Abstract
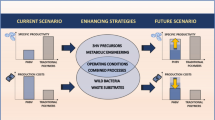
Polyhydroxybutyrate (PHB) is an attractive substitute for petrochemical plastic due to its similar properties, biocompatibility, and biodegradability. The cost of scaled-up PHB production inhibits its widespread usage. Intensive researches are growing to reduce costs and improve thermomechanical, physical, and processing properties of this green biopolymer. Among cheap substrates which are used for reducing total cost of PHB production, some C1 carbon sources, e.g., methane, methanol, and CO2 have received a great deal of attention due to their serious role in greenhouse problem. This article reviews the fundamentals of strategies for reducing PHA production and moves on to the applications of several cheap substrates with a special emphasis on methane, methanol, and CO2. Also, some explanation for involved microorganisms including the hydrogen-oxidizing bacteria and methanotrophs, their history, culture condition, and nutritional requirements are given. After description of some important strains among the hydrogen-oxidizing and methanotrophic producers of PHB, the article is focused on limitations, threats, and opportunities for application and their future trends.



Similar content being viewed by others
References
Ackermann JU, Babel W (1997) Growth-associated synthesis of poly (hydroxybutyric acid) in Methylobacterium rhodesianum as an expression of internal bottleneck. Appl Microbiol Biotechnol 47:144–149
Ackermann JU, Babel W (1998) Approaches to increase the economy of the PHB production. Polymer Degrad Stabil 59:183–186
Akaraonye E, Keshavarz T, Roy I (2010) Production of polyhydroxyalkanoates: the future green materials of choice (review). J Chem Technol Biotechnol 85:732–743
Albuquerque MGE, Eiro M, Torres C, Nunes BR, Reis MAM (2007) Strategies for the development of a side stream process for polyhydroxyalkanoate (PHA) production from sugar cane molasses. J Biotechnol 130:411–421
Ammann ECB, Reed L, Durichek JE (1968) Gas consumption and growth rate of Hydrogenomonas eutropha in continuous culture. Appl Microbiol 16:822–826
Anderson AJ, Dawes EA (1990) Occurrence, metabolism, metabolic role, and industrial uses of bacterial polyhydroxyalkanoates. Microbiol Rev 54:450–472
Anderson AJ, Williams DR, Taidi B, Dawes EA, Ewing DF (1992) Studies on copolyester synthesis by Rhodoccocus ruber and factors influencing the molecular mass of polyhydroxybutyrate accumulated by Methylobacterium extorquens and Alcaligenes etrophus. FEMS Microbiol Rev 103:93–102
Anthony C (1982) The biochemistry of methylotrophs. Academic, New York
Aragno M, Schlegel HG (1992) The mesophilic hydrogen oxidizing (Knallgas) bacteria. In: Balows A, Triiper HG, Dworkin M, Harder W, Schleifer KH (eds) The prokaryotes, 2nd edn. Springer, New York, pp 344–384
Asada Y, Miyake M, Miyake J, Kurane R, Tokiwa Y (1999) Photosynthetic accumulation of poly(hydroxybutyrate) by cyanobacteria the metabolism and potential for CO2 recycling. Int J Biol Macromol 25:37–42
Asenjo JA, Suk J (1986) Microbial conversion of methaneintopoly-beta-hydroxybutrate (PHB)-growth and intracellular product accumulation in a type-II methanotroph. J Ferment Technol 64:271–278
Auman AJ, Speake CC, Lidstrom ME (2001) nifH sequences and nitrogen fixation in type I and type II methanotrophs. Appl Environ Microbiol 67:4009–4016
Babel W (1992) Pecularities of methylotrophs concerning over flow metabolism, especially the synthesis of polyhydroxyalkanoates. FEMS Microbiol Rev 103:141–148
Babel W, Mothes G (1994) Methylobacterium rhodesianum MB 126 possesses two acetoacetyl-CoA reductases. Arch Microbiol 161:277–280
Bae S, Kwak K, Kim S, Chung S, Igarashi Y (2001) Isolation and characterization of CO2-fixing hydrogen-oxidizing marine bacteria. J Biosci Bioeng 91:442–448
Bengtsson S, Hallquist J, Werker A, Welander T (2007) Acidogenic fermentation of industrial wastewaters: effects of chemostat retention time and pH on volatile fatty acids production. J Biochem Eng 40:492–499
Bhubalan K, Loo CY, Lee WH, Yamamoto T, Doi Y, Sudesh K (2008) Controlled biosynthesis and characterization of poly(3-hydroxybutyrate-3-hydroxyvalerate-co-3-hydroxyhexanoate) from mixtures of palm kernel oil and 3HV-precursors. Polymer Degrad Stabil 93:17–23
Bongers L (1970) Energy generation and utilization in hydrogen bacteria. J Bacteriol 104:145–151
Bormann EJ, Leißner M, Roth M, Beer B, Metzner K (1998) Production of polyhydroxybutyrate by Ralstonia eutropha from protein hydrolysates. Appl Microbiol Biotechnol 50:604–607
Bourque D, Ouellette B, Andre G, Groleau D (1992) Production of poly-β-hydroxybutyrate from methanol: characterization of a new isolate of Methylobacterium extorquens. Appl Microbiol Biotechnol 37:7–12
Bourque D, Pomerleau Y, Groleau D (1995) High-cell-density production of poly-β-hydroxybutyrate (PHB) from Methylobacterium extorquens: production of high-molecular-mass PHB. Appl Microbiol Biotechnol 44:367–376
