Abstract
Purpose
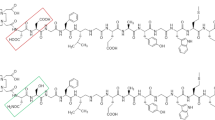
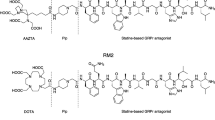
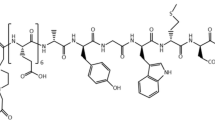
Diethylenetriamine-pentaacetic acid (DTPA)-coupled minigastrins are unsuitable for therapeutic application with the available β-emitting radiometals due to low complex stability. Low tumour-to-kidney ratio of the known radiopharmaceuticals is further limiting their potency. We used macrocyclic chelators for coupling to increase complex stability, modified the peptide sequence to enhance radiolytic stability and studied tumour-to-kidney ratio and metabolic stability using 111In-labelled derivatives.
Methods
Gastrin derivatives with decreasing numbers of glutamic acids were synthesised using 111In as surrogate for therapeutic radiometals for in vitro and in vivo studies. Gastrin receptor affinities of the natIn-metallated compounds were determined by receptor autoradiography using 125I-CCK as radioligand. Internalisation was evaluated in AR4-2J cells. Enzymatic stability was determined by incubating the 111In-labelled peptides in human serum. Biodistribution was performed in AR4-2J-bearing Lewis rats.
Results
IC50 values of the natIn-metallated gastrin derivatives vary between 1.2 and 4.8 nmol/L for all methionine-containing derivatives. Replacement of methionine by norleucine, isoleucine, methionine-sulfoxide and methionine-sulfone resulted in significant decrease of receptor affinity (IC50 between 9.9 and 1,195 nmol/L). All cholecystokinin receptor affinities were >100 nmol/L. All 111In-labelled radiopeptides showed receptor-specific internalisation. Serum mean-life times varied between 2.0 and 72.6 h, positively correlating with the number of Glu residues. All 111In-labelled macrocyclic chelator conjugates showed higher tumour-to-kidney ratios after 24 h (0.37–0.99) compared to 111In-DTPA-minigastrin 0 (0.05). Tumour wash out between 4 and 24 h was low. Imaging studies confirmed receptor-specific blocking of the tumour uptake.
Conclusions
Reducing the number of glutamates increased tumour-to-kidney ratio but resulted in lower metabolic stability. The properties of the macrocyclic chelator-bearing derivatives make them potentially suitable for clinical purposes.




Similar content being viewed by others
References
Wank SA, Pisegna JR, de Weerth A. Cholecystokinin receptor family. Molecular cloning, structure, and functional expression in rat, guinea pig, and human. Ann N Y Acad Sci 1994;713:49–66.
Rehfeld JF. The endoproteolytic maturation of progastrin and procholecystokinin. J Mol Med 2006;84:544–50.
Noble F, Wank SA, Crawley JN, Bradwejn J, Seroogy KB, Hamon M, et al. International Union of Pharmacology. XXI. Structure, distribution, and functions of cholecystokinin receptors. Pharmacol Rev 1999;51:745–81.
Reubi JC. Peptide receptors as molecular targets for cancer diagnosis and therapy. Endocr Rev 2003;24:389–427.
Reubi JC, Waser B, Laderach U, Stettler C, Friess H, Halter F, et al. Localization of cholecystokinin A and cholecystokinin B-gastrin receptors in the human stomach. Gastroenterology 1997;112:1197–205.
Reubi J, Waser B. Unexpected high incidence of cholecystokinin B/gastrin receptors in human medullary thyroid carcinomas. Int J Cancer 1996;67:644–7.
Reubi JC, Schaer JC, Waser B. Cholecystokinin(CCK)-A and CCK-B/gastrin receptors in human tumors. Cancer Res 1997;57:1377–86.
Reubi JC, Waser B. Concomitant expression of several peptide receptors in neuroendocrine tumours: molecular basis for in vivo multireceptor tumour targeting. Eur J Nucl Med Mol Imaging 2003;30:781–93.
Gotthardt M, Behe MP, Beuter D, Battmann A, Bauhofer A, Schurrat T, et al. Improved tumour detection by gastrin receptor scintigraphy in patients with metastasised medullary thyroid carcinoma. Eur J Nucl Med Mol Imaging 2006;33:1273–9.
Gotthardt M, Behe MP, Grass J, Bauhofer A, Rinke A, Schipper ML, et al. Added value of gastrin receptor scintigraphy in comparison to somatostatin receptor scintigraphy in patients with carcinoids and other neuroendocrine tumours. Endocr Relat Cancer 2006;13:1203–11.
