Abstract
Key message
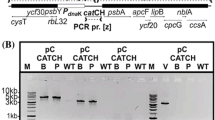
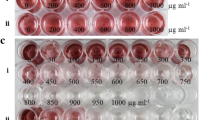
A mutant glutamate 1-semialdehyde aminotransferase gene from the Synechococcus , inserted into tobacco plastid DNA by means of particle bombardment and antibiotic selection, conferred gabaculine resistance allowing to attain homoplasmy.
Abstract
Many plant species are recalcitrant to plastid genome transformation. New selections systems may help to overcome this limitation and to extend the application of this technology. A mutant hemL gene from the photosynthetic cyanobacterium Synechococcus, encoding a gabaculine-insensitive glutamate 1-semialdehyde aminotransferase (GSA), is an efficient selectable marker gene for nuclear transformation of tobacco, alfalfa and durum wheat. Since GSA functions in the plastid, we introduced the mutant hemL gene into the tobacco plastid genome along with the conventional antibiotic resistance aadA gene, in the attempt to develop a new selection system for plastome transformation. Although we were unable to directly regenerate gabaculine resistant transplastomic plants, we demonstrated the functionality of hemL in tobacco plastids by using gabaculine selection in the second and third rounds of in vitro selection that permitted to obtain the homoplasmic state in transgenic plants. Thus, the mutant hemL gene functions as a secondary selection marker in tobacco plastids. Our results encourage further attempts to test gabaculine resistant GSA for plastome transformation of crop plants in which gabaculine has stronger regeneration-inhibiting effects with respect to tobacco.





Similar content being viewed by others
References
Ahlert D, Ruf S, Bock R (2003) Plastid protein synthesis is required for plant development in tobacco. Proc Natl Acad Sci USA 100:15730–15735. doi:10.1073/pnas.2533668100
Ayliffe MA, Timmis JN (1992) Tobacco nuclear DNA contains long tracts of homology to chloroplast DNA. Theor Appl Genet 85:229–238. doi:10.1007/BF00222864
Barone P, Zhang X-H, Widholm JM (2009) Tobacco plastid transformation using the feedback-insensitive anthranilate synthase α-subunit of tobacco (ASA2) as a new selectable marker. J Exp Bot 60:3195–3202. doi:10.1093/jxb/erp160
Bellucci M, Alpini A, Paolocci F, Cong L, Arcioni S (2000) Accumulation of γ-zein and γ-zein: KDEL to high levels in tobacco leaves and differential synthesis of BiP in transformants. Theor Appl Genet 101:796–804. doi:10.1007/s001220051546
Bellucci M, De Marchis F, Nicoletti I, Arcioni S (2007) Zeolin is a recombinant storage protein with different solubility and stability properties according to its localization in the endoplasmic reticulum or in the chloroplast. J Biotechnol 131:97–105. doi:10.1016/j.jbiotec.2007.06.004
Bock R (2014) Genetic engineering of the chloroplast: novel tools and new applications. Curr Opin Biotechnol 26:7–13. doi:10.1016/j.copbio.2013.06.004
Bock R (2015) Engineering plastid genomes: methods, tools, and applications in basic research and biotechnology. Annu Rev Plant Biol 66:211–241. doi:10.1146/annurev-arplant-050213-040212
Carrer H, Hockenberry TN, Svab Z, Maliga P (1993) Kanamycin resistance as a selectable marker for plastid transformation in tobacco. Mol Gen Genet 241:49–56
Clarke JL, Daniell H (2011) Plastid biotechnology for crop production: present status and future perspectives. Plant Mol Biol 76:211–220. doi:10.1007/s11103-011-9767-z
Craig W, Lenzi P, Scotti N, De Palma M, Saggese P, Carbone V, McGrath Curran N et al (2008) Transplastomic tobacco plants expressing a fatty acid desaturase gene exhibit altered fatty acid profiles and improved cold tolerance. Transgenic Res 17:769–782. doi:10.1007/s11248-008-9164-9
Daniell H, Datta R, Varma S, Gray S, Lee SB (1998) Containment of herbicide resistance through genetic engineering of the chloroplast genome. Nat Biotechnol 16:345–348. doi:10.1038/nbt0498-345