Bowman JP (2001) Family I. Methylococcaceae and Family V. Methylocystaceae. In: Bergey’s manual of systematic bacteriology. Williams & Wilkins, Baltimore, pp 256–270, pp. 411–420
Bowman JP (2006) The methanotrophs-the families Methylococcacceae and Methylocystaceae. In: The prokaryotes a: handbook on the biology of bacteria. Springer, NewYork, pp 266–289
Bowman JP, Sly LI, Nichols PD, Hayward A (1993) Revised taxonomy of the methanotrophs: description of Methylobacter gen. nov., emendation of Methylococcus, validation of Methylosinus and Methylocystis species, and a proposal that the family Methylococcaceae includes only the group I methanotrophs. Int J Syst Bacteriol 43:735–753
Brandi H, Gross RA, Lenz RW, Fuller RC (1990) Plastic from bacteria and for bacteria: poly (β-hydroxyalkanoates) as natural, biocompatible, and biodegradable polyesters. Adv Biochem Eng Biotechnol 41:77–93
Braunegg G, Lefebvre G, Genser KF (1998) Poly hydroxyalkanoate, biopolyesters from renewable resources: physiological and engineering aspects. J Biotechnol 65:127–161
Budde CF, Riedel SL, Hübner F, Risch S, Popović MK, ChoKyun R, Sinskey AJ (2011) Growth and polyhydroxybutyrate production by Ralstonia eutropha in emulsified plant oil medium. Appl Microbiol Biotechnol 89:1611–1619
Byrom D (1987) Polymer synthesis by microorganisms: technology and economics. Trends Biotechnol 5:246–250
Cebron A, Bodrossy L, Stralis-Pavese N, Singer AC, Thompson IP, Prosser JI, Murrell JC (2007) Nutrient amendments in soil DNA stable isotope probing experiments reduce the observed methanotroph diversity. Appl Environ Microbiol 73:798–807
Chanprateep S (2010) Current trends in biodegradable polyhydroxyalkanoates (review). J Biosci Bioeng 110:621–632
Chen CW, Don TM, Yen HF (2006) Enzymatic extruded starch as a carbon source for the production of poly(3-hydroxybutyrateco-3-hydroxyvalerate by Haloferax mediterranei. Process Biochem 41:2289–2296
Choi J, Lee SY (1997) Process analysis and economic evaluation for PHB production by fermentation. Bioprocess Eng 17:335–342
Choi J, Lee SY (1999a) Factors affecting the economics of polyhydroxyalkanoate production by bacterial fermentation. Appl Microbiol Biotechnol 51:13–21
Choi J, Lee SY (1999b) High-level production of poly(3-hydroxybutyrateco-3-hydroxyvalerate) by fed-batch culture of recombinant Escherichia coli. Appl Environ Microbiol 65:4363–4368
Choi J, Kim JH, Daneial M, Lebeault JM (1989) Optimization of growth medium and poly-β-hydroxybutyric acid production from methanol in Methylobacterium organophilium. Korean J Appl Microbiol Bioeng 17:392–396
Dalton H (1981) Methane mono-oxygenase from a variety of microbes. In: Microbial growth on C, compounds.Heyden & Son, London, pp 1–10
Daniel M, Choi JH, Kim JH, Lebeault JM (1992) Effect of nutrient deficiency on accumulation and relative molecular weight of poly-β-hydroxybutyric acid by methylotrophic bacterium, Pseudomonas 135. Appl Microbiol Biotechnol 37:702–706
Dedysh SN (2002) Methanotrophic bacteria of acids phagnum bogs. Mikrobiologiia 71:741–754
Dedysh SN, Berestovskaya YY, Vasylieva LV, Belova SE, Khmelenina VN, Suzina NE, Trotsenko YA, Liesack W, Zavarzin GA (2004) Methylocella tundrae sp. nov., a novel methanotrophic bacterium from acidic tundra peatlands. Int J Syst Evol Microbiol 54:151–156
Di Donato P, Anzelmo G, Tommonaro G, Fiorentino G, Nicolaus B, Poli A (2009) Vegetable wastes as suitable biomass feedstock for biorefineries. New Biotechnol 25(Suppl):S257
Dobroth ZT, Hu S, Coats ER, McDonald RG (2011) Polyhydroxybutyrate synthesis on biodiesel wastewater using mixed microbial consortia. Bioresour Technol 102:3352–3359
Doi Y (1990) Microbial polyesters. VHC Publishers, New York
Doi Y, Steinbüchel A (2002) Biopolymers. Wiley, Weinheim
Doronina NV, Ezhov VA, Trotsenko YA (2008) Growth of Methylobacteriumtrichosporium OB3b on methanol and poly-β-hydroxybutyrate biosynthesis. Appl Biochem Microbiol 44:182–184
Du G, Chen J, Yu J, Lun S (2001) Continuous production of poly-3-hydroxybutyrate by Ralstonia eutropha in a two-stage culture system. J Biotechnol 88:59–65
Du G, Chen LXL, Yu J (2004) High-efficiency production of bioplastics from biodegradable organic solids. J Polym Environ 12:89–94
Du C, Sabirova J, Soetaert W, Lin SKC (2012) Polyhydroxyalkanoates production from low-cost sustainable raw. Materials Curr Chem Biol 6(1):14–25
Dunfield PF, Khmelenina VN, Suzina NE, Trotsenko YA, Dedysh SN (2003) Methylocella silvestris sp nov., a novel methanotroph isolated from an acidic forest cambisol. Int J Syst Evol Microbiol 53:1231–1239
Dunfield PF, Yuryev A, Senin P, Smirnova AV, Stott MB, Hou S, Ly B, Saw JH, Zhou Z, Ren Y, Wang J, Mountain BW, Crowe MA, Weatherby TM, Bodelier PL, Liesack W, Feng L, Wang L, Alam M (2007) Methane oxidation by an extremely acidophilic bacterium of the phylum Verrucomicrobia. Nature 450:879–882
Fidler S, Dennis D (1992) Polyhydroxyalkanoate production in recombinant Escherichia coli. FEMS Microbiol Rev 103:231–236
Follner CG, Babel W, Valentin HE, Steinbüchel A (1993) Expression of polyhydroxy alkanoic-acid-biosynthesis genes in methylotrophic bacteria relying on the ribulose monophosphate pathway. Appl Microbiol Biotechnol 40:284–291
