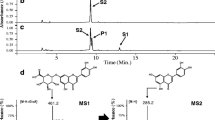
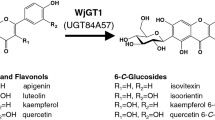
Abstract
Two plant-originated C-glucosyltransferases (CGTs) UGT708D1 from Glycine max and GtUF6CGT1 from Gentiana triflora were accessed for glucosylation of selected flavones chrysin and luteolin. Uridine diphosphate (UDP)-glucose pool was enhanced in Escherichia coli cell cytosol by introducing heterologous UDP-glucose biosynthetic genes, i.e., glucokinase (glk), phosphoglucomutase (pgm2), and glucose 1-phosphate uridylyltransferase (galU), along with glucose facilitator diffusion protein from (glf) from different organisms, in a multi-monocistronic vector with individual T7 promoter, ribosome binding site, and terminator for each gene. The C-glucosylated products were analyzed by high-performance liquid chromatography-photodiode array, high-resolution quadruple time-of-flight electrospray ionization mass spectrometry, and one-dimensional nuclear magnetic resonance analyses. Fed-batch shake flask culture showed 8% (7 mg/L; 16 μM) and 11% (9 mg/L; 22 μM) conversion of chrysin to chrysin 6-C-β-D-glucoside with UGT708D1 and GtUF6CGT1, respectively. Moreover, the bioengineered E. coli strains with exogenous UDP-glucose biosynthetic genes and glucose facilitator diffusion protein enhanced the production of chrysin 6-C-β-D-glucoside by approximately 1.4-fold, thus producing 10 mg/L (12%, 24 μM) and 14 mg/L (17%, 34 μM) by UGT708D1 and GtUF6CGT1, respectively, without supplementation of additional UDP-glucose in the medium. The biotransformation was further elevated when the bioengineered strain was scaled up in lab-scale fermentor at 3 L volume. HPLC analysis of fermentation broth extract revealed 50% (42 mg/L, 100 μM) conversion of chrysin to chrysin 6-C-β-D-glucoside at 48 h upon supplementation of 200 μM of chrysin. The maximum conversion of luteolin was 38% (34 mg/L, 76 μM) in 50-mL shake flask fermentation at 48 h. C-glucosylated derivative of chrysin was found to be more soluble and more stable to high temperature, different pH range, and β-glucosidase enzyme, than O-glucosylated derivative of chrysin.








Similar content being viewed by others
References
Abad-García B, Garmón-Lobato S, Berrueta LA, Gallo B, Vicente F (2008) New features on the fragmentation and differentiation of C-glycosidic flavone isomers by positive electrospray ionization and triple quadrupole mass spectrometry. Rapid Commun Mass Spectrom 22(12):1834–1842. https://doi.org/10.1002/rcm.3560
Baliga MS, Baliga BR, Kandathil SM, Bhat HP, Vayalil PK (2011) A review of the chemistry and pharmacology of the date fruits (Phoenix dactylifera L.) Food Res Int 44(3):1812–1822. https://doi.org/10.21010/ajtcam.v14i3.26
Bililign T, Hyun CG, Williams JS, Czisny AM, Thorson JS (2004) The hedamycin locus implicates a novel aromatic PKS priming mechanism. Chem Biol 11(7):959–969. https://doi.org/10.1016/j.chembiol.2004.04.016
Bowles D, Isayenkova J, Lim EK, Poppenberger B (2005) Glycosyltransferases: managers of small molecules. Curr Opin Plant Biol 8(3):254–263. https://doi.org/10.1016/j.pbi.2005.03.007
Brazier-Hicks M, Evans KM, Gershater MC, Puschmann H, Steel PG, Edwards R (2009) The C-glycosylation of flavonoids in cereals. J Biol Chem 284(27):17926–17934. https://doi.org/10.1074/jbc.M109.009258
Bungaruang L, Gutmann A, Nidetzky B (2013) Leloir glycosyltransferases and natural product glycosylation: biocatalytic synthesis of the C-glucoside nothofagin, a major antioxidant of redbush herbal tea. Adv Synth Catal 355(7):2757–2763. https://doi.org/10.1002/biot.201600557