Daniell H, Muthukumar B, Lee SB (2001) Marker free transgenic plants: engineering the chloroplast genome without the use of antibiotic selection. Curr Genet 39:109–116. doi:10.1007/s002940100185
Day A, Goldschmidt-Clermont M (2011) The chloroplast transformation toolbox: selectable markers and marker removal. Plant Biotechnol J 9:540–553. doi:10.1111/j.1467-7652.2011.00604.x
De Cosa B, Moar W, Lee SB, Miller M, Daniell H (2001) Overexpression of the Bt cry2Aa2 operon in chloroplasts leads to formation of insecticidal crystals. Nat Biotechnol 19:71–74. doi:10.1038/83559
De Marchis F, Pompa A, Mannucci R, Morosinotto T, Bellucci M (2011) A plant secretory signal peptide targets plastome-encoded recombinant proteins to the thylakoid membrane. Plant Mol Biol 76:427–444. doi:10.1007/s11103-010-9676-6
De Marchis F, Pompa A, Bellucci M (2012) Plastid proteostasis and heterologous protein accumulation in transplastomic plants. Plant Physiol 160:571–581. doi:10.1104/pp.112.203778
Dhingra A, Portis AR Jr, Daniell H (2004) Enhanced translation of a chloroplast-expressed RbcS gene restores small subunit levels and photosynthesis in nuclear RbcS antisense plants. Proc Natl Acad Sci USA 101:6315–6320. doi:10.1073/pnas.0400981101
Dix P, Kavanagh T (1995) Transforming the plastome—genetic-markers and DNA delivery systems. Euphytica 85:29–34. doi:10.1007/BF00023927
Dufourmantel N, Dubald M, Matringe M, Canard H, Garcon F, Job C, Kay E, Wisniewski JP, Ferullo JM, Pelissier B, Sailland A, Tissot G (2007) Generation and characterization of soybean and marker-free tobacco plastid transformants over-expressing a bacterial 4-hydroxyphenylpyruvate dioxygenase which provides strong herbicide tolerance. Plant Biotechnol J 5:118–133. doi:10.1111/j.1467-7652.2006.00226.x
Dunne A, Maple- Grødem J, Gargano D, Haslam R, Napier J, Chua NH, Russel R, Møller SG (2014) Modifying fatty acid profiles through a new cytokinin-based plastid transformation system. Plant J 80:1131–1138. doi:10.1111/tpj.12684
Falk J, Brosch M, Schafer A, Braun S, Krupinska K (2005) Characterization of transplastomic tobacco plants with a plastid localized barley 4-hydroxyphenylpyruvate dioxygenase. J Plant Physiol 162:738–742. doi:10.1016/j.jplph.2005.04.005
Fernández-San Millán A, Obregón P, Veramendi J (2011) Over-expression of peptide deformylase in chloroplasts confers actinonin resistance, but is not a suitable selective marker system for plastid transformation. Transgenic Res 20:613–624. doi:10.1007/s11248-010-9447-9
Ferradini N, Nicolia A, Capomaccio S, Veronesi F, Rosellini D (2011) A point mutation in the Medicago sativa GSA gene provides a novel, efficient, selectable marker for plant genetic engineering. J Biotechnol 146:147–152. doi:10.1016/j.jbiotec.2011.08.015
Fleischmann TT, Scharff LB, Alkatib S, Hasdorf S, Schöttler MA, Bock R (2011) Nonessential plastid-encoded ribosomal proteins in tobacco: a developmental role for plastid translation and implications for reductive genome evolution. Plant Cell 23:3137–3155. doi:10.1105/tpc.111.088906
Giancaspro A, Rosellini D, Blanco A, Gadaleta A (2012) Gabaculine selection using bacterial and plant marker genes (GSA-AT) in durum wheat transformation. Plant Cell Tissue Organ Culture 109:447–455. doi:10.1007/s11240-011-0109-2
Gisby MF, Mudd EA, Day A (2012) Growth of transplastomic cells expressing d-amino acid oxidase in chloroplasts is tolerant to d-alanine and inhibited by d-valine1. Plant Physiol 160:2219–2226. doi:10.1104/pp.112.204107
Goldschmidt-Clermont M (1991) Transgenic expression of aminoglycoside adenine transferase in the chloroplast: a selectable marker of site-directed transformation of Chlamydomonas. Nucleic Acids Res 19:4083–4089
Gough KC, Hawes WS, Kilpatrick J, Whitelam GC (2001) Cyanobacterial GR6 glutamate-1-semialdehyde aminotransferase: a novel enzyme-based selectable marker for plant transformation. Plant Cell Rep 20:296–300. doi:10.1007/s002990100337
Grimm B, Smith AJ, Kannangara CG, Smith M (1991) Gabaculine resistant glutamate 1-semialdehyde aminotransferase of Synechococcus. J Biol Chem 266:12495–12501
