Abstract
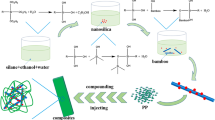
In this elucidation, we synthesized novel molecularly imprinted polymers (MIPOH1-4) that were used as organic fillers on bagasse pulp to form cellulosic paper sheets, the formation of MIPOH1-4 from the reaction of enaminone 3 with methyl methacrylate monomer in the presence of ethylene glycol dimethyl acrylate and 2,2’-azobisisobutyronitrile. The obtained MIPOH1-4 was confirmed via FT-IR and SEM with different pore sizes. And we noticed that the increase of template 3 increases the hydrogen bond between monomer and template and gave sponge surface of MIPOH4. Furthermore, the computational energies of the template with different monomers utilize DFT/B3LYP/6-31G (d) level to know the stability of polymers and hydrogen bonding between them. So that we applied that in paper manufacture through the adsorption of MIPOH4 as an organic filler on bagasse pulp to form novel cellulosic paper sheets with different concentrations and these sheets which characteristic through FT-IR, SEM, sizing test, tensile strength, and elucidation their antimicrobial activity with theoretical and docking studies. The MIPOH4 and cellulosic paper sheets showed the thermal stability and high tendency of sizability of these paper sheets and concluded the interaction between cellulose fibers of pulp with MIPOH polymer with physical hydrogen bonding.







Similar content being viewed by others
References
Kumar A, Negi YS, Choudhary V, Bhardwaj NK (2014) Characterization of cellulose nanocrystals produced by acid-hydrolysis from sugarcane bagasse as agro-waste. J Mater Phys Chem 2(1):1–8
Ibrahim MA, Mobarak F, Salah El-Din EI, Ebaid A, Youssef MA (2009) Modified Egyptian talc as internal sizing agent for papermaking. J Carbohydr Polym 75:130–134
Dacrory S, Fahim AM (2020) Synthesis, anti-proliferative activity, computational studies of tetrazole cellulose utilizing different homogenous catalyst. Carbohydr Polym 229:115537
Fahim AM, Mohamed A, Ibrahim MA (2021) Experimental and theoretical studies of some propiolate esters derivatives. J Mol Struct 1236:130281. https://doi.org/10.1016/j.molstruc.2021.130281
Fahim AM, Farag AM, Mermer A, Bayrak H, Şirin Y (2021) Synthesis of novel β-lactams: antioxidant activity, acetylcholinesterase inhibition and computational studies. J Mol Struct 1233:130092. https://doi.org/10.1016/j.molstruc.2021.130092
Wondraczek H, Petzold-Welcke K, Fardim P et al (2013) Nanoparticles from conventional cellulose esters: evaluation of preparation methods. Cellulose 20:751–760
Kostag M, Köhler S, Liebert T et al (2010) Pure cellulose nanoparticles from trimethylsilyl cellulose. Macromol Symp 294(2):96–106
Schulz L, Burchard W, Dönges R (1998) Evidence of supramolecular structures of cellulose derivatives in solution. In: Heinze T, Glasser WG (eds) Cellulose derivatives: modification, characterization, and nanostructures, vol 688. ACS symposium series. American Chemical Society, Washington DC, pp 218–238
Mehanny S, Abu-El Magd EE, Ibrahim M, Farag M, Gil-San-Millan R, Navarro J, El-Habbak AEH, El-Kashif E (2021) Extraction and characterization of nanocellulose from three types of palm residues. J Mater Res Technol. https://doi.org/10.1016/j.jmrt.2020.12.027
Zhao Chengke, Zhang Hongjie, Zeng Xu, Li Hailong, Sun Dongyun (2016) Enhancing the inter-fiber bonding properties of cellulosic fibers by increasing different fiber charges. Cellulose. https://doi.org/10.1007/s10570-016-0941-y
Hu X, Pan J, Hu Y, Huo Y, Li G (2009) Preparation and evaluation of solid-phase microextraction fiber based on molecularly imprinted polymers for trace analysis of tetracyclines in complicated samples. J Chromatogr A 1216:190–197
Sellergren B (2001) Molecularly imprinted polymers. Man-made. Elsevier, Amsterdam, p 113
