Abstract
Key message
A newly developed copper-inducible gene expression system overcame the mixed results reported earlier, worked well both in cultured cells and a whole plant, and enabled to control flowering timing.
Abstract
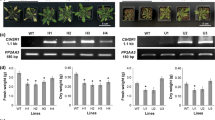
Copper is one of the essential microelements and is readily taken up by plants. However, to date, it has rarely been used to control the expression of genes of interest, probably due to the inefficiency of the gene expression systems. In this study, we successfully developed a copper-inducible gene expression system that is based on the regulation of the yeast metallothionein gene. This system can be applied in the field and regulated at approximately one-hundredth of the rate used for registered copper-based fungicides. In the presence of copper, a translational fusion of the ACE1 transcription factor with the VP16 activation domain (VP16AD) of herpes simplex virus strongly activated transcription of the GFP gene in transgenic Arabidopsis. Interestingly, insertion of the To71 sequence, a 5′-untranslated region of the 130k/180k gene of tomato mosaic virus, upstream of the GFP gene reduced the basal expression of GFP in the absence of copper to almost negligible levels, even in soil-grown plants that were supplemented with ordinary liquid nutrients. Exposure of plants to 100 μM copper resulted in an over 1,000-fold induction ratio at the transcriptional level of GFP. This induction was copper-specific and dose-dependent with rapid and reversible responses. Using this expression system, we also succeeded in regulating floral transition by copper treatment. These results indicate that our newly developed copper-inducible system can accelerate gene functional analysis in model plants and can be used to generate novel agronomic traits in crop species.








Similar content being viewed by others
References
Abe M, Kobayashi Y, Yamamoto S, Daimon Y, Yamaguchi A, Ikeda Y, Ichinoki H, Notaguchi M, Goto K, Araki T (2005) FD, a bZIP protein mediating signals from the floral pathway integrator FT at the shoot apex. Science 309:1052–1056
Amirsadeghi S, McDonald AE, Vanlerberghe GC (2007) A glucocorticoid-inducible gene expression system can cause growth defects in tobacco. Planta 226:453–463
Andersen SU, Cvitanich C, Hougaard BK, Roussis A, Gronlund M, Jensen DB, Frokjaer LA, Jensen EO (2003) The glucocorticoid-inducible GVG system causes severe growth defects in both root and shoot of the model legume Lotus japonicus. Mol Plant Microbe Interact 16:1069–1076
Aoyama T, Chua NH (1997) A glucocorticoid-mediated transcriptional induction system in transgenic plants. Plant J 11:605–612
Boetti H, Chevalier L, Denmat LA, Thomas D, Thomasset B (1999) Efficiency of physical (light) or chemical (ABA, tetracycline, CuSO4 or 2-CBSU)-stimulus-dependent gus gene expression in tobacco cell suspensions. Biotechnol Bioeng 64:1–13
Caddick MX, Greenland AJ, Jepson I, Krause KP, Qu N, Riddell KV, Salter MG, Schuch W, Sonnewald U, Tomsett AB (1998) An ethanol inducible gene switch for plants used to manipulate carbon metabolism. Nat Biotechnol 16:177–180
Corbesier L, Vincent C, Jang S, Fornara F, Fan Q, Searle I, Giakountis A, Farrona S, Gissot L, Turnbull C, Coupland G (2007) FT protein movement contributes to long-distance signaling in floral induction of Arabidopsis. Science 316:1030–1033
Corrado G, Karali M (2009) Inducible gene expression systems and plant biotechnology. Biotechnol Adv 27:733–743
Culotta VC, Hsu T, Hu S, Furst P, Hamer D (1989) Copper and the ACE1 regulatory protein reversibly induce yeast metallothionein gene transcription in a mouse extract. Proc Natl Acad Sci USA 86:8377–8381
Czarnecka E, Key JL, Gurley WB (1989) Regulatory domains of the Gmhsp17.5-E heat shock promoter of soybean. Mol Cell Biol 9:3457–3463
Feinbaum RL, Storz G, Ausubel FM (1991) High intensity and blue light regulated expression of chimeric chalcone synthase genes in transgenic Arabidopsis thaliana plants. Mol Gen Genet 226:449–456
Furst P, Hu S, Hackett R, Hamer D (1988) Copper activates metallothionein gene transcription by altering the conformation of a specific DNA binding protein. Cell 55:705–717
