Abstract
Purpose
To investigate the ability of 18F-FDG PET/CT to assess the response of patients with metastatic melanoma to immunotherapy.
Methods
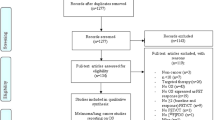
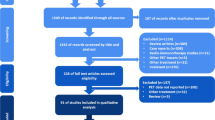
A comprehensive search of the literature for studies examining the prognostic value of 18F-FDG PET/CT in monitoring the response of patients with metastatic melanoma to immunotherapy was performed. We also screened the references of the selected articles to identify any other relevant studies. Detailed data were extracted and categorized. Comprehensive meta-analysis software was used for analysis.
Results
Twenty four eligible articles were included in the systematic review. Based on the baseline 18F-FDG PET/CT imaging, the pooled hazard ratios of MTV, SLR, SUV/SULmax, SUV/SULpeak, and TLG for overall survival (OS) were 1.777 (95%CI: 1.389–2.275, p < 0.001), 3.425 (95%CI: 1.707–6.869, p = 0.001), 0.941 (95%CI: 0.599–1.477, p = 0.791), 1.704 (95%CI: 1.253–2.316, p = 0.016), and 1.755 (95%CI: 1.315–2.342, p < 0.001), respectively. The conventional and modified response assessment criteria exhibited a pooled sensitivity of 64% (95%CI: 46–79%) and 94% (95%CI: 81–99%) and a pooled specificity of 80% (95%CI: 59–93%) and 84% (95%CI: 64–95%), respectively, for the early 18F-FDG PET/CT scan. On the other hand, based on the late 18F-FDG PET/CT scan, the pooled sensitivity of 67% (95%CI: 35–90%) and 92% (95%CI: 73–99%) and pooled specificity of 77% (95%CI: 56–91%) and 76% (95%CI: 50–93%) were observed for the conventional and modified criteria, respectively. PET-detectable immune-related adverse events (irAEs) were associated with the response to therapy.
Conclusions
The baseline SUVpeak, MTV, and TLG parameters represent promising predictors of the final response of metastatic melanoma patients to immunotherapy. Modified response assessment criteria are potentially an appropriate method for monitoring immunotherapy. irAEs are also valuable for predicting eventual clinical benefit of treatment.







Similar content being viewed by others
Data availability
Data are available upon request.
References
Rossi S, Toschi L, Castello A, Grizzi F, Mansi L, Lopci E. Clinical characteristics of patient selection and imaging predictors of outcome in solid tumors treated with checkpoint-inhibitors. Eur J Nucl Med Mol Imaging. 2017;44(13):2310–25. https://doi.org/10.1007/s00259-017-3802-5.
Bauckneht M, Piva R, Sambuceti G, Grossi F, Morbelli S. Evaluation of response to immune checkpoint inhibitors: is there a role for positron emission tomography? World J Radiol. 2017;9(2):27–33. https://doi.org/10.4329/wjr.v9.i2.27.
Guldbrandsen KF, Hendel HW, Langer SW, Fischer BM. Nuclear molecular imaging strategies in immune checkpoint inhibitor therapy. Diagnostics (Basel). 2017;7(2). https://doi.org/10.3390/diagnostics7020023.
Kwak JJ, Tirumani SH, Van den Abbeele AD, Koo PJ, Jacene HA. Cancer immunotherapy: imaging assessment of novel treatment response patterns and immune-related adverse events. Radiographics. 2015;35(2):424–37. https://doi.org/10.1148/rg.352140121.
McCracken MN, Tavare R, Witte ON, Wu AM. Advances in PET detection of the antitumor T cell response. Adv Immunol. 2016;131:187–231. https://doi.org/10.1016/bs.ai.2016.02.004.
Gilardi L, Grana CM, Paganelli G. Evaluation of response to immunotherapy: new challenges and opportunities for PET imaging. Eur J Nucl Med Mol Imaging. 2014;41(11):2090–2. https://doi.org/10.1007/s00259-014-2848-x.
Perng P, Marcus C, Subramaniam RM. (18)F-FDG PET/CT and melanoma: staging, immune modulation and mutation-targeted therapy assessment, and prognosis. AJR Am J Roentgenol. 2015;205(2):259–70. https://doi.org/10.2214/AJR.14.13575.
Solinas C, Porcu M, Hlavata Z, De Silva P, Puzzoni M, Willard-Gallo K, et al. Critical features and challenges associated with imaging in patients undergoing cancer immunotherapy. Crit Rev Oncol Hematol. 2017;120:13–21. https://doi.org/10.1016/j.critrevonc.2017.09.017.