Farrington K, Regan F (2007) Investigation of the nature of MIP recognition: the development and characterization of a MIP for Ibuprofen. Biosens Bioelectron 22:1138–1146
Fahim AM, Wasiniak B, Łukaszewicz JP (2020) Molecularly imprinted polymer and computational study of (E)-4-(2- cyano-3-(dimethylamino)acryloyl)benzoic acid from poly(ethylene terephthalate) plastic waste. Curr Anal Chem 16(2):119–137
Balakrishna C, Kandula V, Gudipati R, Yennam S, Uma Devi P, Behera M (2018) An efficient microwave-assisted propylphosphonic anhydride (T3P®)-mediated one-pot chromone synthesis via enaminones. Synlett 29(08):1087–1091. https://doi.org/10.1055/s-0036-1591898
Akl E, Dacrory S, Kamel Mohamed, Fahim AM (2020) Preparation and characterization of novel antibacterial blended films based on modified carboxymethyl cellulose/phenolic compounds. Polym Bull. https://doi.org/10.1007/s00289-020-03148-w
Peng C, Ayala PY, Schlegel HB, Frisch MJ (1996) Using redundant internal coordinates to optimize equilibrium geometries and transition states. J Comput Chem 17:49–56
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Hratchian X, Li HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, revision a. 1. Gaussian, Inc., Wallingford
Scott AP, Radom L (1996) Harmonic vibrational Frequencies: an evaluation of Hartree-Fock, MøllerPlesset, quadratic configuration interaction, density functional theory, and semiempirical scale factors. J Phys Chem 100:16502–16513
Foresman J, Frish E (1996) Exploring chemistry. Gaussian Inc., Pittsburg
Dennington R, Keith T, Millam J (2009) GaussView, version 5. Semichem Inc., Shawnee Mission
Fahim AM, Ismael EHI (2019) Synthesis, antimicrobial activity and quantum calculations of novel sulphonamide derivatives. Egypt J Chem 62(8):1427–1440
Ray GB, Cook JW (2005) Molecular modeling of heme proteins using MOE: bio-inorganic and structure-function activity for undergraduates*S. Biochem Mol Biol Educ 33:194–201. https://doi.org/10.1002/bmb.2005.494033032449
Monika P, Umesh K, Michael B (2014) Docking simulations, molecular properties and ADMET studies of novel Chromane-6,7-diol analogues as potential inhibitors of Mushroom tyrosinase. Gene Ther Mol Biol 16:201–217
Morris GM, Huey R, Lindstrom W, Sanner M, Belew MF, Goodsell RK, Olson AJ (2009) Autodock4 and AutoDockTools4: automated docking with selective receptor flexibility. J Comput Chem 16:2785–2791
Li HJ, Zhang N, Truglio JJH, Mishra S, Kisker C, Garcia-Diaz M, Tonge PJ (2011) Mechanism of the intramolecular claisen condensation reaction catalyzed by MenB, a crotonase superfamily member. Biochemistry 50:9532–9544
Hagelueken G, Huang H, Mainprize IL, Whitfield C, Naismith JH (2009) Crystal structures of Wzb of Escherichia Coli and Cpsb of streptococcus pneumoniae, representatives of two families of tyrosine phosphatases that regulate capsule assembly. J Mol Biol 392(2009):678
Yoshida H, Sakai G, Mori K, Kojima K, Kamitori S, Sode K (2015) Structural analysis of fungus-derived FAD glucose dehydrogenase. Sci Rep 5:13498–13498
Juo ZS, Chiu TK, Leiberman PM, Baikalov I, Berk AJ, Dickerson RE (1996) How proteins recognize the TATA box. J Mol Biol 261:239–254
Chattopadhyay B, Ghosh S, Mondal S, Mukherjee M, Mukherjee A (2012) Structural study of three o-hydroxy acetophenone derivatives using X-ray powder diffraction: interplay of weak intermolecular interactions. CrystEngComm 14:837–846. https://doi.org/10.1039/C1CE05920C
Fahim AM, Elshikh MS, Darwish NM (2020) Synthesis, antitumor activity, molecular docking and DFT study of novel pyrimidiopyrazole derivatives. Curr Comput-Aided Drug Des 16(4):486–499
Fahim AM, Farag AM (2020) Synthesis, antimicrobial evaluation, molecular docking and theoretical calculations of novel pyrazolo[1,5-a]pyrimidine derivatives. J Mol Struct 1199:127025
