, Zaidi S, Zhou W, Cheng F (2015) Effects of high temperature at anthesis on spikelet fertility and grain weight in relation to floral positions within a panicle of rice (Oryza sativa L.). Crop Pasture Sci 66:922–929
Choudhury S, Panda SK (2004) Role of salicylic acid in regulating cadmium induced oxidative stress in Oryza sativa L. roots. Bulg J Plant Physiol 30:95–110
Das S, Krishnan P, Nayak M, Ramakrishnan B (2014) High temperature stress effects on pollens of rice (Oryza sativa L.) genotypes. Environ Exp Bot 101:36–46
Endo M, Tsuchiya T, Hamada K, Kawamura S, Yano K, Ohshima M, Higashitani A, Watanabe M, Kawagishi-Kobaya M (2009) High temperatures cause male sterility in rice plants with transcriptional alterations during pollen development. Plant Cell Physiol 50:1911–1922
Foyer CH, Noctor G (2000) Oxygen processing in photosynthesis: regulation and signaling. New Phytol 146:359–388
Frank G, Pressman E, Ophir R, Althan L, Shaked R, Freedman M, Shen S, Firon N (2009) Transcriptional profiling of maturing tomato microspores reveals the involvement of heat shock proteins, ROS scavengers, hormones, and sugars in the heat stress response. J Exp Bot 60:3891–3908
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Govindaraj K (1986) Biochemical characterization of normal and male sterile anthers in rice (Oryza sativa L.). Indian J Genet Plant Breed 1:13–15
Hasanuzzaman M, Nahar K, Alam MM, Fujita M (2014) Modulation of antioxidant machinery and the methylglyoxal detoxification system in selenium-supplemented Brassica napus seedlings confers tolerance to high temperature. Biol Trace Elem Res 161:297–307
Hayat Q, Hayat S, Irfan M, Ahmad A (2010) Effect of exogenous salicylic acid under changing environment: a review. Environ Exp Bot 68:14–25
Horváth E, Janda T, Szalai G, Páldi E (2002) In vitro salicylic acid inhibition of catalase activity in maize: differences between the isozymes and a possible role in the induction of chilling tolerance. Plant Sci 163:1129–1135
Iwamoto M, Higo H, Higo K (2000) Differential diurnal expression of rice catalase genes: the 5′-flanking region of CatA is not sufficient for circadian control. Plant Sci 151:39–46
Jagadish SVK, Craufurd PQ, Wheeler TR (2007) High temperature stress and spikelet fertility in rice (Oryza sativa L.). J Exp Bot 58:1627–1635
James DF, Foyer CH, Scott IM (1998) Parallel changes in H2O2 and catalase during thermo-tolerance induced by salicylic acid or heat acclimation in mustard seedlings. Plant Physiol 116:1351–1357
Janda T, Szalai G, Tari I, Paldi E (1999) Hydroponic treatment with salicylic acid decreases the effect of chilling injury in maize (Zea mays L.) plants. Planta 208:175–180
Jiang P, Zhang Y, Zhu W, Zhu H, Wang X (2007) Metabolism of reactive oxygen species in cotton cytoplasmic male sterility and its restoration. Plant Cell Rep 26:1627–1634
Kawaiyamada M, Ohori Y, Uchimiya H (2004) Dissection of Arabidopsis Bax inhibitor-1 suppressing Bax-, hydrogen peroxide-, and salicylic acid-induced cell death. Plant Cell 16:21–32
Klessig DF, Malamy J et al (1994) The salicylic acid signal plants. Plant Mol Biol 26:439–1458
Krantev A, Yordanova R, Janda T, Szalai G, Popova L (2008) Treatment with salicylic acid decreases the effect of cadmium on photosynthesis in maize plants. J Plant Physiol 165:920–931
Krishnan P, Ramakrishnan B, Reddy KR, Reddy VR (2011) High-temperature effects on rice growth, yield, and grain quality. Adv Agron 111:87–206
Li S, Wan C, Kong J, Zhang Z, Li Y, Zhu Y (2004) Programmed cell death during microgenesis in a Honglian CMS line of rice is correlated with oxidative stress in mitochondria. Funct Plant Biol 31:369–376
Matsui T, Omasa K (2002) Rice (Oryza sativa L.) cultivars tolerant to high temperature at flowering: anther characteristics. Ann Bot 89:683–687