Gallie DR, Walbot V, Hershey JW (1988) The ribosomal fraction mediates the translational enhancement associated with the 5′-leader of tobacco mosaic virus. Nucleic Acids Res 16:8675–8694
Gardner MJ, Baker AJ, Assie JM, Poethig RS, Haseloff JP, Webb AAR (2009) GAL4 GFP enhancer trap lines for analysis of stomatal guard cell development and gene expression. J Exp Bot 60:213–226
Gatz C, Frohberg C, Wendenburg R (1992) Stringent repression and homogeneous de-repression by tetracycline of a modified CaMV 35S promoter in intact transgenic tobacco plants. Plant J 2:397–404
Granger CL, Cyr RJ (2000) Microtubule reorganization in tobacco BY-2 cells stably expressing GFP-MBD. Planta 210:502–509
Granger CL, Cyr RJ (2001) Characterization of the yeast copper-inducible promoter system in Arabidopsis thaliana. Plant Cell Rep 20:227–234
Gunl M, Liew EF, David K, Putterill J (2009) Analysis of a post-translational steroid induction system for GIGANTEA in Arabidopsis. BMC Plant Biol 9:141
Jaeger KE, Wigge PA (2007) FT protein acts as a long-range signal in Arabidopsis. Curr Biol 17:1050–1054
Jung C, Muller AE (2009) Flowering time control and applications in plant breeding. Trends Plant Sci 14:563–573
Kang HG, Fang Y, Singh KB (1999) A glucocorticoid-inducible transcription system causes severe growth defects in Arabidopsis and induces defense-related genes. Plant J 20:127–133
Kardailsky I, Shukla VK, Ahn JH, Dagenais N, Christensen SK, Nguyen JT, Chory J, Harrison MJ, Weigel D (1999) Activation tagging of the floral inducer FT. Science 286:1962–1965
Kobayashi Y, Kaya H, Goto K, Iwabuchi M, Araki T (1999) A pair of related genes with antagonistic roles in mediating flowering signals. Science 286:1960–1962
Labbe S, Thiele DJ (1999) Copper ion inducible and repressible promoter systems in yeast. Methods Enzymol 306:145–153
Lam E, Benfey PN, Gilmartin PM, Fang RX, Chua NH (1989) Site-specific mutations alter in vitro factor binding and change promoter expression pattern in transgenic plants. Proc Natl Acad Sci USA 86:7890–7894
Lin MK, Belanger H, Lee YJ, Varkonyi-Gasic E, Taoka K, Miura E, Xoconostle-Cázares B, Gendler K, Jorgensen RA, Phinney B, Lough TJ, Lucas WJ (2007) FLOWERING LOCUS T protein may act as the long-distance florigenic signal in the cucurbits. Plant Cell 19:1488–1506
Mathieu J, Warthmann N, Küttner F, Schmid M (2007) Export of FT protein from phloem companion cells is sufficient for floral induction in Arabidopsis. Curr Biol 17:1055–1060
McKenzie MJ, Mett VV, Stewart Reynolds PH, Jameson PE (1998) Controlled cytokinin production in transgenic tobacco using a copper-inducible promoter. Plant Physiol 116:969–977
Mett VL, Lochhead LP, Reynolds PH (1993) Copper-controllable gene expression system for whole plants. Proc Natl Acad Sci USA 90:4567–4571
Mett VL, Podivinsky E, Tennant AM, Lochhead LP, Jones WT, Reynolds PH (1996) A system for tissue-specific copper-controllable gene expression in transgenic plants: nodule-specific antisense of aspartate aminotransferase-P2. Transgenic Res 5:105–113
Mohamed R, Meilan R, Strauss SH (2001) Complex behavior of a copper-inducible gene expression system in transgenic poplar. For Genet 8:69–72
Moore I, Samalova M, Kurup S (2006) Transactivated and chemically inducible gene expression in plants. Plant J 45:651–683
Notaguchi M, Abe M, Kimura T, Daimon Y, Kobayashi T, Yamaguchi A, Tomita Y, Dohi K, Mori M, Araki T (2008) Long-distance, graft-transmissible action of Arabidopsis FLOWERING LOCUS T protein to promote flowering. Plant Cell Physiol 49:1645–1658
Odell JT, Nagy F, Chua NH (1985) Identification of DNA sequences required for activity of the cauliflower mosaic virus 35S promoter. Nature 313:810–812
Okuzaki A, Konagaya K, Nanasato Y, Tsuda M, Tabei Y (2011) Estrogen-inducible GFP expression patterns in rice (Oryza sativa L.). Plant Cell Rep 30:529–538
Padidam M (2003) Chemically regulated gene expression in plants. Curr Opin Plant Biol 6:169–177
Pena MM, Koch KA, Thiele DJ (1998) Dynamic regulation of copper uptake and detoxification genes in Saccharomyces cerevisiae. Mol Cell Biol 18:2514–2523
Roslan HA, Salter MG, Wood CD, White MR, Croft KP, Robson F, Coupland G, Doonan J, Laufs P, Tomsett AB, Caddick MX (2001) Characterization of the ethanol-inducible alc gene-expression system in Arabidopsis thaliana. Plant J 28:225–235