Behr TM, Jenner N, Radetzky S, Behe M, Gratz S, Yucekent S, et al. Targeting of cholecystokinin-B/gastrin receptors in vivo: preclinical and initial clinical evaluation of the diagnostic and therapeutic potential of radiolabelled gastrin. Eur J Nucl Med 1998;25:424–30.
de Jong M, Bakker W, Bernard B, Valkema R, Kwekkeboom D, Reubi J, et al. Preclinical and initial clinical evaluation of 111In-labeled nonsulfated CCK8 analog: a peptide for CCK-B receptor targeted scintigraphy and radionuclide therapy. J Nucl Med 1999;40:2081–7.
Behr TM, Jenner N, Behe M, Angerstein C, Gratz S, Raue F, et al. Radiolabeled peptides for targeting cholecystokinin-B/gastrin receptor-expressing tumors. J Nucl Med 1999;40:1029–44.
Reubi JC. CCK receptors in human neuroendocrine tumors: clinical implications. Scand J Clin Lab Invest Suppl 2001;234:101–4.
Kwekkeboom DJ, Bakker WH, Kooij PP, Erion J, Srinivasan A, de Jong M, et al. Cholecystokinin receptor imaging using an octapeptide DTPA-CCK analogue in patients with medullary thyroid carcinoma. Eur J Nucl Med 2000;27:1312–7.
Reubi JC, Maecke HR, Krenning EP. Candidates for peptide receptor radiotherapy today and in the future. J Nucl Med 2005;46:67S–75S.
von Guggenberg E, Behe M, Behr TM, Saurer M, Seppi T, Decristoforo C. 99mTc-labeling and in vitro and in vivo evaluation of HYNIC- and (Na-His)acetic acid-modified [d-Glu1]-minigastrin. Bioconjug Chem 2004;15:864–71.
Behe M, Becker W, Gotthardt M, Angerstein C, Behr TM. Improved kinetic stability of DTPA-dGlu as compared with conventional monofunctional DTPA in chelating indium and yttrium: preclinical and initial clinical evaluation of radiometal labelled minigastrin derivatives. Eur J Nucl Med Mol Imaging 2003;30:1140–6.
Behe M, Behr TM. Cholecystokinin-B (CCK-B)/gastrin receptor targeting peptides for staging and therapy of medullary thyroid cancer and other CCK-B receptor expressing malignancies. Biopolymers 2002;66:399–418.
Nock BA, Maina T, Behe M, Nikolopoulou A, Gotthardt M, Schmitt JS, et al. CCK-2/Gastrin receptor-targeted tumor imaging with 99mTc-labeled minigastrin analogs. J Nucl Med 2005;46:1727–36.
Aloj L, Caraco C, Panico M, Zannetti A, Del Vecchio S, Tesauro D, et al. In vitro and in vivo evaluation of 111In-DTPAGlu-G-CCK8 for cholecystokinin-B receptor imaging. J Nucl Med 2004;45:485–94.
Behr TM, Behe MP. Cholecystokinin-B/Gastrin receptor-targeting peptides for staging and therapy of medullary thyroid cancer and other cholecystokinin-B receptor-expressing malignancies. Semin Nucl Med 2002;32:97–109.
Harrison A, Walker C, Parker D. The in vivo release of 90Y from cyclic and acyclic ligand-antibody conjugates. Nucl Med & Biol 1991;18:469–76.
Eisenwiener KP, Powell P, Maecke HR. A convenient synthesis of novel bifunctional prochelators for coupling to bioactive peptides for radiometal labelling. Bioorg Med Chem Lett 2000;10:2133–5.
Good S, Maecke H. Kinetics of formation and dissociation of two DOTA-based chelator conjugates with 177Lu. Eur J Nucl Med Mol Imaging 2003;30(Suppl 2):S319.
Good S, Maecke H. Stability and kinetics of formation of two macrocyclic chelator-conjugates. Nuklearmedizin 2004;43:A11.
Heppeler A, Froidevaux S, Mäcke HR, Jermann E, Béhé M, Powell P, et al. Radiometal-labelled macrocyclic chelator-derivatised somatostatin analogue with superb tumour-targeting properties and potential for receptor-mediated internal radiotherapy. Chemistry A European Journal 1999;5:1016–23.
Eisenwiener KP, Prata MI, Buschmann I, Zhang HW, Santos AC, Wenger S, et al. NODAGATOC, a new chelator-coupled somatostatin analogue labeled with [67/68Ga] and [111In] for SPECT, PET, and targeted therapeutic applications of somatostatin receptor (hsst2) expressing tumors. Bioconjug Chem 2002;13:530–41.