Matsumura H, Nirasawa S, Kiba A, Urasaki N, Saitoh HM, Kawai-Yamada M, Uchimiya H, Terauchi R (2003) Overexpression of Bax inhibitor suppresses the fungal elicitor-induced cell death in rice (Oryza sativa L.) cells. Plant J Cell Molecular Biol 33:425–434
Metwally A, Finkemeier I, Georgi M, Dietz KJ (2003) Salicylic acid alleviates the cadmium toxicitybarley seedlings. Plant Physiol 1321:272–281
Mhamdi A, Queval G, Chaouch S, Vanderauwera S, Breusegem FV, Noctor G (2010) Catalase function in plants: a focus on Arabiopsis mutants as stress-mimic models. J Exp Bot 61:4197–4220
Mohammed AR, Tarpley L (2011) Effects of night temperature, spikelet position and salicylic acid on yield and yield-related parameters of rice (Oryza sativa L.) plants. J Agron Crop Sci 33:117–123
Nguyen GN, Sutton BG (2009) Water deficit reduced fertility of young microspores resulting in a decline of viable mature pollen and grain set in rice. J Agron Crop Sci 195:11–18
Ozgur R, Turkan I, Uzilday B, Sekmen AH (2014) Endoplasmic reticulum stress triggers ROS signaling, changes the redox state, and regulates the antioxidant defence of Arabidopsis thaliana. J Exp Bot 65:1377–1390
Panda SK, Patra HK (2007) Effect of salicylic acid potentiates cadmium-induced oxidative damage in Oryza sativa L. leaves. Acta Physiol Plant 29:567–575
Prasad PVV, Boote KJ, Allen JLH, Sheehy JE, Thomas JMG (2006) Species, ecotype and cultivar differences in spikelet fertility and harvest index of rice in response to high temperature stress. Field Crop Res 95:398–411
Rao MY, Davis KR (1999) Ozone-induced cell death occurs via two distinct mechanisms in Arabidopsis: the role of salicylic acid. Plant J 17:603–614
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protocols 3:1101–1108
Selote DS, Khanna-Chopra R (2010) Antioxidant response of wheat roots to drought acclimation. Protoplasma 245:153–163
Teixeira EI, Fischer G, Velthuizen HV, Walter C, Ewert F (2013) Global hot-spots of heat stress on agricultural crops due to climate change. Agric Forest Meteorol 170:206–215
Tiwari BS, Belengh B, Levine A (2002) Oxidative stress increased respiration and generation of reactive oxygen species, resulting in ATP depletion, opening of mitochondrial permeability transition, and programmed cell death. Plant Physiol 128:1271–1281
Wan CX, Li SQ, Wen L, Kong J, Wang K, Zhu YG (2007) Damage of oxidative stress on mitochondria during microspores development in Honglian CMS line of rice. Plant Cell Rep 26:373–382
Wang LJ, Fan L, Loescher W, Duan W, Liu GJ, Cheng JS, Luo HB, Li SH (2010) Salicylic acid alleviates decreases in photosynthesis under heat stress and accelerates recovery in grapevine leaves. BMC Plant Biol 10:34
Willekens H, Chamnongpol S, Davey M, Schraudner M, Langebartels C (1997) Catalase is a sink for H2O2 and is indispensable for stress defense in C3 plants. Embo J 14:4806–4816
Zhang C, Fu G, Yang X, Yang Y, Zhao X, Chen T, Zhang X, Jin Q, Tao L (2015) Heat stress effects are stronger on spikelets than on flag leaves in rice due to differences in dissipation capacity. J Agron Crop Sci 202:394–408
Acknowledgements
The authors are indebted to National Key Research and Development Plan of China (No. 2016YFD0300502), National Natural Science Foundation of China (No. 31571602) and Zhejiang Provincial Natural Science Foundation of China (LZ15C130001) for its financial support to this research project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Kang Chong.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhao, Q., Zhou, L., Liu, J. et al. Involvement of CAT in the detoxification of HT-induced ROS burst in rice anther and its relation to pollen fertility. Plant Cell Rep 37, 741–757 (2018). https://doi.org/10.1007/s00299-018-2264-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-018-2264-y