Fahim AM, Farag AM, Yakout EMA, Nawwar GA, Ragab EA (2018) Sundegradation and synthesis of new antimicrobial and antioxidant utilizing poly(ethylene terephthalate) waste. Int J Environ Waste Manag 22:239–259
Fahim AM, Farag AM, Yakout EMA, Nawwar GAM, Ragab EA (2016) Synthesis, biological evaluation of 1,3,4-oxadiazole, triazole and uracil derivatives from poly (ethylene terephthalate) waste. Egypt J Chem 59:285–303
Fahim AM, Shalaby MA, Ibrahim MA (2019) Microwave-assisted synthesis of novel 5-aminouracil-based compound with DFT calculations. J Mol Struct 2019(1194):211–226
Fahim AM, Shalaby MA (2019) Synthesis, biological evaluation, molecular docking and DFT calculations of novel benzenesulfonamide derivatives. J Mol Struct 1176:408–421
Fahim AM, Yakout EAM, Nawwar GA (2014) Facile synthesis of in-vivo insecticidal and antimicrobial evaluation of bis heterocyclic moiety from pet waste. Online J Biol Sci 14:196–208
Farag AM, Fahim AM (2019) Synthesis, biological evaluation and DFT calculation of novel pyrazole and pyrimidine derivatives. J Mol Struct 1179:304–314
Runge E, Gross EKU (1984) Density-functional theory for time-dependent systems. Phys Rev Lett 52:997–1000
Ibrahim MM, Dufresne A, El-Zawawy WK, Agblevor FA (2010) Banana fibers and microfibrils as lignocellulosic reinforcements in polymer composites. Carbohydr Polym 81(4):811–819
Soror S, Fahim AM, Elabbady S, Nassar E, Aboelnaga A (2021) Synthesis, antimicrobial activities, docking studies and computational calculations of new bis-1,4-phenylene-1H-1,2,3-triazole derivatives utilized ultrasonic energy. J Biomol Struct Dyn. https://doi.org/10.1080/07391102.2021.1875051
Saha SK, Hens A, RoyChowdhury A, Lohar AK, Murmu NC, Banerjee B (2014) Molecular dynamics and density functional theory study on corrosion inhibitory action of three substituted pyrazine derivatives on steel surface. Can Chem Trans 2(4):489–503
Griffith JS, Orgel LE (1957) Ligand-field theory. Q Rev Chem Soc 11:381–393
Hasan M, Lai TK, Chong EWN, Gopakumar DA, Rizal S, Hossain MS, Nurul Fazita MR, Haafiz MKM, Paridah MT, Abdul Khalil HPS (2019) Organic and inorganic fillers’ role on the amelioration of Kappaphycus spp.-based biopolymer films’ performance. BioResources 14(4):9198–9213
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Andrews JM (2001) Determination of minimum inhibitory concentrations. J Antimicrob Chemother 48:5–16
Tanak H, Toy M (2013) Molecular structure, vibrational spectra, NLO and MEP analysis of bis[2-hydroxy-кO-N-(2-pyridyl)-1-naphthaldiminato-кN]zinc(II). Spectrochim Acta Mol Biomol Spectrosc 115:145–153
Yoon S-D, Byun H-S (2013) Molecularly imprinted polymers for selective separation of acetaminophen and aspirin by using supercritical fluid technology. Chem Eng J 226:171–180
Fahim AM, Elshikh MS, Darwish NM (2020) Synthesis, antitumor activity, molecular docking and DFT study of novel pyrimidiopyrazole derivatives. Curr Comput Aided Drug Des 16(4):486–499. https://doi.org/10.2174/1573409915666190710094425
Aboelnaga A, Fahim AM, El-Sayed TH (2020) Computer aid screening for potential antimalarial choroquinone compounds as Covid 19 utilizing computational calculations and molecular docking study. Online J Biol Sci 20(4):207–220. https://doi.org/10.3844/ojbsci.2020.207.220
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fahim, A.M., Abu-El Magd, E.E. Performance efficiency of MIPOH polymers as organic filler on cellulose pulp waste to form cellulosic paper sheets with biological evaluation and computational studies. Polym. Bull. 79, 4099–4131 (2022). https://doi.org/10.1007/s00289-021-03685-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-021-03685-y