Reubi J, Waser B, Schaer J, Laederach U, Erion J, Srinivasan A, et al. Unsulfated DTPA- and DOTA-CCK analogs as specific high-affinity ligands for CCK-B receptor-expressing human and rat tissues in vitro and in vivo. Eur J Nucl Med 1998;25:481–90.
Reubi JC, Kvols LK, Waser B, Nagorney DM, Heitz PU, Charboneau JW, et al. Detection of somatostatin receptors in surgical and percutaneous needle biopsy samples of carcinoids and islet cell carcinomas. Cancer Res 1990;50:5969–77.
Christophe J. Pancreatic tumoral cell line AR42J: an amphicrine model. Am J Physiol 1994;266:G963–G71.
Wild D, Schmitt JS, Ginj M, Maecke HR, Bernard BF, Krenning E, et al. DOTA-NOC, a high-affinity ligand of somatostatin receptor subtypes 2, 3 and 5 for labelling with various radiometals. Eur J Nucl Med Mol Imaging 2003;30:1338–47.
Storch D, Behe M, Walter MA, Chen J, Powell P, Mikolajczak R, et al. Evaluation of [99mTc/EDDA/HYNIC0]octreotide derivatives compared with [111In-DOTA0,Tyr3, Thr8]octreotide and [111In-DTPA0]octreotide: does tumor or pancreas uptake correlate with the rate of internalization? J Nucl Med 2005;46:1561–9.
Maecke H, Good S. Radiometals (non-Tc, non-Re) and bifunctional labeling chemistry. In: Vertes A, Nagy S, Klencsar Z, editors. Handbook of nuclear chemistry. Vol. 4. Netherlands; 2003, p. 279–314.
Bogni A, Bombardieri E, Iwata R, Cadini L, Pascali C. Stability of l-[S-methyl-11C]methionine solutions. J Radioanal Nucl Chem 2003;256:199–203.
Liu S, Edwards DS. Stabilization of 90Y-labeled DOTA-biomolecule conjugates using gentisic acid and ascorbic acid. Bioconjug Chem 2001;12:554–8.
Good S, Maecke H, Merlo A, Reubi JC. Development of NK-1 receptor mediated radiopharmaceuticals. Nuklearmedizin 2004;43:A7.
Breeman WA, de Jong M, Kwekkeboom DJ, Valkema R, Bakker WH, Kooij PP, et al. Somatostatin receptor-mediated imaging and therapy: basic science, current knowledge, limitations and future perspectives. Eur J Nucl Med 2001;28:1421–9.
Trejtnar F, Laznicek M. Analysis of renal handling of radiopharmaceuticals. Q J Nucl Med 2002;46:181–94.
de Jong M, Barone R, Krenning E, Bernard B, Melis M, Visser T, et al. Megalin is essential for renal proximal tubule reabsorption of 111In-DTPA-octreotide. J Nucl Med 2005;46:1696–700.
Behe M, Kluge G, Becker W, Gotthardt M, Behr TM. Use of polyglutamic acids to reduce uptake of radiometal-labeled minigastrin in the kidneys. J Nucl Med 2005;46:1012–5.
von Guggenberg E, Rupprich M, Virgolini I, Decristoforo C. 99mTc-HYNIC-Minigastrin: improved in vitro and in vivo properties of a short chain analogue. Eur J Nucl Med Mol Imaging 2005;32(Suppl 1):S35.
Behe M, Reubi JC, Nock B. Evaluation of a DOTA-minigastrin derivative for therapy and diagnosis for CCK-2 receptor positive tumours. Eur J Nucl Med Mol Imaging 2005;32(Suppl 1):S78.
Behr TM, Goldenberg DM, Becker W. Reducing the renal uptake of radiolabeled antibody fragments and peptides for diagnosis and therapy: present status, future prospects and limitations. Eur J Nucl Med 1998;25:201–12.
Acknowledgements
Financial support from the Swiss National Science Foundation (Grant No. 320000-114043) is gratefully acknowledged. The work was performed within the European Cooperation in the field of Scientific and Technical Research, COST Action B12: “Radiotracers for in vivo assessment of biological functions” and the European Molecular Imaging Laboratories network of excellence (EMIL).
Author information
Authors and Affiliations
Corresponding author
Additional information
SG and MAW contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Table 1:
Complete list of biodistribution organ uptake (DOC 52.0 KB)
Rights and permissions
About this article
Cite this article
Good, S., Walter, M.A., Waser, B. et al. Macrocyclic chelator-coupled gastrin-based radiopharmaceuticals for targeting of gastrin receptor-expressing tumours. Eur J Nucl Med Mol Imaging 35, 1868–1877 (2008). https://doi.org/10.1007/s00259-008-0803-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-008-0803-4