Cartwright AM, Lim EK, Kleanthous C, Bowles DJ (2008) A kinetic analysis of regiospecific glucosylation by two glycosyltransferases of Arabidopsis thaliana domain swapping to introduce new activities. J Biol Chem 283(23):15724–15731. https://doi.org/10.1074/jbc.M801983200
Chaudhary AK, Park JW, Yoon YJ, Kim BG, Sohng JK (2013) Re-engineering of genetic circuit for 2-deoxystreptamine (2-DOS) biosynthesis in Escherichia coli BL21 (DE3). Biotechnol Lett 35(2):285–293. https://doi.org/10.1007/s10529-012-1077-2
Cheynier V, Comte G, Davies KM, Lattanzio V, Martens S (2013) Plant phenolics: recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol Biochem 72:1–20. https://doi.org/10.1016/j.plaphy.2013.05.009
Courts FL, Williamson G (2015) The occurrence, fate and biological activities of C-glycosyl flavonoids in the human diet. Crit Rev Food Sci Nutr 55(10):1352–1367. https://doi.org/10.1080/10408398.2012.694497
Day AJ, Gee JM, DuPont MS, Johnson IT, Williamson G (2003) Absorption of quercetin-3-glucoside and quercetin-4-glucoside in the rat small intestine: the role of lactase phlorizin hydrolase and the sodium-dependent glucose transporter. Biochem Pharmacol 65(7):1199–1206. https://doi.org/10.1016/S0006-2952(03)00039-X
De Bruyn F, Maertens J, Beauprez J, Soetaert W, De Mey M (2015) Biotechnological advances in UDP-sugar based glycosylation of small molecules. Biotechnol Adv 33(2):288–302. https://doi.org/10.1016/j.biotechadv.2015.02.005
de Villiers A, Venter P, Pasch H (2016) Recent advances and trends in the liquid-chromatography–mass spectrometry analysis of flavonoids. J Chromatogr A 1430:16–78. https://doi.org/10.1016/j.chroma.2015.11.077
Delazar A, Gibbons S, Kosari AR, Nazemiyeh H, Modarresi M, Nahar L, Sarker SD (2006) Flavone C-glycosides and cucurbitacin glycosides from Citrullus colocynthis. Daru 14:109–114
Dennis JW, Granovsky M, Warren CE (1999) Protein glycosylation in development and disease. BioEssays 21(5):412–421. https://doi.org/10.1002/(SICI)1521-1878(199905)21:5<412::AID-BIES8>3.0.CO;2-5
Durand GE, Seta N (2000) Protein glycosylation and diseases: blood and urinary oligosaccharides as markers for diagnosis and therapeutic monitoring. Clin Chem 46(6 Pt 1):795–805
Ferreyra ML, Rodriguez E, Casas MI, Labadie G, Grotewold E, Casati P (2013) Identification of a bifunctional maize C- and O-glucosyltransferase. J Biol Chem 288(44):31678–31688. https://doi.org/10.1074/jbc.M113.510040
Fischbach MA, Lin H, Liu DR, Walsh CT (2005) In vitro characterization of IroB, a pathogen-associated C-glycosyltransferase. Proc Natl Acad Sci U S A 102(3):571–576. https://doi.org/10.1073/pnas.0408463102
Gantt RW, Peltier-Pain P, Thorson JS (2011) Enzymatic methods for glycol (diversification/randomization) of drugs and small molecules. Nat Prod Rep 28(11):1811–1853. https://doi.org/10.1039/C1NP00045D
Gurung RB, Kim EH, Oh TJ, Sohng JK (2013) Enzymatic synthesis of apigenin glucosides by glucosyltransferase (YjiC) from Bacillus licheniformis DSM 13. Mol Cells 36(4):355–361. https://doi.org/10.1007/s10059-013-0164-0
Gutmann A, Lepak A, Diricks M, Desmet T, Nidetzky B (2017) Glycosyltransferase cascades for natural product glycosylation: use of plant instead of bacterial sucrose synthases improves the UDP-glucose recycling from sucrose and UDP. Biotechnol J 12(7):1600557. https://doi.org/10.1002/biot.201600557
Han SH, Kim BG, Yoon JA, Chong Y, Ahn JH (2014) Synthesis of flavonoid O-pentosides by Escherichia coli through engineering of nucleotide sugar pathways and glycosyltransferase. Appl Environ Microbiol 80(9):2754–2762. https://doi.org/10.1128/AEM.03797-13