Foster JF, Litchfield JH (1964) A continuous culture apparatus for the microbial utilization of hydrogen produced by electrolysis of water in closed-cycle space systems. Biotechnol Bioeng 6:44l–456l
Fukui T, Doi Y (1998) Efficient production of polyhydroxyalkanoates from plant oils by Alcaligenes eutrophus and its recombinant strain. Appl Microbiol Biotechnol 49:333–336
Ganduri VSRK, Ghosh S, Patnaik PR (2005) Mixing control as a device to increase PHB production in batch fermentation with co-cultures of lactobacillus delbrueckii and Ralstonia eutropha. Process Biochem 40:257–264
Ghatnekar MS, Pai JS, Ganesh M (2002) Production and recovery of poly-3-hydroxybutyrate from Methylobacterium sp V49. J Chem Technol Biotechnol 77:444–448
Gouda MK, Swellam AE, Omar SH (2001) Production of PHB by a Bacillus megaterium strain using sugarcane molasses and corn steep liquor as sole carbon and nitrogen sources. Microbiol Res 15:201–207
Govorukhina NI, Trotsenko YA (1991) Poly-β-hydroxybutyrate contents of methylotrophic bacteria with different routes methanol assimilation. Appl Biochem Microbiol 27:80–83
Graham DW, Chaudhary JA, Hanson RS, Arnold RG (1993) Factors affecting competition between type I and type II methanotrophs in two-organism, continuous-flow reactors. Microb Ecol 25:1–17
Grothe E, Moo-Young M, Chisti Y (1999) Fermentation optimization for the production of poly (β-hydroxybutyric acid) microbial thermoplastic. Enz Microbial Technol 25:132–141
Haas R, Jin B, Zepf FT (2008) Production of poly(3-hydroxybutyrate) from waste potato starch. Biosci Biotechnol Biochem 72:253–256
Halami PM (2008) Production of polyhydroxyalkanoate from starch by the native isolate Bacillus cereus CFR06. World J Microbiol Biotechnol 24:805–812
Hanson RS, Hanson TE (1996) Methanotrophic bacteria. Microbiol Rev 60:439–471
Hayashi NR, Peerapornpisal Y, Nishihara H, Ishii M, Igarashi Y, Kodama T (1994) Isolation and cultivation of thermophilic cyanobacteria from hot springs of northern Thailand. J Ferment Bioeng 78:179–181
Haywood GW, Anderson AJ, Dawes EA (1989) A survey of the accumulation of novel polyhydroxyalkanoates by bacteria. Biotechnol Lett 11:471–476
Haywood GW, Anderson AJ, Ewing DF, Dawes EA (1990) Accumulation of a polyhydroxyalkanoates containing primarily 3-hydroxydecanoate from simple carbohydrate substrates by Pseudomonas sp. Strain NCIMB 40135. Appl Environ Microbiol 56:3354–3359
Hazer DB, Kılıçay E, Hazer B (2012) Poly(3-hydroxyalkanoate)s: diversification and biomedical applications: a state of the art review. Mater Sci Eng 32:637–647
Heinzle E, Lafferty RM (1980) A kinetic model for growth and syntheseis of poly-β-hydroxybutyric acid (PHB) in Alkaligenes etruphus H16. Eur J Appl Microbiol Biotechnol 11:8–16
Helm J, Wendlandt KD, Rogge G, Kappelmeyer U (2006) Characterizing a stable methane-utilizing mixed culture used in the synthesis of a high-quality biopolymer in an open system. J Appl Microbiol 101:387–395
Helm J, Wendlandt KD, Jechorek M, Stottmeister U (2008) Potassium deficiency results in accumulation of ultra-high molecular weight poly-beta-hydroxybutyrate in a methane utilizing mixed culture. J Appl Microbiol 105:1054–1061
Heyer J, Berger U, Hardt M, Dunfield PF (2005) Methylohalobius crimeensis gen. nov., sp. nov., a moderately halophilic, methanotrophic bacterium isolated from hyper-saline lakes of Crimea. Int J Syst Evol Microbiol 55:1817–1826
Hilger U, Sattler K, Littkowsky U (1991) Studies on the growth associated accumulation of poly-hydroxybutyric acid with Methylobacterium rhodesianum Z. Zentralbl Mikrobiol 146:83–88
Hofer P, Vermette P, Groleau D (2011) Production and characterization of polyhydroxyalkanoates by recombinant Methylobacterium extorquens: combining desirable thermal properties with functionality. Biochem Eng J 54:26–33
Hori K, Kaneko M, Tanji Y, Xing XH, Unno H (2002) Construction of self-disruptive Bacillus megaterium in response to substrate exhaustion for polyhydroxybutyrate production. Appl Microbiol Biotechnol 59:211–216
Huang TY, Duan KJ, Huang SY, Chen CW (2006) Production of polyhydroxyalkanoates from inexpensive extruded rice bran and starch by Haloferax mediterranei. J Ind Microbiol Biotechnol 33:701–706
Ibrahim MHA, Steinbüchel A (2010) High-cell-density cyclic fed-batch fermentation of a poly(3-hydroxybutyrate)-accumulating thermophile, Chelatococcus sp. strain MW10. Appl Environ Microbiol 76:7890–7895
Ishii M, Miyake T, Satoh T, Sugiyama H, Oshima Y, Kodama T, Igarashi Y (1997) Autotrophic carbon dioxide fixation in Acidianus brierleyi. Arch Microbiol 166:368–371
Ishizaki A, Tanaka K (1990) Batch culture of Alcaligenes eutrophus ATCC 17697T using recycled gas closed circuit culture system. J Ferment Bioeng 69:170–174
Ishizaki A, Tanaka K (1991) Production of poly-β-hydroxybutyric acid from carbon dioxide by Alcaligenes eutrophus ATCC 17697T. J Ferment Bioeng 70:254–25
Ishizaki A, Tanaka K, Taga N (2001) Microbial production of poly(hydroxybutyrate) from CO2. Appl Microbiol Biotechnol 57:6–12
