Abstract
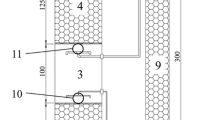
The compartmentalized anaerobic reactor (CAR) is a patent novel high-rate reactor, which consists of three compartments. The reactor has a great potential for application due to its many advantages. In this work, the microbial consortium, spatial distribution, and their relationship with performance of CAR were investigated by means of polymerase chain reaction, denaturing gradient gel electrophoresis, and fluorescence in situ hybridization. The results showed that the predominant archaea were Methanobacterium, Methanosaeta, and Methanospirillum, and the predominant bacteria were Firmicutes, Deltaproteobacteria, Spirochaetes, Actinobacteria, and Gammaproteobacteria in the microbial consortium. The methanogenic archaea (MA), the hydrogen-producing acetogenic bacteria (HAB), and the hydrolytic fermentative bacteria (HFB) were found to be predominant in the upper, middle, and bottom compartments, respectively. The results revealed that the granular sludge took on a stratified microbial structure. The HFB, HAB, and MA were located in the outer shell, middle layer, and core, respectively. The microbial populations from the bottom compartment were relatively homogeneous in the granular sludge, and from the middle and upper compartments, they were relatively heterogeneous in the granular sludge. The microbial consortia and their spatial distribution were in accordance with the organic loading rate and chemical components in the three compartments.








Similar content being viewed by others
References
Amann RI (1995) Molecular microbial ecology manual. Springer, Berlin, pp. 331–345
Amann RI, Ludwig W, Schleifer KH (1995) Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiol Rev 59(1):143–169
APHA–AWWA–WEF (2012) Standard methods for the examination of water and wastewater, 22nd edn. American Public Health Association, Washington, DC
Bialek K, Kim J, Lee C, Collins G, Mahony T, O’Flaherty V (2011) Quantitative and qualitative analyses of methanogenic community development in high-rate anaerobic bioreactors. Water Res 45(3):1298–1308
Calderón K, Rodelas B, Cabirol N, González-López J, Noyola A (2011) Analysis of microbial communities developed on the fouling layers of a membrane-coupled anaerobic bioreactor applied to wastewater treatment. Bioresour Technol 102(7):4618–4627
Cavinato C, Fatone F, Bolzonella D, Pavan P (2010) Thermophilic anaerobic co-digestion of cattle manure with agro-wastes and energy crops: comparison of pilot and full scale experiences. Bioresour Technol 101(2):545–550
Cho S.-K., Kim D.-H., Jeong I.-S., Shin, H.-S. and Oh, S.-E. (2013) Application of low-strength ultrasonication to the continuous anaerobic digestion processes: UASBr and dry digester. Bioresour Technol. doi:10.1016/j.biortech.2013.03.116
Demirel B, Yenigun O, Onay TT (2005) Anaerobic treatment of dairy wastewaters: a review. Process Biochem 40(8):2583–2595
Gao R, Cao Y, Yuan X, Zhu W, Wang X, Cui Z (2012) Microbial diversity in a full-scale anaerobic reactor treating high concentration organic cassava wastewater. Afr J Biotechnol 11(24):6494–6500
Gutell RR, Larsen N, Woese CR (1994) Lessons from an evolving rRNA: 16S and 23S rRNA structures from a comparative perspective. Microbiol Rev 58(1):10–26
Hansen KH, Ahring BK, Raskin L (1999) Quantification of syntrophic fatty acid-β-oxidizing bacteria in a mesophilic biogas reactor by oligonucleotide probe hybridization. Appl Environ Microbiol 65(11):4767–4774
Harmsen H, Akkermans A, Stams A, De Vos W (1996a) Population dynamics of propionate-oxidizing bacteria under methanogenic and sulfidogenic conditions in anaerobic granular sludge. Appl Environ Microbiol 62(6):2163–2168
Harmsen H, Kengen H, Akkermans A, Stams A, De Vos W (1996b) Detection and localization of syntrophic propionate-oxidizing bacteria in granular sludge by in situ hybridization using 16S rRNA-based oligonucleotide probes. Appl Environ Microbiol 62(5):1656–1663
Ji J, Zheng P, Zhang J, Lu H (2011) Performance of compartmentalized anaerobic reactor. Transactions of the CSAE 27(11):252–256 (in Chinese with English abstract)
