Abstract
Purpose
An imaging-based stratification tool is needed to identify melanoma patients who will benefit from anti Programmed Death-1 antibody (anti-PD1). We aimed at identifying biomarkers for survival and response evaluated in lymphoid tissue metabolism in spleen and bone marrow before initiation of therapy.
Methods
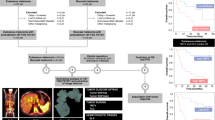
This retrospective study included 55 patients from two institutions who underwent 18F-FDG PET/CT before anti-PD1. Parameters extracted were SUVmax, SUVmean, HISUV (SUV-based Heterogeneity Index), TMTV (total metabolic tumor volume), TLG (total lesion glycolysis), BLR (Bone marrow-to-Liver SUVmax ratio), and SLR (Spleen-to-Liver SUVmax ratio). Each parameter was dichotomized using the median as a threshold. Association with survival, best overall response (BOR), and transcriptomic analyses (NanoString assay) were evaluated using Cox prediction models, Wilcoxon tests, and Spearman’s correlation, respectively.
Results
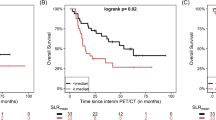
At 20.7 months median follow-up, 33 patients had responded, and 29 patients died. Median PFS and OS were 11.4 (95%CI 2.7–20.2) and 28.5 (95%CI 13.4–43.8) months. TMTV (>25cm3), SLR (>0.77), and BLR (>0.79) correlated with shorter survival. High TMTV (>25 cm3), SLR (>0.77), and BLR (>0.79) correlated with shorter survival, with TMTV (HR PFS 2.2, p = 0.02, and HR OS 2.5, p = 0.02) and BLR (HR OS 2.3, p = 0.04) remaining significant in a multivariable analysis. Low TMTV and TLG correlated with BOR (p = 0.03). Increased glucose metabolism in bone marrow (BLR) was associated with transcriptomic profiles including regulatory T cell markers (p < 0.05).
Conclusion
Low tumor burden correlates with survival and objective response while hematopoietic tissue metabolism correlates inversely with survival. These biomarkers should be further evaluated for potential clinical application.



Similar content being viewed by others
Abbreviations
- 18F-FDG:
-
18fluor-fluoro-deoxy-glucose
- AJCC:
-
American joint committee on cancer
- BLR:
-
Bone marrow-to-liver maximum standard uptake value ratio
- BM:
-
Bone marrow
- BOR:
-
Best overall response
- CD(4/8):
-
Cluster of differentiation
- CI:
-
Confidence interval
- CR:
-
Complete response
- CT:
-
Computed tomography
- FOXP3:
-
Forkhead box P3
- G-CSF:
-
Granulocyte-colony stimulating factor
- GM-CSF:
-
Granulocyte-macrophage colony stimulating factor.
- HISUV:
-
Heterogeneity index standard uptake value-based
- HR:
-
Hazard ratio
- ICI:
-
Immune checkpoint inhibitor
- IgG:
-
Immunoglobulin G
- IL3RA:
-
Interleukin 3 receptor subunit alpha
- iRECIST:
-
Immune response evaluation criteria in solid tumors
- LDH:
-
Lactate dehydrogenase
- LSO:
-
Lu2SiO5:Ce (lutetium, orthosilicate, cerium)
- LYSO:
-
Lu1.8Y.2SiO5:Ce (lutetium, yttrium, orthosilicate, cerium)
- MDSC:
-
Myeloid-derived suppressor cell
- MRI:
-
Magnetic resonance imaging
- OS:
-
Overall survival
- PET:
-
Positron emission tomography
- PD:
-
Progression disease
- PD-1:
-
Programmed cell death-1
- PERCIST:
-
Positron emission tomography evaluation response criteria in solid tumors
- PFS:
-
Progression-free survival
- PR:
-
Partial response
- RECIST 1.1:
-
Response evaluation criteria in solid tumors version 1.1
- RNA:
-
Ribonucleic acid
- SD:
-
Stable disease
- SLR:
-
Spleen-to-liver maximum standard uptake value ratio
- SUVmax:
-
Maximum standard uptake value
- SUVmean:
-
Mean standard uptake value
- TAM:
-
Tumor-associated macrophage
- TAN:
-
Tumor-associated neutrophil
- TLG:
-
Total lesion glycolysis
- TMTV:
-
Total metabolic tumor volume
- Tregs:
-
Regulatory T cells
- VOI:
-
Volume of interest
References
Robert C, Long GV, Brady B, Dutriaux C, Maio M, Mortier L, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320–30.