Islam T, Jensen S, Reigstad LJ, Larsen O, Birkeland NK (2008) Methane oxidation at 55 degrees C and pH 2 by a thermo acidophilic bacterium belonging to the Verrucomicrobia phylum. Proc Natl Acad Sci USA 105:300–304
Jing D, Jiaying X (2011) Biosynthesis of PHB, a new packaging material by methane-utilizing mixed culture HD6T. Adv Mater Res 380:244–247
João MBT, Cavalheiro M, Catarina MD, Grandfils C, Fonseca MMR (2009) Poly(3-hydroxybutyrate) production by Cupriavidus necator using waste glycerol. Proc Biochem 44:509–515
Kabilan S, Ayyasamy M, Jayavel S, Paramasamy G (2012) Pseudomonas sp. as a source of medium chain length polyhydroxyalkanoates for controlled drug delivery: perspective. Int J Microbiol 2012:317828
Kaewkannetra P, Tanonkeo P, Tanamool V, Imai I (2008) Biorefinery of sweet sorghum juice into value added product of biopolymer. J Biotechnol 136:S412
Kallio RE, Harrington AA (1960) Sudanophilic granules and lipid of Pseudomonas methanica. J Bacteriol 80:321–324
Kessler B, Witholt B (2001) Factors involved in the regulatory network of polyhydroxyalkanoate metabolism. J Biotechnol 86:97–104
Khanna S, Srivastava AK (2005a) Statistical media optimization studies for growth and PHB production by Ralstosnia eutropha. Process Biochem 40:2173–2182
Khanna S, Srivastava AK (2005b) Recent advances in microbial polyhydroxyalkanoates. Process Biochem 40:607–619
Khanna S, Srivastava AK (2005c) A simple structured mathematical model for biopolymer (PHB) production. Biotech Prog 21:830–838
Khosravi-Darani K, Vasheghani-Farahani E (2005a) Microorganisms and systems for production of poly(hydroxybutyrate) as a biodegradable polymer. Iran J Chem Chemical Eng 24:1–19
Khosravi-Darani K, Vasheghani-Farahani E (2005b) Application of supercritical fluid extraction in biotechnology. Crit Rev Biotechnol 25:1–12
Khosravi-Darani K, Vasheghani-Farahani E, Shojaosadati SA (2003a) Application of the Plackett–Burman design for the optimization of poly(hydroxybutyrate) production by Ralstonia eutropha. Iran J Biotechnol 1:155–161
Khosravi-Darani K, Vasheghani-Farahani E, Yamini Y (2003b) Solubility of poly hydroxybutyrate in supercritical carbon dioxide. J Chem Eng Data 48:860–863
Khosravi-Darani K, Vasheghani-Farahani E, Shojaosadati SA (2004a) Application of the Taguchi design for production of poly(hydroxybutyrate) by Ralstonia eutropha. Iran J Chem Chemical Eng 23:131–136
Khosravi-Darani K, Vasheghani-Farahani E, Shojaosadati SA, Yamini Y (2004b) The effect of process variable on poly(hydroxybutyrate) recovery by supercritical fluid cell disruption. Biotechnol Prog 20:1757–1765
Khosravi-Darani K, Vasheghani-Farahani E, Tanaka K (2006) Hydrogen-oxidizing bacteria as poly(hydroxybutyrate) producers. Iran J Biotechnol 4:193–196
Kim SB (2000) Production of poly(hydroxybutyrate) from inexpensive substrates. Enz Microb Technol 27:774–777
Kim SW, Kim P, Lee HS, Kim JH (1996) High production of poly-β-hydroxybutyrate (PHB) from Methylobacterium organophilum under potassium limitation. Biotechnol Lett 18:25–30
Kodama T, Igarashi Y, Minoda Y (1975) Isolation and culture conditions of a bacterium grown on hydrogen and carbon dioxide. Agr Biol Chem 36:77–82
Koller M, Bona R, Braunegg G, Hermann C, Horvat P, Kroutil M, Martinz J, Neto J, Pereira L, Varila P (2005) Production of polyhydroxyalkanoates from agricultural waste and surplus materials. Biomacromol 6:561–565
Koller M, Bona R, Chiellini E, Fernandes EG, Horvat P, Kutschera C, Hesse P, Braunegg G (2008) Polyhydroxyalkanoate production from whey by Pseudomonas hydrogenovora. Bioresour Technol 99:4854–4863
Koller M, Hesse P, Salerno A, Reiterer A, Braunegg G (2011) A viable antibiotic strategy against microbial contamination in biotechnological production of polyhydroxyalkanoates from surplus whey. Biomass Bioenerg 35:748–753
Korotkova N, Lidstrom ME (2001) Connection between poly-betahydroxybutyrate biosynthesis and growth on C1 and C2 compounds in the methylotroph Methylobacterium extorquens AM1. J Bacteriol 183:1038–1046
Kozhevnikov IV, Volova TG, Hai T, Steinbüchel A (2010) Cloning and molecular organization of the polyhydroxyalkanoic acid synthase gene (phaC) of Ralstonia eutropha strain B5786. Appl Biochem Microbiol 46:140–147
Kunasundari B, Sudesh K (2011) Isolation and recovery of microbial polyhydroxyalkanoates. Exp Poly Lett 5:620–634
Lafferty RM (1979) Microbiological method. US Patent 4138291
Lara LM, Huisman GW (1999) Metabolic engineering of poly(3-hydroxyalkanoates): from DNA to plastic. Microbiol Mol Biol Rev 63:21–53
Lee SY (1996) Bacterial polyhydroxyalkanoates. Biotechnol Bioeng 49:1–14
Lee SY, Choi J, Wong HH (1999) Recent advances in poly(hydroxylalkanoate) production by bacterial fermentation: mini-review. Int J Biol Macromol 25:31–36
Lemos PC, Serafim LS, Reis MAM (2006) Synthesis of polyhydroxyalkanoates from different short-chain fatty acids by mixed cultures submitted to aerobic dynamic feeding. J Biotechnol 122:226–238