Hao B, Caulfield JC, Hamilton ML, Pickett JA, Midega CA, Khan ZR, Wang J, Hooper AM (2016) Biosynthesis of natural and novel C-glycosylflavones utilising recombinant Oryza sativa C-glycosyltransferase (OsCGT) and Desmodium incanum root proteins. Phytochemistry 125:73–87. https://doi.org/10.1016/j.phytochem.2016.02.013
Hirade Y, Kotoku N, Terasaka K, Saijo-Hamano Y, Fukumoto A, Mizukami H (2015) Identification and functional analysis of 2-hydroxyflavanone C-glucosyltransferase in soybean (Glycine max). FEBS Lett 589(15):1778–1786. https://doi.org/10.1016/j.febslet.2015.05.010
Hooper AM, Tsanuo MK, Chamberlain K, Tittcomb K, Scholes J, Hassanali A, Khan ZR, Pickett JA (2010) Isoschaftoside, a C-glycosylflavonoid from Desmodium uncinatum root exudate, is an allelochemical against the development of Striga. Phytochemistry 71(8-9):904–908. https://doi.org/10.1016/j.phytochem.2010.02.015
Hultin PG (2005) Bioactive C-glycosides from bacterial secondary metabolism. Curr Top Med Chem 5(14):1299–1331. https://doi.org/10.2174/156802605774643015
Ito T, Fujimoto S, Suito F, Shimosaka M, Taguchi G (2017) C-glycosyltransferases catalyzing the formation of di-C-glucosyl flavonoids in citrus plants. Plant J 91(2):187–198. https://doi.org/10.1111/tpj.13555
Jay M (1994) C-glycosylflavonoids. In the flavonoids: advances in research since 1986. Chapman & Hall, London
Jeong KM, Yang M, Jin Y, Kim EM, Ko J, Lee J (2017) Identification of major flavone C-glycosides and their optimized extraction from Cymbidium kanran using deep eutectic solvents. Molecules 22(11):2006. https://doi.org/10.3390/molecules22112006
Kim DH, Kim BG, Lee HJ, Lim Y, Hur HG, Ahn JH (2005a) Enhancement of isoflavone synthase activity by co-expression of P450 reductase from rice. Biotechnol Lett 27(17):1291–1294. https://doi.org/10.1007/s10529-005-0221-7
Kim YC, Jun M, Jeong WS, Chung SK (2005b) Antioxidant properties of flavone C-glycosides from Atractylodes japonica leaves in human low-density lipoprotein oxidation. J Food Sci 70(9):S575–S580. https://doi.org/10.1111/j.1365-2621.2005.tb08331.x
Kim BG, Sung SH, Ahn JH (2012) Biological synthesis of quercetin 3-O-N-acetylglucosamine conjugate using engineered Escherichia coli expressing UGT78D2. Appl Microbiol Biotechnol 93(6):2447–2453. https://doi.org/10.1007/s00253-011-3747-8
Kitta K, Hagiwara Y, Shibamoto T (1992) Antioxidative activity of an isoflavonoid, 2″-O-glycosylisovitexin isolated from green barley leaves. J Agric Food Chem 40(10):1843–1845. https://doi.org/10.1021/jf00022a023
Koirala N, Pandey RP, Parajuli P, Jung HJ, Sohng JK (2014) Methylation and subsequent glycosylation of 7, 8-dihydroxyflavone. J Biotechnol 184:128–137. https://doi.org/10.1016/j.jbiotec.2014.05.005
Lairson LL, Henrissat B, Davies GJ, Withers SG (2008) Glycosyltransferases: structures, functions, and mechanisms. Ann Rev Biochem 77(1):521–555. https://doi.org/10.1146/annurev.biochem.76.061005.092322
Lee EM, Lee SS, Chung BY, Cho JY, Lee IC, Ahn SR, Jang SJ, Kim TH (2010) Pancreatic lipase inhibition by C-glycosidic flavones isolated from Eremochloa ophiuroides. Molecules 15(11):8251–8259. https://doi.org/10.3390/molecules15118251
Levy DE, Tang C (1995) The chemistry of C-glycosides. Pergamon, Oxford
Li QM, Van den Heuvel H, Dillen L, Claeys M (1992) Differentiation of 6-C-and 8-C-glycosidic flavonoids by positive ion fast atom bombardment and tandem mass spectrometry. Biol Mass Spectrom 21(4):213–221. https://doi.org/10.1002/bms.1200210406
Lin H, Fischbach MA, Liu DR, Walsh CT (2005) In vitro characterization of salmochelin and enterobactin trilactone hydrolases IroD, IroE, and Fes. J Am Chem Soc 127(31):11075–11084. https://doi.org/10.1021/ja0522027