Hindie E. Metastatic melanoma: can FDG-PET predict success of anti-PD-1 therapy and help determine when it can be discontinued? Eur J Nucl Med Mol Imaging. 2020. https://doi.org/10.1007/s00259-020-04826-7.
Seban RD, Moya-Plana A, Antonios L, Yeh R, Marabelle A, Deutsch E, et al. Prognostic 18F-FDG PET biomarkers in metastatic mucosal and cutaneous melanoma treated with immune checkpoint inhibitors targeting PD-1 and CTLA-4. Eur J Nucl Med Mol Imaging. 2020. https://doi.org/10.1007/s00259-020-04757-3.
Yentz S, Lao CD. Immunotherapy for mucosal melanoma. Ann Transl Med. 2019;7(Suppl 3):S118. https://doi.org/10.21037/atm.2019.05.62.
Michielin O, van Akkooi ACJ, Ascierto PA, Dummer R, Keilholz U. Clinicalguidelines@esmo.org EGCEa. Cutaneous melanoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-updagger. Ann Oncol. 2019;30(12):1884–901. https://doi.org/10.1093/annonc/mdz411.
Coit DG, Thompson JA, Albertini MR, Barker C, Carson WE, Contreras C, et al. Cutaneous melanoma, version 2.2019, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw. 2019;17(4):367–402. https://doi.org/10.6004/jnccn.2019.0018.
Hicks RJ, Iravani A, Sandhu S. (18)F-fluorodeoxyglucose positron emission tomography/computed tomography for assessing tumor response to immunotherapy in solid tumors: melanoma and beyond. PET Clin. 2020;15(1):11–22. https://doi.org/10.1016/j.cpet.2019.08.007.
Ito K, Schoder H, Teng R, Humm JL, Ni A, Wolchok JD, et al. Prognostic value of baseline metabolic tumor volume measured on (18)F-fluorodeoxyglucose positron emission tomography/computed tomography in melanoma patients treated with ipilimumab therapy. Eur J Nucl Med Mol Imaging. 2019;46(4):930–9. https://doi.org/10.1007/s00259-018-4211-0.
Amrane K, Le Goupil D, Quere G, Delcroix O, Gouva S, Schick U, et al. Prediction of response to immune checkpoint inhibitor therapy using 18F-FDG PET/CT in patients with melanoma. Medicine (Baltimore). 2019;98(29):e16417. https://doi.org/10.1097/MD.0000000000016417.
Anwar H, Sachpekidis C, Winkler J, Kopp-Schneider A, Haberkorn U, Hassel JC, et al. Absolute number of new lesions on (18)F-FDG PET/CT is more predictive of clinical response than SUV changes in metastatic melanoma patients receiving ipilimumab. Eur J Nucl Med Mol Imaging. 2018;45(3):376–83. https://doi.org/10.1007/s00259-017-3870-6.
Boursi B, Werner TJ, Gholami S, Margalit O, Baruch E, Markel G, et al. Physiologic colonic fluorine-18-fluorodeoxyglucose uptake may predict response to immunotherapy in patients with metastatic melanoma. Melanoma Res. 2019;29(3):318–21. https://doi.org/10.1097/cmr.0000000000000566.
Breki CM, Dimitrakopoulou-Strauss A, Hassel J, Theoharis T, Sachpekidis C, Pan L, et al. Fractal and multifractal analysis of PET/CT images of metastatic melanoma before and after treatment with ipilimumab. EJNMMI Res. 2016;6(1):61. https://doi.org/10.1186/s13550-016-0216-5.
Cho SY, Lipson EJ, Im HJ, Rowe SP, Gonzalez EM, Blackford A, et al. Prediction of response to immune checkpoint inhibitor therapy using early-time-point (18)F-FDG PET/CT imaging in patients with advanced melanoma. J Nucl Med. 2017;58(9):1421–8. https://doi.org/10.2967/jnumed.116.188839.
de Heer EC, Brouwers AH, Boellaard R, Sluiter WJ, Diercks GFH, Hospers GAP, et al. Mapping heterogeneity in glucose uptake in metastatic melanoma using quantitative 18F-FDG PET/CT analysis. EJNMMI Res. 2018;8(1):101. https://doi.org/10.1186/s13550-018-0453-x.