Lidstrom ME (2006) Aerobic methylotrophic prokaryotes. In: The prokaryotes, volume 2: ecophysiology and biochemistry. Springer, New York, pp 618–634
Listewnik HF, Wendlandt KD, Jechorek M, Mirschel G (2007) Process design for the microbial synthesis of poly-β-hydroxybutyrate (PHB) from natural gas. Eng Life Sci 7:278–282
López-Cuellar MR, Alba-Flores J, Gracida Rodríguez JN, Pérez-Guevara F (2011a) A viable antibiotic strategy against microbial contamination in biotechnologica production of polyhydroxyalkanoates from surplus whey. Biomass Bioenerg 35:748–753
López-Cuellar MR, Alba-Flores J, Gracida Rodríguez JN, Pérez-Guevara F (2011b) Production of polyhydroxyalkanoates (PHAs) with canola oil as carbon source. Int J Biol Macromol 48:74–80
Lu X, Zhang J, Wu Q, Chen GQ (2003) Enhanced production of poly (hydroxybutyrate-co-hydroxyhexanoate) via manipulation the fatty acid β-oxidation pathway in E. coli. FEMS Microbiol Lett 221:97–101
Madison LL, Huisman GW (1999) Metabolic engineering of poly(3-hydroxyalkanoates): from DNA to plastic. Microbiol Mol Biol Rev 63:21–53
Malik KA, Schlegel HG (1980) Enrichment and isolation of new nitrogen-fixing hydrogen oxidizing bacteria. FEMS Microbiol Lett 8:101–104
Miyake M, Erata M, Asada Y (1996) A thermophilic cyanobacterium, Synechococcus sp. MA19, capable of accumulating poly-β-hydroxybutyrate. J Ferment Bioeng 82(5):512–514
Miyake M, Takase K, Narato M, Khatipov E, Schnackenberg J, Shirai M, Kurane R, Asada Y (2000) Polyhydroxybutyrate production from carbon dioxide by cyanobacteria. Appl Biochem Biotechnol A Enzy Eng Biotechnol 84–86:991–1002
Mokhtari-Hosseini ZB, Vasheghani-Farahani E, Heidarzadeh-Vazifekhoran A, Shojaosadati SA, Karimzadeh R, Khosravi-Darani K (2009a) Statistical media optimization for growth and PHB production from methanol by a methylotrophic bacterium. Bioresour Technol 100:2436–2443
Mokhtari-Hosseini ZB, Vasheghani-Farahani E, Shojaosadati SA, Karimzadeh R, Heidarzadeh-Vazifekhoran A (2009b) Effect of feed composition on PHB production from methanol by HCDC Methylobacterium extorquens (DSMZ 1340). J Chem Technol Biotechnol 84:1136–1139
Morgan-Sagastume F, Karlsson A, Johansson P, Pratt S, Boon N, Lant P, Werker A (2010) Production of polyhydroxyalk in open, mixed cultures from a waste sludge stream containing high levels of soluble organics, nitrogen and phosphorus. Water Res 44:5196–5211
Morse M, Liao Q, Criddle CS, Frank CW (2011) An aerobic biodegradation of the microbial copolymer poly(3-hydroxybutyrate-co-3-hydroxyhexanoate): effects of comonomer content, processing history, and semi-crystalline morphology. Polym 52:547–555
Mothes G, Rivera HS, Babel B (1997) Competition between β-ketothiolase and citrate synthase during poly (hydroxybutyrate) synthesis in Methylobacterium rhodesianum. Arch Microbiol 166:405–410
Mothes G, Ackermann JU, Babel W (1998) Regulation of poly(β-hydroxybutyrate) synthesis in Methylobacterium rhodesianum MB 126 growing on methanol or fructose. Arch Microbiol 169:360–363
Mulchandani A, Luong JHT, Grom C (1989) Substrate inhibition kinetics for microbial growth and syntheseis of poly-β-hydroxybutyric acid in Alkaligenes etruphus ATCC17679. Appl Microbiol Biotechnol 30:11–17
Murrell J, Dalton H (1983) Nitrogen-fixation in obligate methanotrophs. J Gen Microbiol 129:3481–3486
Nguyen HH, Elliott SJ, Yip JH, Chan SI (1998) The particulate methane monooxygenase from M. capsulatus (Bath) is a novel copper-containing three-subunit enzyme. J Biol Chem 273:7957–7966
Nikel PI, Almeida AD, Melillo EC, Galvagno MA, Pettinari MJ (2006) New recombinant Escherichia coli strain tailored for the production of poly(3-hydroxybutyrate) from agro-industrial by-products. Appl Environ Microbiol 72:3949–3954
Nishihara H, Igarashi Y, Kodama T (1991) Growth characteristics and high cell-density cultivation of a marine obligately chemolithoautotrophic hydrogen-oxidizing bacterium Hydrogenovibrio marinus strain MH-110 under a continuous gas-flow system. J Ferment Bioeng 72:358–361
Oakley C, Murrell J (1988) Nifh genes in the obligate methane oxidizing bacteria. FEMS Microbiol Lett 49:53–57
Omar S, Rayes A, Eqaab A, Viss I, Steinbüchel A (2011) Optimization of cell growth and poly(3-hydroxbutyrate) accumulation on date syrup by a Bacillus megaterium strain. Biotechnol Lett 23:1119–1123
Op den Camp HJM, Islam T, Stott MB, Harhangi HR, Hynes A, Schouten S, Jetten MSM, Birkeland NK, Pol A, Dunfield PF (2009) Environmental, genomic, and taxonomic perspectives on methanotrophic Verrucomicrobia. Env Microbiol Rep 1:293–306
Pantazaki AA, Papaneophytou CP, Pritsa AG, Liakopoulou-Kyriakides M, Kyriakidis DA (2009) Production of polyhydroxyalkanoates from whey by Thermus thermophilus HB8. Process Biochem 44:847–853
Papaneophytou CP, Pantazaki AA, Kyriakidis DA (2009) An extracellular polyhydroxybutyrate depolymerase in Thermus thermophilus HB8. Appl Microbiol Biotechnol 83:659–668
Park SJ, Ahn WS, Green PR, Lee SY (2001) Biosynthesis of Poly(hydroxybutyrate-co-3-hydroxyvalerate-co-3-hydroxyhexanoate) by metabolically engineered Escherichia coli strains. Biotechnol Bioeng 74:81–86