Malla S, Pandey RP, Kim BG, Sohng JK (2013) Regiospecific modifications of naringenin for astragalin production in Escherichia coli. Biotechnol Bioeng 110(9):2525–2535. https://doi.org/10.1002/bit.24919
March RE, Lewars EG, Stadey CJ, Miao XS, Zhao X, Metcalfe CD (2006) A comparison of flavonoid glycosides by electrospray tandem mass spectrometry. Int J Mass Spectrom 248(1-2):61–85. https://doi.org/10.1016/j.ijms.2005.09.011
Markham KR, Mitchell KA (2003) The mis-identification of the major antioxidant flavonoids in young barley (Hordeum vulgare) leaves. Z Naturforsch C 58(1-2):53–56. https://doi.org/10.1515/znc-2003-1-209
McNally DJ, Wurms KV, Labbé C, Bélanger RR (2003) Synthesis of C-glycosyl flavonoid phytoalexins as a site-specific response to fungal penetration in cucumber. Physiol Mol Plant Pathol 63(6):293–303. https://doi.org/10.1016/j.pmpp.2004.03.005
Nagatomo Y, Usui S, Ito T, Kato A, Shimosaka M, Taguchi G (2014) Purification, molecular cloning and functional characterization of flavonoid C-glucosyltransferases from Fagopyrum esculentum M.(buckwheat) cotyledon. Plant J 80(3):437–448. https://doi.org/10.1111/tpj.12645
Pandey RP, Malla S, Simkhada D, Kim BG, Sohng JK (2013) Production of 3-O-xylosyl quercetin in Escherichia coli. Appl Microbiol Biotechnol 97(5):1889–1901. https://doi.org/10.1007/s00253-012-4438-9
Pandey RP, Gurung RB, Parajuli P, Koirala N, Sohng JK (2014) Assessing acceptor substrate promiscuity of YjiC-mediated glycosylation toward flavonoids. Carbohydr Res 393:26–31. https://doi.org/10.1016/j.carres.2014.03.011
Pandey RP, Parajuli P, Koffas MA, Sohng JK (2016) Microbial production of natural and non-natural flavonoids: pathway engineering, directed evolution and systems/synthetic biology. Biotechnol Adv 34(5):634–662. https://doi.org/10.1016/j.biotechadv.2016.02.012
Pandey RP, Parajuli P, Pokhrel AR, Sohng JK (2017) Biosynthesis of novel 7, 8-dihydroxyflavone glycoside derivatives and in silico study of their effects on BACE1 inhibition. Biotechnol Appl Biochem. https://doi.org/10.1002/bab.1570
Parajuli P, Pandey RP, Trang NT, Chaudhary AK, Sohng JK (2015) Synthetic sugar cassettes for the efficient production of flavonol glycosides in Escherichia coli. Microb Cell Factories 14(1):76. https://doi.org/10.1186/s12934-015-0261-1
Parajuli P, Pandey RP, Nguyen TT, Shrestha B, Yamaguchi T, Sohng JK (2017) Biosynthesis of natural and non-natural genistein glycosides. RSC Adv 7(26):16217–16231. https://doi.org/10.1039/C6RA28145A
Peng J, Fan G, Hong Z, Chai Y, Wu Y (2005) Preparative separation of isovitexin and isoorientin from Patrinia villosa Juss by high-speed counter-current chromatography. J Chromatogr A 1074(1-2):111–115. https://doi.org/10.1016/j.chroma.2005.03.067
Plaza M, Pozzo T, Liu J, Gulshan Ara KZ, Turner C, Nordberg Karlsson E (2014) Substituent effects on in vitro antioxidizing properties, stability, and solubility in flavonoids. J Agric Food Chem 62(15):3321–3333. https://doi.org/10.1021/jf405570u
Prasain JK, Jones K, Kirk M, Wilson L, Smith-Johnson M, Weaver C, Barnes S (2003) Profiling and quantification of isoflavonoids in kudzu dietary supplements by high-performance liquid chromatography and electrospray ionization tandem mass spectrometry. J Agric Food Chem 51(15):4213–4218. https://doi.org/10.1021/jf030174a
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor, New York
Sasaki N, Nishizaki Y, Yamada E, Tatsuzawa F, Nakatsuka T, Takahashi H, Nishihara M (2015) Identification of the glucosyltransferase that mediates direct flavone C-glucosylation in Gentiana triflora. FEBS Lett 589(1):182–187. https://doi.org/10.1016/j.febslet.2014.11.045