Iravani A, Osman MM, Weppler AM, Wallace R, Galligan A, Lasocki A, et al. FDG PET/CT for tumoral and systemic immune response monitoring of advanced melanoma during first-line combination ipilimumab and nivolumab treatment. Eur J Nucl Med Mol Imaging. 2020. https://doi.org/10.1007/s00259-020-04815-w.
Ito K, Teng R, Schoder H, Humm JL, Ni A, Michaud L, et al. (18)F-FDG PET/CT for monitoring of ipilimumab therapy in patients with metastatic melanoma. J Nucl Med. 2019;60(3):335–41. https://doi.org/10.2967/jnumed.118.213652.
Kong BY, Menzies AM, Saunders CA, Liniker E, Ramanujam S, Guminski A, et al. Residual FDG-PET metabolic activity in metastatic melanoma patients with prolonged response to anti-PD-1 therapy. Pigment Cell Melanoma Res. 2016;29(5):572–7. https://doi.org/10.1111/pcmr.12503.
Lang N, Dick J, Slynko A, Schulz C, Dimitrakopoulou-Strauss A, Sachpekidis C, et al. Clinical significance of signs of autoimmune colitis in (18)F-fluorodeoxyglucose positron emission tomography-computed tomography of 100 stage-IV melanoma patients. Immunotherapy. 2019;11(8):667–76. https://doi.org/10.2217/imt-2018-0146.
Nakamoto R, Zaba LC, Rosenberg J, Reddy SA, Nobashi TW, Davidzon G, et al. Prognostic value of volumetric PET parameters at early response evaluation in melanoma patients treated with immunotherapy. Eur J Nucl Med Mol Imaging. 2020. https://doi.org/10.1007/s00259-020-04792-0.
Nobashi T, Baratto L, Reddy SA, Srinivas S, Toriihara A, Hatami N, et al. Predicting response to immunotherapy by evaluating tumors, lymphoid cell-rich organs, and immune-related adverse events using FDG-PET/CT. Clin Nucl Med. 2019;44(4):e272–e9. https://doi.org/10.1097/RLU.0000000000002453.
Sachpekidis C, Larribere L, Pan L, Haberkorn U, Dimitrakopoulou-Strauss A, Hassel JC. Predictive value of early 18F-FDG PET/CT studies for treatment response evaluation to ipilimumab in metastatic melanoma: preliminary results of an ongoing study. Eur J Nucl Med Mol Imaging. 2015;42(3):386–96. https://doi.org/10.1007/s00259-014-2944-y.
Sachpekidis C, Anwar H, Winkler JK, Kopp-Schneider A, Larribere L, Haberkorn U, et al. Longitudinal studies of the (18)F-FDG kinetics after ipilimumab treatment in metastatic melanoma patients based on dynamic FDG PET/CT. Cancer Immunol Immunother. 2018;67(8):1261–70. https://doi.org/10.1007/s00262-018-2183-3.
Sachpekidis C, Anwar H, Winkler J, Kopp-Schneider A, Larribere L, Haberkorn U, et al. The role of interim (18)F-FDG PET/CT in prediction of response to ipilimumab treatment in metastatic melanoma. Eur J Nucl Med Mol Imaging. 2018;45(8):1289–96. https://doi.org/10.1007/s00259-018-3972-9.
Sachpekidis C, Kopp-Schneider A, Hakim-Meibodi L, Dimitrakopoulou-Strauss A, Hassel JC. 18F-FDG PET/CT longitudinal studies in patients with advanced metastatic melanoma for response evaluation of combination treatment with vemurafenib and ipilimumab. Melanoma Res. 2019;29(2):178–86. https://doi.org/10.1097/CMR.0000000000000541.
Sachpekidis C, Larribere L, Kopp-Schneider A, Hassel JC, Dimitrakopoulou-Strauss A. Can benign lymphoid tissue changes in (18)F-FDG PET/CT predict response to immunotherapy in metastatic melanoma? Cancer Immunol Immunother. 2019;68(2):297–303. https://doi.org/10.1007/s00262-018-2279-9.
Sanli Y, Leake J, Odu A, Xi Y, Subramaniam RM. Tumor heterogeneity on FDG PET/CT and immunotherapy: an imaging biomarker for predicting treatment response in patients with metastatic melanoma. AJR Am J Roentgenol. 2019:1–9. https://doi.org/10.2214/AJR.18.19796.
