Abstract
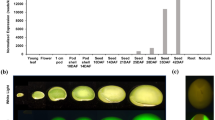
Soybean was used as a model for studies of chemical induction of gene expression in seeds. A chimeric transcriptional activator, VGE, driven by the soybean seed glycinin G1 promoter, was used to induce the expression of an ER-targeted GFPKDEL reporter protein upon addition of the chemical ligand, methoxyfenozide. The chemical gene switch activated gene expression under in vitro conditions in somatic cotyledonary embryos and zygotic seed embryos cultured from transgenic soybean plants, as well as in seeds in planta under greenhouse conditions. The efficiency of induction of GFP expression under different growth conditions was strongly influenced by the developmental stage of the seed and availability of the inducer. The formation of ER-derived GFP-containing protein bodies in seed storage parenchyma cells was correlated with the level of induced expression.






Similar content being viewed by others
Abbreviations
- TSP:
-
Total soluble protein
- MOF:
-
Methoxyfenozide
- ER:
-
Endoplasmic reticulum
- 35SCaMV promoter:
-
Cauliflower mosaic virus 35S RNA promoter
References
Andersen SU, Cvitanich C, Hougaard BK et al (2003) The glucocorticoid-inducible GVG system causes severe growth defects in both root and shoot of the model legume Lotus japonicus. Mol Plant Microbe Interact 16:1069–1076
Aoyama T, Chua NH (1997) A glucocorticoid-mediated transcriptional induction system in transgenic plants. Plant J 11:605–612
Brand L, Hörler M, Nüesch E et al (2006) A versatile and reliable two-component system for tissue-specific gene induction in Arabidopsis. Plant Physiol 141:1194–1204
Dai S, Zheng P, Marmey P et al (2001) Comparative analysis of transgenic rice plants obtained by Agrobacterium-mediated transformation and particle bombardment. Mol Breed 7:25–33
Dietrich CR, Han G, Chen M et al (2008) Loss-of-function mutations and inducible RNAi suppression of Arabidopsis LCB2 genes reveal the critical role of sphingolipids in gametophytic and sporophytic cell viability. Plant J 54:284–298
Egli DB, Fraser J, Leggett JE et al (1981) Control of Seed Growth in Soya Beans [Glycine max (L.) Merrill]. Ann Bot 48:171–176
Fischer RL, Goldberg RB (1982) Structure and flanking regions of soybean seed protein genes. Cell 29:651–660
Gälweiler L, Conlan RS, Mader P et al (2000) Technical advance: the DNA-binding activity of gal4 is inhibited by methylation of the gal4 binding site in plant chromatin. Plant J 23:143–157
Garbarino JE, Belknap WR (1994) Isolation of a ubiquitin-ribosomal protein gene (ubi3) from potato and expression of its promoter in transgenic plants. Plant Mol Biol 24:119–127
Herman EM, Tague BW, Hoffman LM et al (1990) Retention of phytohemagglutinin with carboxyterminal tetrapeptide KDEL in the nuclear envelope and the endoplasmic reticulum. Planta 182:305–312
Holowach LP, Thompson JF, Madison JT (1984) Storage protein composition of soybean cotyledons grown in vitro in media of various sulfate concentrations in the presence and absence of exogenous L-methionine. Plant Physiol 74:584–589
Hughes EH, Hong S-B, Gibson SI et al (2004) Expression of a feedback-resistant anthranilate synthase in Catharanthus roseus hairy roots provides evidence for tight regulation of terpenoid indole alkaloid levels. Biotech Bioeng 86:18–27
Koo JC, Asurmendi S, Bick J et al (2004) Ecdysone agonist-inducible expression of a coat protein gene from tobacco mosaic virus confers viral resistance in transgenic Arabidopsis. Plant J 37:439–448
Kourtz L, Dillon K, Daughtry S et al (2007) Chemically inducible expression of the PHB biosynthetic pathway in Arabidopsis. Transgenic Res 16:759–769
Martinez A, Sparks C, Hart CA et al (1999) Ecdysone agonist inducible transcription in transgenic tobacco plants. Plant J 19:97–106
Meinke DW, Chen J, Beachy RN (1981) Expression of storage-protein genes during soybean seed development. Planta 153:130–139
Miernyk JA, Johnston ML, Antoine W et al (2009) A directed-proteomics platform for systems analysis of soybean seed development. J Integr Plant Biol (in press)
Moore I, Samalova M, Kurup S (2006) Transactivated and chemically inducible gene expression in plants. Plant J 45:651–683
Moravec T, Schmidt MA, Herman EM et al (2007) Production of Escherichia coli heat labile toxin (LT) B subunit in soybean seed and analysis of its immunogenicity as an oral vaccine. Vaccine 25:1647–1657
Muasya RM, Lommen WJM, Auma EO et al (2006) Relationship between variation in quality of individual seeds and bulk seed quality in common bean (Phaseolus vulgaris L.) seed lots. NJAS 54:5–16