Robert C, Schachter J, Long GV, Arance A, Grob JJ, Mortier L, et al. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015;372:2521–32.
Ribas A, Hamid O, Daud A, Hodi FS, Wolchok JD, Kefford R, et al. Association of Pembrolizumab with tumor response and survival among patients with advanced melanoma. JAMA. 2016;315:1600–9.
Tan AC, Emmett L, Lo S, Liu V, Kapoor R, Carlino MS, et al. FDG-PET response and outcome from anti-PD-1 therapy in metastatic melanoma. Ann Oncol. 2018;29:2115–20.
Dercle L, Ammari S, Champiat S, Massard C, Ferté C, Taihi L, et al. Rapid and objective CT scan prognostic scoring identifies metastatic patients with long-term clinical benefit on anti-PD-1/−L1 therapy. Eur J Cancer. 2016;65:33–42.
Qin B-D, Jiao X-D, Zang Y-S. Tumor mutation burden to tumor burden ratio and prediction of clinical benefit of anti-PD-1/PD-L1 immunotherapy. Med Hypotheses. 2018;116:111–3.
Dercle L, Seban R-D, Lazarovici J, Schwartz LH, Houot R, Ammari S, et al. 18F-FDG PET and CT scans detect new imaging patterns of response and progression in patients with Hodgkin lymphoma treated by anti-programmed death 1 immune checkpoint inhibitor. J Nucl Med. 2018;59:15–24.
Dercle L, Ammari S, Seban R-D, Schwartz LH, Houot R, Labaied N, et al. Kinetics and nadir of responses to immune checkpoint blockade by anti-PD1 in patients with classical Hodgkin lymphoma. Eur J Cancer. 2018;91:136–44.
Joseph RW, Elassaiss-Schaap J, Kefford RF, Hwu W-J, Wolchok JD, Joshua AM, et al. Baseline tumor size is an independent prognostic factor for overall survival in patients with melanoma treated with Pembrolizumab. Clin Cancer Res. 2018;24(20):4960–7.
Robert C, Ribas A, Hamid O, Daud A, Wolchok JD, Joshua AM, et al. Durable complete response after discontinuation of Pembrolizumab in patients with metastatic melanoma. J Clin Oncol. 2018;36:1668–74.
Son SH, Kang SM, Jeong SY, Lee S-W, Lee S-J, Lee J, et al. Prognostic value of volumetric parameters measured by pretreatment 18F FDG PET/CT in patients with cutaneous malignant melanoma. Clin Nucl Med. 2016;41:e266–73.
Tomita M, Yasui H, Higashikawa K, Nakajima K, Takakura H, Shiga T, et al. Anti PD-1 treatment increases [18F]FDG uptake by cancer cells in a mouse B16F10 melanoma model. EJNMMI Res. 2018;8:82.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–74.
Angelova M, Mlecnik B, Vasaturo A, Bindea G, Fredriksen T, Lafontaine L, et al. Evolution of metastases in space and time under immune selection. Cell. 2018;175:751–765.e16.
Gerlinger M, Rowan AJ, Horswell S, Math M, Larkin J, Endesfelder D, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. 2012;366:883–92.
Salamon J, Derlin T, Bannas P, Busch JD, Herrmann J, Bockhorn M, et al. Evaluation of intratumoural heterogeneity on 18F-FDG PET/CT for characterization of peripheral nerve sheath tumours in neurofibromatosis type 1. Eur J Nucl Med Mol Imaging. 2013;40:685–92.
Dercle L, Hartl D, Rozenblum-Beddok L, Mokrane F-Z, Seban R-D, Yeh R, et al. Diagnostic and prognostic value of 18F-FDG PET, CT, and MRI in perineural spread of head and neck malignancies. Eur Radiol. 2018;28:1761–70.