Huang FC, Klaus SM, Herz S, Zou Z, Koop HU, Golds TJ (2002) Efficient plastid transformation in tobacco using the aphA-6 gene and kanamycin selection. Mol Genet Genomics 268:19–27. doi:10.1007/s00438-002-0738-6
Iamtham S, Day A (2000) Removal of antibiotic resistance genes from transgenic tobacco plastids. Nat Biotechnol 18:1172–1176. doi:10.1038/81161
Li W, Ruf S, Bock R (2011) Chloramphenicol acetyltransferase as selectable marker for plastid transformation. Plant Mol Biol 76:443–451. doi:10.1007/s11103-010-9678-4
Lutz KA, Knapp JE, Maliga P (2001) Expression of bar in the plastid genome confers herbicide resistance. Plant Physiol 125:1585–1590
Maliga P, Bock R (2011) Plastid biotechnology: food, fuel, and medicine for the 21st century. Plant Physiol 155:1501–1510. doi:10.1104/pp.110.170969
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Rogalski M, Ruf S, Bock R (2006) Tobacco plastid ribosomal protein S18 is essential for cell survival. Nucleic Acids Res 34:4537–4545. doi:10.1093/nar/gkl634
Rosellini D (2012) Selectable markers and reporter genes: a well furnished toolbox for plant science and genetic engineering. Crit Rev Plant Sci 31:401–453. doi:10.1080/07352689.2012.683373
Rosellini D, LaFayette P, Barone P, Veronesi F, Parrott WA (2004) Kanamycin resistant alfalfa has a point mutation in the plastid 16S rDNA. Plant Cell Rep 22:774–779. doi:10.1007/s00299-004-0757-3
Rosellini D, Capomaccio S, Ferradini N, Savo Sardaro ML, Nicolia A, Veronesi F (2007) Non-antibiotic, efficient selection for alfalfa genetic engineering. Plant Cell Rep 26:1035–1044. doi:10.1007/s00299-007-0321-z
Scotti N, Rigano M, Cardi T (2011) Production of foreign proteins using plastid transformation. Biotechnol Adv 30:387–397. doi:10.1016/j.biotechadv.2011.07.019
Scotti N, Bellucci M, Cardi T (2013) The chloroplasts as platform for recombinant proteins production. In: Duchêne A-M (ed) Translation in mitochondria and other organelle. Springer, Berlin, pp 225–262
Svab Z, Maliga P (1993) High-frequency plastid transformation in tobacco by selection for a chimeric aadA gene. Proc Natl Acad Sci USA 90:913–917. doi:10.1073/pnas.90.3.913
Svab Z, Hajdukiewicz P, Maliga P (1990) Stable transformation of plastids in higher plants. Proc Natl Acad Sci USA 87:8526–8530. doi:10.1073/pnas.87.21.8526
Verma D, Daniell H (2007) Chloroplast vector systems for biotechnology applications. Plant Physiol 145:1129–1143. doi:10.1104/pp.107.106690
Verma D, Samson NP, Koya V, Daniell H (2008) A protocol for expression of foreign genes in chloroplasts. Nat Protoc 3:739–758. doi:10.1038/nprot.2007.522
Watson J, Koya V, Leppla SH, Daniell H (2004) Expression of Bacillus anthracis protective antigen in transgenic chloroplasts of tobacco, a non-food/feed crop. Vaccine 22:4374–4384. doi:10.1016/j.vaccine.2004.01.069
Wei Z, Liu Y, Lin C, Wang Y, Cai Q, Dong Y, Xing S (2011) Transformation of alfalfa chloroplasts and expression of green fluorescent protein in a forage crop. Biotechnol Lett 33:2487–2494. doi:10.1007/s10529-011-0709-2
Ye GN, Hajdukiewicz PT, Broyles D, Rodriguez D, Xu CW, Nehra N, Staub JM (2001) Plastid-expressed 5-enolpyruvylshikimate-3-phosphate synthase genes provide high level glyphosate tolerance in tobacco. Plant J 25:261–270
Acknowledgments
The projects “Ricerca di Base 2015” (PI Daniele Rosellini) and “Coesistenza nell’agricoltura italiana” (PI Fabio Veronesi) of the University of Perugia are acknowledged for partial funding of this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by A. Dhingra.
Rights and permissions
About this article
Cite this article
Bellucci, M., De Marchis, F., Ferradini, N. et al. A mutant Synechococcus gene encoding glutamate 1-semialdehyde aminotransferase confers gabaculine resistance when expressed in tobacco plastids. Plant Cell Rep 34, 2127–2136 (2015). https://doi.org/10.1007/s00299-015-1856-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-015-1856-z