Patnaik PR (2005) Perspectives in the modelling and optimization of PHB production by pure and mixed cultures. CritRev Biotechnol 25:153–171
Patwardhan PR, Srivastava AK (2004) Model-based fed-batch cultivation of R. eutropha for enhanced biopolymer production. Biochem Eng J 20:21–28
Pfluger AR, Wu WM, Pieja AJ, Wan J, Rostkowski KH, Criddle CS (2011) Selection of type I and type II methanotrophic proteobacteria in a fluidized bed reactor under non-sterile conditions. Bioresour Technol 102:9919–9926
Pieja AJ, Rostkowski KH, Criddle CS (2011a) Distribution and selection of poly-hydroxybutyrate production capacity in methanotrophic proteobacteria. Microb Ecol 62:564–573
Pieja AJ, Sundstrom ER, Criddle CS (2011b) Poly-hydroxybutyrate metabolism in the type II methanotroph Methylocystis parvus OBBP. Appl Environ Microbiol 77:6012–6019
Pieja AJ, Sundstrom ER, Criddle CS (2012) Cyclic, alternating methane and nitrogen limitation increases PHB production in a methanotrophic community. Bioresour Technol 107:385–392
Pilla S (2011). Handbook of bioplastics and biocomposites engineering applications. Wiley, New York, pp. 373–396
Pinkwart M, Schneider K, Schlegel HG (1983) Purification and properties of the membrane-bound hydrogenase from N2-fixing Alcaligenes latus. Biochim Biophys Acta Protein Struct Mol Enzymol 745:267–278
Pol A, Heijmans K, Harhangi HR, Tedesco D, Jetten MS, OpdenCamp HJ (2007) Methanotrophy below pH1 by a new Verrucomicrobia species. Nature 450:874–878
Povolo S (2010) Production of polyhydroxyalkanoates (PHAs) with canola oil as carbon source. Int J Biol Macromol 48:74–80
Povolo S, Casella S (2003) Bacterial production of PHA from lactose and cheese whey permeate. Macromol Symp 197:1–9
Povolo S, Toffano P, Basaglia M, Casella S (2010) Polyhydroxyalkanoates production by engineered Cupriavidus necator from waste material containing lactose. Bioresour Technol 101:7902–7907
Powell KA, Collinson BA, Richardson KR (1980) Microbiological process for the production of poly(beta-hydroxybutyric acid) and microorganisms for use therein. Eur Patent Appl 80300432.4
Quillaguamán J, Hashim S, Bento F, Mattiasson B, Hatti-Kaul R (2005) Poly(β-hydroxybutyrate) production by a moderate halophile, Halomonas boliviensis LC1 using starch hydrolysate as substrate. J Appl Microbiol 99:151–157
Raje P, Srivastava AK (1998) Updated mathematical model fed-batch strategies for poly-β-hydroxybutyrate (PHB) ptoduction by Alkaligenes etruphus. Bioresour Technol 64:185–192
Ramadas NV, Singh SK, Soccol CR, Pandey A (2009) Polyhydroxybutyrate production using agro-industrial residue as substrate by Bacillus sphaericus NCIM 5149. Braz Arch Biol Technol 52:17–23
Ramadas NV, Soccol CR, Pandey A (2010) A statistical approach for optimization of polyhydroxybutyrate production by Bacillus sphaericus ncim 5149 under submerged fermentation using central composite design. Appl Biochem Biotechnol 162:996–1007
Reddy CSK, Ghai R, Rashmi R, Kalia VC (2003) Polyhydroxyalkanoates: an overview. Bioresour Technol 87:137–146
Repask R (1966) Characteristics of hydrogen bacteria. Biotechnol Bioeng 8:217–235
Ribera RG, Monteoliva-Sanchez M, Ramos-Cormenzana A (2001) Production of polyhydroxyalkanoates by Pseudomonas putida KT2442 harbouring pSK2665 in waste water from olive oil mills (alpechin). J Biotechnol 4:116–119
Rudnik E (2008). Compostable polymer materials. Elsevier, Amsterdam, p. 21
Ryu HW, Hahn SK, Chang YK, Chang HN (1997) Production of poly(3-hydroxybutyrate) by high cell density fed-batch culture of Alcaligenes eutrophus with phosphate limitation. Biotechnol Bioeng 55:28–32
Santimano MC, Prabhu NN, Garg S (2009) PHA production using low-cost agro-industrial wastes by Bacillus sp. strain COL1/A6. J Microbiol 4:89–96
Schink B, Schlegel H (1978) Hydrogen, metabolism in aerobic hydrogen oxidizing bacteria. Biochimie 60(3):297–305
Schlegel HG, Gottschalk G, Von Bartha R (1961) Formation and utilization of poly-β-hydroxybutyic acid by Knallgas bacteria (Hydrogenomonas). Nature 191:463
Schrader J, Schilling M, Holtmann D, Sell D, Filho MV, Marx A, Vorholt JA (2009) Methanol-based industrial biotechnology: current status and future perspectives of methylotrophic bacteria (review). Trends Biotechnol 27:107–115
Scott D, Brannan J, Higgins IJ (1981) The effect of growth conditions on intracytoplasmic membranes and methane mono-oxygenase activities in Methylosinus trichosporium OB3b. J Gen Microbiol 125:63–72
Shah NN, Hanna ML, Jackson KJ, Taylor RT (1996a) Batch cultivation of Methylosinus trichosporium OB3B: IV production of hydrogen-driven soluble or particulate methane monooxygenase activity. Biotechnol Bioeng 45:229–238
Shah NN, Hanna ML, Taylor RT (1996b) Batch cultivation of Methylosinus trichosporiumOB3b. 5: characterization of poly(hydroxybutyrate) production under methane-dependent growth conditions. Biotechnol Bioeng 49:161–171
Shah-Hosseini S, Sadeghi MT, Khosravi-Darani K (2003) Simulation and model validation of batch poly(β-hydroxybutyrate) production process using Ralstonia eutropha. Iran J Chem Chemical Eng 22:35–41