Sachpekidis C, Larribère L, Kopp-Schneider A, Hassel JC, Dimitrakopoulou-Strauss A. Can benign lymphoid tissue changes in 18F-FDG PET/CT predict response to immunotherapy in metastatic melanoma? Cancer Immunol Immunother. 2019;68(2):297–303.
Zhao E, Xu H, Wang L, Kryczek I, Wu K, Hu Y, et al. Bone marrow and the control of immunity. Cell Mol Immunol. 2012;9:11–9.
Jordan KR, Kapoor P, Spongberg E, Tobin RP, Gao D, Borges VF, et al. Immunosuppressive myeloid-derived suppressor cells are increased in splenocytes from cancer patients. Cancer Immunol Immunother. 2017;66:503–13.
Zilio S, Serafini P. Neutrophils and granulocytic MDSC: the Janus god of cancer immunotherapy. Vaccines (Basel). 2016. https://doi.org/10.3390/vaccines4030031.
Cortez-Retamozo V, Etzrodt M, Newton A, Rauch PJ, Chudnovskiy A, Berger C, et al. Origins of tumor-associated macrophages and neutrophils. Proc Natl Acad Sci USA. 2012;109:2491–6.
Seban R-D, Robert C, Dercle L, Yeh R, Dunant A, Reuze S, et al. Increased bone marrow SUVmax on 18F-FDG PET is associated with higher pelvic treatment failure in patients with cervical cancer treated by chemoradiotherapy and brachytherapy. Oncoimmunology. 2019;8:e1574197.
Nam H-Y, Kim S-J, Kim I-J, Kim B-S, Pak K, Kim K. The clinical implication and prediction of diffuse splenic FDG uptake during cancer surveillance. Clin Nucl Med. 2010;35:759–63.
Sade-Feldman M, Yizhak K, Bjorgaard SL, Ray JP, de Boer CG, Jenkins RW, et al. Defining T cell states associated with response to checkpoint immunotherapy in melanoma. Cell. 2018;175:998–1013.e20.
Chung HW, Lee KY, Kim HJ, Kim WS, So Y. FDG PET/CT metabolic tumor volume and total lesion glycolysis predict prognosis in patients with advanced lung adenocarcinoma. J Cancer Res Clin Oncol. 2014;140:89–98.
Lee JW, Kang CM, Choi HJ, Lee WJ, Song SY, Lee J-H, et al. Prognostic value of metabolic tumor volume and total lesion glycolysis on preoperative 18F-FDG PET/CT in patients with pancreatic cancer. J Nucl Med. 2014;55:898–904.
Kang S, Ahn B-C, Hong CM, Song B-I, Lee HJ, Jeong SY, et al. Can (18)F-FDG PET/CT predict recurrence in patients with cutaneous malignant melanoma? Nuklearmedizin. 2011;50:116–21.
Wong ANM, Callahan J, Beresford J, Herschtal A, Fullerton S, Milne D, et al. Spleen to liver ratio (SLR): novel PET imaging biomarker for prediction of overall survival after ipilimumab and anti-PD1 in patients with metastatic melanoma. JCO. 2016;34:9523.
Sun R, Limkin EJ, Vakalopoulou M, Dercle L, Champiat S, Han SR, et al. A radiomics approach to assess tumour-infiltrating CD8 cells and response to anti-PD-1 or anti-PD-L1 immunotherapy: an imaging biomarker, retrospective multicohort study. Lancet Oncol. 2018;19:1180–91.
Kaplan RN, Riba RD, Zacharoulis S, Bramley AH, Vincent L, Costa C, et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature. 2005;438:820–7.
Hiratsuka S, Nakamura K, Iwai S, Murakami M, Itoh T, Kijima H, et al. MMP9 induction by vascular endothelial growth factor receptor-1 is involved in lung-specific metastasis. Cancer Cell. 2002;2:289–300.
Morrison SJ, Scadden DT. The bone marrow niche for haematopoietic stem cells. Nature. 2014;505:327–34.
Kamran N, Li Y, Sierra M, Alghamri MS, Kadiyala P, Appelman HD, et al. Melanoma induced immunosuppression is mediated by hematopoietic dysregulation. Oncoimmunology. 2018;7:e1408750.