Ji J, Zheng K, Xing Y. and Zheng P (2012a) Hydraulic characteristics and their effects on working performance of compartmentalized anaerobic reactor. Bioresour Technol 116:47–52
Ji J, Zheng p, Zhang J, Zhang M (2012b) Vertical distribution of compartmentalized anaerobic reactor (CAR) performances. J Chem Eng Chin Univ 6:028 (in Chinese with English abstract)
Lee C, Kim J, Hwang K, O’Flaherty V, Hwang S (2009) Quantitative analysis of methanogenic community dynamics in three anaerobic batch digesters treating different wastewaters. Water Res 43(1):157–165
Liu F, Wang S, Zhang J, Zhang J, Yan X, Zhou H, Zhao G, Zhou Z (2009) The structure of the bacterial and archaeal community in a biogas digester as revealed by denaturing gradient gel electrophoresis and 16S rDNA sequencing analysis. J Appl Microbiol 106(3):952–966
Merlino G, Rizzi A, Villa F, Sorlini C, Brambilla M, Navarotto P, Bertazzoni B, Zagni M, Araldi F, Daffonchio D (2012) Shifts of microbial community structure during anaerobic digestion of agro-industrial energetic crops and food industry byproducts. J Chem Technol Biotechnol 87:1302–1311
Nelson MC, Morrison M, Schanbacher F, Yu Z (2012) Shifts in microbial community structure of granular and liquid biomass in response to changes to infeed and digester design in anaerobic digesters receiving food-processing wastes. Bioresour Technol 107:135–143
Panichnumsin P, Ahring B, Nopharatana A, Chaiprasert P (2012) Microbial community structure and performance of an anaerobic reactor digesting cassava pulp and pig manure. Water Sci Technol 66(7):1590
Poh P, Chong M (2009) Development of anaerobic digestion methods for palm oil mill effluent (POME) treatment. Bioresour Technol 100(1):1–9
Satoh H, Miura Y, Tsushima I, Okabe S (2007) Layered structure of bacterial and archaeal communities and their in situ activities in anaerobic granules. Appl Environ Microbiol 73(22):7300–7307
Shin SG, Lee S, Lee C, Hwang K, Hwang S (2010) Qualitative and quantitative assessment of microbial community in batch anaerobic digestion of secondary sludge. Bioresour Technol 101(24):9461–9470
Stahl DA, Flesher B, Mansfield HR, Montgomery L (1988) Use of phylogenetically based hybridization probes for studies of ruminal microbial ecology. Appl Environ Microbiol 54(5):1079–1084
Takai K, Horikoshi K (2000) Rapid detection and quantification of members of the archaeal community by quantitative PCR using fluorogenic probes. Appl Environ Microbiol 66(11):5066–5072
Tale V, Maki J, Struble C, Zitomer D (2011) Methanogen community structure-activity relationship and bioaugmentation of overloaded anaerobic digesters. Water Res 45(16):5249–5256
Vanysacker L, Declerck SA, Hellemans B, De Meester L, Vankelecom I, Declerck P (2010) Bacterial community analysis of activated sludge: an evaluation of four commonly used DNA extraction methods. Appl Microbiol Biotechnol 88(1):299–307
Woese CR (1987) Bacterial evolution. Microbiol Rev 51(2):221
Yang K, Yu Y, Hwang S (2003) Selective optimization in thermophilic acidogenesis of cheese-whey wastewater to acetic and butyric acids: partial acidification and methanation. Water Res 37(10):2467–2477
Yu Y, Lee C, Kim J, Hwang S (2005) Group-specific primer and probe sets to detect methanogenic communities using quantitative real-time polymerase chain reaction. Biotechnol Bioeng 89(6):670–679
Yu Y, Kim J, Hwang S (2006) Use of real-time PCR for group-specific quantification of aceticlastic methanogens in anaerobic processes: population dynamics and community structures. Biotechnol Bioeng 93(3):424–433
Acknowledgments
Financial supports of this work by Major Scientific and Technological Specialized Project of Zhejiang Province (No. 2010C13001) and Specialized Research Fund for the Doctoral Program of Higher Education (No. J20120067) are greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xing, Yj., Ji, Jy., Zheng, P. et al. Microbial consortium and its spatial distribution in a compartmentalized anaerobic reactor. Appl Microbiol Biotechnol 98, 1357–1366 (2014). https://doi.org/10.1007/s00253-013-5003-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-013-5003-x