Schnaitman CA, Klena JD (1993) Genetics of lipopolysaccharide biosynthesis in enteric bacteria. Microbiol Rev 57:655–682
Simkhada D, Lee HC, Sohng JK (2010) Genetic engineering approach for the production of rhamnosyl and allosyl flavonoids from Escherichia coli. Biotechnol Bioeng 107(1):154–162. https://doi.org/10.1002/bit.22782
Snijman PW, Joubert E, Ferreira D, Li XC, Ding Y, Green IR, Gelderblom WC (2009) Antioxidant activity of the dihydrochalcones aspalathin and nothofagin and their corresponding flavones in relation to other rooibos (Aspalathus linearis) flavonoids, epigallocatechin gallate, and Trolox. J Agric Food Chem 57(15):6678–6684. https://doi.org/10.1021/jf901417k
Thuan NH, Sohng JK (2013) Recent biotechnological progress in enzymatic synthesis of glycosides. J Ind Microbiol Biotechnol 40(12):1329–1356. https://doi.org/10.1007/s10295-013-1332-0
Van Der Merwe JD, Joubert E, Manley M, De Beer D, Malherbe CJ, Gelderblom WC (2009) In vitro hepatic biotransformation of aspalathin and nothofagin, dihydrochalcones of rooibos (Aspalathus linearis), and assessment of metabolite antioxidant activity. J Agricult Food Chem 58(4):2214–2220. https://doi.org/10.1021/jf903917a
Varki A, Cummings R, Esko J, Freeze H, Hart G, Marth J (1999) Essentials of glycobiology. Cold spring harbor laboratory press, New York
Veitch NC, Grayer RJ (2011) Flavonoids and their glycosides, including anthocyanins. Nat Prod Rep 28(10):1626–1695. https://doi.org/10.1039/C1NP00044F
Wang X (2009) Structure, mechanism and engineering of plant natural product glycosyltransferases. FEBS Lett 583(20):3303–3309. https://doi.org/10.1016/j.febslet.2009.09.042
Waridel P, Wolfender JL, Ndjoko K, Hobby KR, Major HJ, Hostettmann K (2001) Evaluation of quadrupole time-of-flight tandem mass spectrometry and ion-trap multiple-stage mass spectrometry for the differentiation of C-glycosidic flavonoid isomers. J Chromatogr A 926(1):29–41. https://doi.org/10.1016/S0021-9673(01)00806-8
Weymouth-Wilson AC (1997) The role of carbohydrates in biologically active natural products. Nat Prod Rep 14(2):99–110. https://doi.org/10.1039/NP9971400099
Xia T, Eiteman MA (2017) Quercetin glucoside production by engineered Escherichia coli. Appl Microbiol Biotechnol 182(4):1358–1370. https://doi.org/10.1007/s12010-017-2403-x
Xiao J (2017) Dietary flavonoid aglycones and their glycosides: which show better biological significance? Crit Rev Food Sci Nutr 57:1874–1905. https://doi.org/10.1080/10408398.2015.1032400
Xiao J, Chen T, Cao H (2014) Flavonoid glycosylation and biological benefits. Biotechnol Adv. https://doi.org/10.1016/j.biotechadv.2014.05.004
Xiao J, Capanoglu E, Jassbi AR, Miron A (2016) Advance on the flavonoid C-glycosides and health benefits. Crit Rev Food Sci Nutr 56(sup1):29–45. https://doi.org/10.1080/10408398.2015.1067595
Yoon JA, Kim BG, Lee WJ, Lim Y, Chong Y, Ahn JH (2012) Production of a novel quercetin glycoside through metabolic engineering of Escherichia coli. Appl Environ Microbiol 78(12):4256–4262. https://doi.org/10.1128/AEM.00275-12
Acknowledgements
This work was supported by a grant from the Next-Generation BioGreen 21 Program (SSAC, grant # PJ01111901), Rural Development Administration, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 890 kb)
Rights and permissions
About this article
Cite this article
Shrestha, A., Pandey, R.P., Dhakal, D. et al. Biosynthesis of flavone C-glucosides in engineered Escherichia coli. Appl Microbiol Biotechnol 102, 1251–1267 (2018). https://doi.org/10.1007/s00253-017-8694-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8694-6