Semenyuk EG, Schmidt MA, Beachy RN, Moravec T, Woodford-Thomas T (2010) Adaptation of an ecdysone-based genetic switch for transgene expression in soybean seeds. Transgenic Res 19:987–999
Takeda Y, Hirokawa H, Nagata T (1992) The replication origin of proplastid DNA in cultured cells of tobacco. Mol Gen Genet 232:191–198
Tamai A, Meshi T (2001) Tobamoviral movement protein transiently expressed in a single epidermal cell functions beyond multiple plasmodesmata and spreads multicellularly in an infection-coupled manner. Mol Plant Microbe Interact 14:126–134
Tamaki S, Matsuo S, Wong HL, Yokoi S, Shimamoto K (2007) Hd3a protein is a mobile flowering signal in rice. Science 316:1033–1036
Tang W, Luo X, Samuels V (2004) Regulated gene expression with promoters responding to inducers. Plant Sci 166:827–834
Tavva VS, Dinkins RD, Palli SR, Collins GB (2006) Development of a methoxyfenozide-responsive gene switch for applications in plants. Plant J 45:457–469
Tavva VS, Palli SR, Dinkins RD, Collins GB (2007) Applications of EcR gene switch technology in functional genomics. Arch Insect Biochem Physiol 65:164–179
Van de Rhee MD, Van Kan JA, Gonzalez-Jaen MT, Bol JF (1990) Analysis of regulatory elements involved in the induction of two tobacco genes by salicylate treatment and virus infection. Plant Cell 2:357–366
Wigge PA, Kim MC, Jaeger KE, Busch W, Schmid M, Lohmann JU, Weigel D (2005) Integration of spatial and temporal information during floral induction in Arabidopsis. Science 309:1056–1059
Wyatt RE, Ainley WM, Nagao RT, Conner TW, Key JL (1993) Expression of the Arabidopsis AtAu2–11 auxin-responsive gene in transgenic plants. Plant Mol Biol 22:731–749
Yamaguchi-Shinozaki K, Shinozaki K (1993) Characterization of the expression of a desiccation-responsive rd29 gene of Arabidopsis thaliana and analysis of its promoter in transgenic plants. Mol Gen Genet 236:331–340
Yamamoto M, Torikai S, Oeda K (1997) A major root protein of carrots with high homology to intracellular pathogenesis-related (PR) proteins and pollen allergens. Plant Cell Physiol 38:1080–1086
Yang J, Ordiz MI, Semenyuk EG, Kelly B, Beachy RN (2012) A safe and effective plant gene switch system for tissue-specific induction of gene expression in Arabidopsis thaliana and Brassica juncea. Transgenic Res 21:879–883
Yeoh CC, Balcerowicz M, Laurie R, Macknight R, Putterill J (2011) Developing a method for customized induction of flowering. BMC Biotechnol 11:36
Zuo J, Chua NH (2000) Chemical-inducible systems for regulated expression of plant genes. Curr Opin Biotechnol 11:146–151
Zuo J, Niu QW, Chua NH (2000) Technical advance: an estrogen receptor-based transactivator XVE mediates highly inducible gene expression in transgenic plants. Plant J 24:265–273
Acknowledgments
We are grateful to Dr. Y. Niwa of Shizuoka Prefecture University for the kind gift of the 35S-sGFP(S65T) vector, and Dr. M. Mori of Ishikawa Prefecture University for the kind gift of piL.erG3. We thank Dr. R. Sato and Dr. T. Adachi for valuable discussions and suggestions, Dr. Y. Kitamura of Nagasaki University for useful advice on manuscript preparation, and Ms. T. Chikahisa, Ms. T. Nakamura, Ms. M. Uda, Ms. E. Tanaka, Ms. H. Hagita, Mr. M Toya, and Mr. R. Fukui for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by K. Toriyama.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1-4 (MOESM1–4) GFP fluorescent (upper) and bright-field (lower) images of T3 seedlings with or without 3 days of treatment with 100 μM copper. Transgenic plants that were transformed with pSUM21-sGFP, pSUM24-sGFP, pSUM44-sGFP, pSUM48-sGFP, pSUM46-sGFP, and pBI-35S-sGFP are represented by the numbers 21, 24, 44, 48, 46, and 91, respectively. For each construct, 6–9 independent homozygous lines are displayed. Table S1 (MOESM5) A list of all primers and probes used in PCR experiments. Supplementary Materials and Methods (MOESM6) plasmid construction.
Rights and permissions
About this article
Cite this article
Saijo, T., Nagasawa, A. Development of a tightly regulated and highly responsive copper-inducible gene expression system and its application to control of flowering time. Plant Cell Rep 33, 47–59 (2014). https://doi.org/10.1007/s00299-013-1511-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-013-1511-5