Gabrilovich DI, Nagaraj S. Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol. 2009;9:162–74.
Bauckneht M, Piva R, Sambuceti G, Grossi F, Morbelli S. Evaluation of response to immune checkpoint inhibitors: is there a role for positron emission tomography? World J Radiol. 2017;9:27–33.
Munn LL. Cancer and inflammation. Wiley Interdiscip Rev Syst Biol Med. 2017;9(2):e1370.
Margolin K. Introduction to the role of the immune system in melanoma. Hematol Oncol Clin North Am. 2014;28:537–58.
Maru GB, Gandhi K, Ramchandani A, Kumar G. The role of inflammation in skin cancer. Adv Exp Med Biol. 2014;816:437–69.
Jiang H, Gebhardt C, Umansky L, Beckhove P, Schulze TJ, Utikal J, et al. Elevated chronic inflammatory factors and myeloid-derived suppressor cells indicate poor prognosis in advanced melanoma patients. Int J Cancer. 2015;136:2352–60.
Gebhardt C, Sevko A, Jiang H, Lichtenberger R, Reith M, Tarnanidis K, et al. Myeloid cells and related chronic inflammatory factors as novel predictive markers in melanoma treatment with Ipilimumab. Clin Cancer Res. 2015;21:5453–9.
Bastiaannet E, Hoekstra OS, de Jong JR, Brouwers AH, Suurmeijer AJH, Hoekstra HJ. Prognostic value of the standardized uptake value for (18)F-fluorodeoxyglucose in patients with stage IIIB melanoma. Eur J Nucl Med Mol Imaging. 2012;39:1592–8.
Lopci E, Toschi L, Grizzi F, Rahal D, Olivari L, Castino GF, et al. Correlation of metabolic information on FDG-PET with tissue expression of immune markers in patients with non-small cell lung cancer (NSCLC) who are candidates for upfront surgery. Eur J Nucl Med Mol Imaging. 2016;43:1954–61.
Boursi B, Werner TJ, Gholami S, Margalit O, Baruch E, Markel G, et al. Physiologic colonic fluorine-18-fluorodeoxyglucose uptake may predict response to immunotherapy in patients with metastatic melanoma. Melanoma Res. 2018.
Seymour L, Bogaerts J, Perrone A, Ford R, Schwartz LH, Mandrekar S, et al. iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol. 2017;18:e143–52.
Houdek Š, Büchler T, Kindlová E. Comparison of RECIST 1.1 and iRECIST for response evaluation in solid Tumours. Klin Onkol. 2017;30:32–9.
Shen M, Wang J, Yu W, Zhang C, Liu M, Wang K, et al. A novel MDSC-induced PD-1-PD-L1+ B-cell subset in breast tumor microenvironment possesses immuno-suppressive properties. Oncoimmunology. 2018;7:e1413520.
Liu Y, Wei G, Cheng WA, Dong Z, Sun H, Lee VY, et al. Targeting myeloid-derived suppressor cells for cancer immunotherapy. Cancer Immunol Immunother. 2018;67(8):1181–95.
Acknowledgements
Patients Recruitment (CM, JCS, LHS, YS, CR). Clinical Data Collection (SA, AMP, FZM, GR, LB, LD). Imaging Data Collection (RDS, JSN, RY, LD). Transcriptomics Data Cost (AM, YS). Transcriptomics Data Collection (AM, RG, AB, YS, LD). Data analysis (RDS, LD). Manuscript writing (RDS, LD). Manuscript editing (RDS, JSN, YS, LD). Manuscript final approval (all authors). L Dercle work is funded by a grant from Fondation Philanthropia, Geneva, Switzerland and the Fondation Nuovo-Soldati.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors have declared no competing interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology–General.
Electronic supplementary material
ESM 1
(DOCX 344 kb)
Rights and permissions
About this article
Cite this article
Seban, RD., Nemer, J.S., Marabelle, A. et al. Prognostic and theranostic 18F-FDG PET biomarkers for anti-PD1 immunotherapy in metastatic melanoma: association with outcome and transcriptomics. Eur J Nucl Med Mol Imaging 46, 2298–2310 (2019). https://doi.org/10.1007/s00259-019-04411-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-019-04411-7