Sharma L, Mallick N (2008) Exploitation of municipal and aquacultural discharges for poly-β-hydroxybutyrate production in cyanobacterium, Nostoc muscorum. Res J Biotechnol 3:282–287
Sheu DS, Wang YT, Lee CY (2000) Rapid detection of polyhydroxyalkanoate-accumulating bacteria isolated from the environment by colony PCR. Microbiol 146:2019–2025
Simon-Colin C, Raguenes G, Crassous P, Moppert X, Guezennec J (2008) A novel MCL-PHA produced on coprah oil by Pseudomonas guezennei biovar.tikehau, isolated from a ‘kopara’ mat of French Polynesia. Int J BiolMacromol 43:176–181
Slepecky RA, Law JH (1961) Synthesis and degradation of poly-b-hydroxybutyric acid in connection with sporulation of Bacillus megaterium. J Bacteriol 82:37–42
Solaiman D, Ashby R, Hotchkiss A, Foglia T (2006) Biosynthesis of medium-chain-length poly(hydroxyalkanoates) from soy molasses. Biotechnol Lett 28:57–162
Song H, Xin J, Zhang Y, Kong W, Xia C (2011) Poly-3-hydroxybutyrate production from methanol by Methylosinus trichosporium IMV3011 in the non-sterilized fed-batch fermentation. Afr J Microbiol Res 5:5022–5029
Sonnleitner B, Heinzle E, Braunegg G, Lafferty RM (1979) Formal kinetics of poly-β-hydroxybutyric acid (PHB) production in Alkaligenes etruphus H16 and Mycoplana rubera R14 with respect to the dissolved oxygen tension in ammonium limited batch-cultures. Eur J Appl Microbiol Biotechnol 7:1–10
Steinbüchel A, Füchtenbusch B (1998) Bacterial and other biological systems for polyester production. Trends Biotechnol 16:419–427
Steinbüchel A, Lutke-Eversloh T (2003) Metabolic engineering and pathway construction for biotechnological production of relevant polyhydroxyalkanoates in microorganisms. Biochem Eng J 16:81–96
Sudesh K, Doi AY (2000) Synthesis, structure and properties of polyhydroxyalkanoates: biological polyesters. Prog Polym Sci 25:1503–1555
Sugimoto T, Tsuge T, Tanaka K, Ishizaki A (1999) Control of acetic acid concentration by pH-stat continuous substrate feeding in heterotrophic culture phase of two-stage cultivation of Alcaligenes eutrophus for production of PHB from CO2, H2 and O2 under non-explosive condition. Biotechnol Bioeng 62:625–631
Suzuki T, Yamane T, Shimizu S (1986a) Mass production of poly-β-hydroxybutyric acid by fully automatic fed-batch culture of methylotroph. Appl Microbiol Biotechnol 23:322–329
Suzuki T, Yamane T, Shimizu S (1986b) Kinetics and effect of nitrogen source feeding on production of poly(hydroxybutyric acid) by fed-batch culture. Appl Microbiol Biotechnol 23:366–369
Suzuki T, Yamane T, Shimizu S (1986c) Mass production of (poly-hydroxybutyric acid) by fed-batch culture with controlled carbon/nitrogen feeding. Appl Microbiol Biotechnol 24:370–374
Suzuki T, Deguchi H, Yamane T, Shimizu S, Gekko K (1988) Control of molecular weight of (poly-hydroxybutyric acid) produced in fed-batch culture of Protomonas extorquence. Appl Microbiol Biotechnol 27:487–491
Suzuki H, Kishimoto M, Kamoshita Y, Omasa T, Katakura Y, Suga KI (2000) On-line control of feeding of medium components to attain high cell density. Bioprocess Eng 22:433–440
Taga N, Tanaka K, Ishizaki A (1997) Effects of rheological change by addition of carboxymethylcellulose in culture media of an air-lift fermentor on poly-d-3-hydroxybutyric acid productivity in autotrophic culture of hydrogen-oxidizing bacterium. Alcaligenes eutrophus. Biotechnol Bioeng 53:529–533
Taidi B, Anderson AJ, Dawes EA, Byrom D (1994) Effect of carbon source and concentration on the molecular mass of poly(3-hydroxybutyrate) production by Methylobacterium extorquens and Alcaligenes etrophus. Appl Microbiol Biotechnol 40:786–790
Takeshita T, Ishizaki A (1996) Influence of hydrogen limitation on gaseous substrate utilization in autotrophic culture of Alcaligenes eutrophus ATCC 17697T. J Ferment Bioeng 81:83–86
Takeshita T, Tanaka K, Ishizaki A, Stanbury PF (1993a) Development of a dissolved hydrogen sensor and its application to evaluation of hydrogen mass transfer. J Ferment Bioeng 76:148–150
Takeshita T, Tanaka K, Ishizaki A, Stanbury PF (1993b) Studies on dissolved hydrogen behavior in autotrophic culture of A. eutrophus 17697T. J Fac Agr Kyushu Univ 38:55–64
Tanaka K, Ishizaki A (1994) Production of poly-d-3-hydroxybutyric acid from carbon dioxide by a two-stage culture method employing Alcaligenes eutrophus ATCC 17697T. J Ferment Bioeng 77:425–427
Tanaka K, Ishizaki A, Takeshita T, Kanemaru T, Shimoji T, Kawano T (1993) Equipment and operation for fermentative PHB production using gaseous substrate to guarantee safety from explosion. J Chem Eng Japan 26:225–227
Tanaka K, Ishizaki A, Kanamaru T, Kawano T (1995) Production of poly(d-3-hydroxybutyrate) from CO2, H2, and CO2 by high cell density autotrophic cultivation of Alcaligenes eutrophus. Biotechnol Bioeng 45:268–275
Tanaka K, Miyawaki K, Yamaguchi A, Khosravi-Darani K, Matsusaki H (2011) Cell growth and P(3HB) accumulation from CO2 of a carbon monoxide-tolerant hydrogen-oxidizing bacterium, Ideonella sp. O-1. Appl Microbiol Biotechnol 92:1161–1169