Seban RD, Nemer JS, Marabelle A, Yeh R, Deutsch E, Ammari S, et al. Prognostic and theranostic 18F-FDG PET biomarkers for anti-PD1 immunotherapy in metastatic melanoma: association with outcome and transcriptomics. Eur J Nucl Med Mol Imaging. 2019;46(11):2298–310. https://doi.org/10.1007/s00259-019-04411-7.
Seith F, Forschner A, Schmidt H, Pfannenberg C, Guckel B, Nikolaou K, et al. 18F-FDG-PET detects complete response to PD1-therapy in melanoma patients two weeks after therapy start. Eur J Nucl Med Mol Imaging. 2018;45(1):95–101. https://doi.org/10.1007/s00259-017-3813-2.
Tan AC, Emmett L, Lo S, Liu V, Kapoor R, Carlino MS, et al. FDG-PET response and outcome from anti-PD-1 therapy in metastatic melanoma. Ann Oncol. 2018;29(10):2115–20. https://doi.org/10.1093/annonc/mdy330.
Wong ANM, McArthur GA, Hofman MS, Hicks RJ. The advantages and challenges of using FDG PET/CT for response assessment in melanoma in the era of targeted agents and immunotherapy. Eur J Nucl Med Mol Imaging. 2017;44(Suppl 1):67–77. https://doi.org/10.1007/s00259-017-3691-7.
Zamora J, Abraira V, Muriel A, Khan K, Coomarasamy A. Meta-DiSc: a software for meta-analysis of test accuracy data. BMC Med Res Methodol. 2006;6:31. https://doi.org/10.1186/1471-2288-6-31.
Wong A, Callahan J, Keyaerts M, Neyns B, Mangana J, Aberle S, et al. (18)F-FDG PET/CT based spleen to liver ratio associates with clinical outcome to ipilimumab in patients with metastatic melanoma. Cancer Imaging. 2020;20(1):36. https://doi.org/10.1186/s40644-020-00313-2.
Pennock GK, Waterfield W, Wolchok JD. Patient responses to ipilimumab, a novel immunopotentiator for metastatic melanoma: how different are these from conventional treatment responses? Am J Clin Oncol. 2012;35(6):606–11. https://doi.org/10.1097/COC.0b013e318209cda9.
Dimitrakopoulou-Strauss A. Monitoring of patients with metastatic melanoma treated with immune checkpoint inhibitors using PET-CT. Cancer Immunol Immunother. 2019;68(5):813–22. https://doi.org/10.1007/s00262-018-2229-6.
Prigent K, Aide N. (18)F-fludeoxyglucose PET/computed tomography for assessing tumor response to immunotherapy and detecting immune-related side effects: a checklist for the PET reader. PET Clin. 2020;15(1):1–10. https://doi.org/10.1016/j.cpet.2019.08.006.
Wachsmann JW, Ganti R, Peng F. Immune-mediated disease in ipilimumab immunotherapy of melanoma with FDG PET-CT. Acad Radiol. 2017;24(1):111–5. https://doi.org/10.1016/j.acra.2016.08.005.
Aide N, Hicks RJ, Le Tourneau C, Lheureux S, Fanti S, Lopci E. FDG PET/CT for assessing tumour response to immunotherapy : report on the EANM symposium on immune modulation and recent review of the literature. Eur J Nucl Med Mol Imaging. 2019;46(1):238–50. https://doi.org/10.1007/s00259-018-4171-4.
Teng F, Meng X, Kong L, Yu J. Progress and challenges of predictive biomarkers of anti PD-1/PD-L1 immunotherapy: a systematic review. Cancer Lett. 2018;414:166–73. https://doi.org/10.1016/j.canlet.2017.11.014.
Author information
Authors and Affiliations
Contributions
Conceptualization: All authors.
Methodology: Z. Kiamanesh, R. Sadeghi, N. Ayati
Formal analysis: R. Sadeghi, Z. Kiamanesh, N. Ayati
Writing—Original draft preparation: N. Ayati, Z. Kiamanesh
Writing—review and editing: R. Sadeghi, S.R. Zakavi, S.T. Lee, A. M. Scott
Supervision: R. Sadeghi, A. M. Scott
All authors have read and approved the submitted manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology – General
Rights and permissions
About this article
Cite this article
Ayati, N., Sadeghi, R., Kiamanesh, Z. et al. The value of 18F-FDG PET/CT for predicting or monitoring immunotherapy response in patients with metastatic melanoma: a systematic review and meta-analysis. Eur J Nucl Med Mol Imaging 48, 428–448 (2021). https://doi.org/10.1007/s00259-020-04967-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-020-04967-9