Nielsen NC, Dickinson CD, Cho TJ et al (1989) Characterization of the glycinin gene family in soybean. Plant Cell 1:313–328
Ouwerkerk PB, de Kam RJ, Hoge JH et al (2001) Glucocorticoid-inducible gene expression in rice. Planta 213:370–378
Padidam M (2003) Chemically regulated gene expression in plants. Current Opin Plant Biol 6:169–177
Padidam M, Gore M, Lu DL et al (2003) Chemical-inducible, ecdysone receptor-based gene expression system for plants. Transgenic Res 12:101–109
Perez-Grau L, Goldberg RB (1989) Soybean seed protein genes are regulated spatially during embryogenesis. Plant Cell 1:1095–1109
Reddy MS, Dinkins RD, Collins GB (2003) Gene silencing in transgenic soybean plants transformed via particle bombardment. Plant Cell Rep 21:676–683
Saab IN, Obendorf RL (1989) Soybean seed water relations during in situ and in vitro growth and maturation. Plant Physiol 89:610–616
Samac DA, Hironaka CM, Yallaly PE et al (1990) Isolation and characterization of the genes encoding basic and acidic chitinase in Arabidopsis thaliana. Plant Physiol 93:907–914
Schardl CL, Byrd AD, Benzion G, Altschuler MA et al (1987) Design and construction of a versatile system for the expression of foreign genes in plants. Gene 61:1–11
Schmidt MA, Herman EM (2008) Proteome rebalancing in soybean seeds can be exploited to enhance foreign protein accumulation. Plant Biotechnol J 6:832–842
Schmidt MA, Tucker DM, Cahoon EB et al (2005) Towards normalization of soybean somatic embryo maturation. Plant Cell Rep 24:383–391
Sims TL, Goldberg RB (1989) The glycinin Gy1 gene from soybean. Nucleic Acids Res 17:4386
Sudarshana MR, Plesha MA, Uratsu SL et al (2006) A chemically inducible cucumber mosaic virus amplicon system for expression of heterologous proteins in plant tissues. Plant Biotech J 4:551–559
Sun J, Niu QW, Tarkowski P (2003) The Arabidopsis AtIPT8/PGA22 gene encodes an isopentenyl transferase that is involved in de novo cytokinin biosynthesis. Plant Physiol 131:167–176
Tang W, Newton RJ (2004) Glucocorticoid-inducible transgene expression in loblolly pine (Pinus taeda L.) cell suspension cultures. Plant Sci 166:1351–1358
Tavva VS, Dinkins RD, Palli SR et al (2006) Development of a methoxyfenozide-responsive gene switch for applications in plants. Plant J 45:457–469
Thibaud-Nissen F, Shealy RT, Khanna A et al (2003) Clustering of microarray data reveals transcript patterns associated with somatic embryogenesis in soybean. Plant Physiol 132:118–136
Thompson JF, Madison JT, Muenster A-ME (1977) In vitro culture of immature cotyledons of soya bean (Glycine max L. Merr.). Ann Bot 41:29–39
Trick HN, Dinkins RD, Santarem ER et al (1997) Recent advances in soybean transformation. Plant Tissue Cult Biotech 3:9–26
Unger E, Cigan AM, Trimnell M et al (2002) A chimeric ecdysone receptor facilitates methoxyfenozide-dependent restoration of male fertility in ms45 maize. Transgenic Res 11:455–465
Wang TL, Domoney C, Hedley CL et al (2003) Can we improve the nutritional quality of legume seeds? Plant Physiol 131:886–891
Wielopolska A, Townley H, Moore I et al (2005) A high-throughput inducible RNAi vector for plants. Plant Biotech J 3:583–590
Wolswinkel P (1992) Transport of nutrients into developing seeds: a review of physiological mechanisms. Seed Sci Res 2:59–73
Zuo J, Niu QW, Ikeda Y et al (2002) Marker-free transformation: increasing transformation frequency by the use of regeneration-promoting genes. Curr Opin Biotechnol 13:173–180
Acknowledgments
Dr. Eliot Herman is gratefully acknowledged for providing stimulating discussions and intellectual support. We also thank Dr. Howard Berg in the Integrated Microscopy Facility and Kevin Lutke from Tissue Culture Facility at the Danforth Plant Science Center for their kind assistance throughout this study, and Dr. Mala Padidam (Rohm and Haas Company), Dr. Wayne Parrott (University of Georgia) and Dr. Ed Cahoon (University of Nebraska) for providing certain genetics components for the gene switch and expression systems. This work was supported by grants from the USDA/Illinois-Missouri Alliance for Biotechnology, IMBA (TWT; 2006-5) and from The Consortium for Plant Biotechnology Research, Inc. (CPBR, Inc.) (RNB; GO12026-266).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Semenyuk, E.G., Schmidt, M.A., Beachy, R.N. et al. Adaptation of an ecdysone-based genetic switch for transgene expression in soybean seeds. Transgenic Res 19, 987–999 (2010). https://doi.org/10.1007/s11248-010-9377-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-010-9377-6