Tohyama M, Patarinska T, Qiang Z, Shimizu K (2002) Modeling of the mixed culture and periodic control for PHB production. Biochem Eng J 10:157–173
US Environmental Protection Agency. Methane: sources and emissions. http://www.epa.gov/outreach/sources.html. Accessed April21, 2011
Ueda S, Matsumoto S, Takagi A, Yamane T (1992) Synthesis of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) from methanol and n-amyl alkohol by methylotrophic bacteria Paraccocus denitrificans and Methylobacterium extorquens. Appl Environ Microbiol 58:3574–3579
Van Dien SJ, Lidstrom ME (2002) Stiochimetric model for evaluating the methabolic capabilities of the facultative methylotroph Methylobacterium extorquens AM1, with application to reconstruction of C3 and C4 methabolism. Biotechnol Bioeng 78:296–312
Vandamme P, Coenye T (2004) Taxonomy of the genus Cupriavidus: a tale of lost and found. Int J Syst Evol Microbiol 54:2285–2289
Van-Thuoc D, Quillaguamán J, Mamo G, Mattiasson B (2008) Utilization of agricultural residues for poly(3-hydroxybutyrate) production by Halomonas boliviensis LC1. J Appl Microbiol 104:420–428
Vecherskaya M, Dijkema C, Stams AJ (2001) Intracellular PHB conversion in a type II methanotroph studied by13CNMR. J Ind Microbiol Biotechnol 26:15–21
Vincenzini M, De Philippis R (1999) Polyhydroxyalkanoates. In: Chemicals from Microalgae, Cohen Z, London, Taylor and Francis, pp 292-352
Volova TG, Voĭnov NA (2004) Study of Ralstonia eutropha culture producing polyhydroxyalkanoates on products of coal processing. Prikl Biokhim Mikrobiol 40:296–300
Volova TG, Kalacheva GS, Altukhova OV (2002) Autotrophic synthesis of polyhydroxyalkanoates by the bacteria Ralstonia eutropha in the presence of carbon monoxide. App Microbiol Biotechnol 58:675–678
Wang J, Yu HQ (2007) Biosynthesis of polyhydroxybutyrate and extracellular polymeric substances by Ralstonia eutropha ATCC 17699 in batch cultures. Appl Microbiol Biotechnol 75:871–878
Wendlandt KD, Jechorek M, Helm J, Stottmeister U (1998) Production of PHB with a high molecular mass from methane. Poly Degrad Stabil 59:191–194
Wendlandt KD, Jechorek M, Helm J, Stottmeister U (2001) Producing poly-3-hydroxybutyrate with a high molecular mass from methane. J Biotechnol 86:127–133
Wendlandt KD, Geyer W, Mirschel G, Al-HajHemidi F (2005) Possibilities for controlling a PHB accumulation process using various analytical methods. J Biotechnol 117:119–129
Wendlandt KD, Stottmeister U, Helm J, Soltmann B, Jechorek M, Beck M (2010) The potential of methane-oxidizing bacteria for applications in environmental biotechnology (review). Eng Life Sci 10:87–102
Whittenbury R, Phillips KC, Wilkinson JF (1970) Enrichment, isolation and some properties of methane-utilizing bacteria. J Gen Microbiol 61:205–218
Wise MG, McArthur JV, Shimkets LJ (1999) Methanotroph diversity in land fill soil: isolation of novel type I and type II methanotrophs whose presence was suggested by culture independent 16S ribosomal DNA analysis. Appl Environ Microbiol 65:4887–4897
Wong HH, Lee SY (1998) Poly(3-hydroxybutyrate) production from whey by high density cultivation of recombinant Escherichia coli. Appl Microbiol Biotechnol 50:30–33
Wong AL, Chua H, Yu PH (2000) Microbial production of polyhydroxyalkanoates by bacteria isolated from oil wastes. App Biochem Biotechnol 84–86:843–857
Xin JY, Zhang YX, Zhang S, Xia CG, Li SB (2007) Methanol production from CO2 by resting cells of the methanotrophic bacterium Methylosinus trichosporium IMV3011. J Basic Microbiol 47:426–435
Xin J, Zhang Y, Dong J, Song H, Xia C (2011) An experimental study on molecular weight of polyhydroxybutyrate (PHB) accumulated in Methylosinus trichosporium IMV 3011. Afr J Biotechnol 10:7078–7087
Yamane T (1993) Yield of poly-D-3-hydroxybutyrate from various, carbon sources: a theoretical study. Biotechnol Bioeng 41:165–170
Yamane T, Chen XF, Ueda S (1996a) Growth associated production of poly(3-hydroxyvalerate) from n-pentanol by a methylotrophic bacterium. Paracoccus denitrificans. Appl Environ Microbiol 62:380–384
Yamane, Fukunaga M, Dee YW (1996b) Increase PHB production by high-cell-density fed-batch culture of Alcaligunes latus, a growth associated PHB producer. Biotechnol Bioeng 50:197–202
Yan S, Tyagi RD, Surampalli RY (2006) Polyhydroxyalkanoates (PHA) production using wastewater as carbon source and activated sludge as microorganisms. Water Sci Technol 53:175–180
Yezza A, Fournier D, Halasz A, Hawari J (2006) Production of polyhydroxyalkanoates from methanol by a new methylotrophic bacterium Methylobacterium sp. GW2. Appl Microbiol Biot 73:211–218
Yoo S, Kim WS (1994) Cybernetic model for synthesis of poly-β-hydroxybutyric acid in Alcaligenes etrophus. Biotechnol Bioeng 43:1043–1051
Zahn JA, DiSpirito AA (1996) Membrane-associated methane monooxygenase from M. capsulatus (Bath). J Bacteriol 178:1018–1029
Zhang Y, Xin J, Chen L, Song H, Xia C (2008) Biosynthesis of poly-3-hydroxybutyrate with a high molecular weight by methanotroph from methane and methanol. J Natural Gas Chem 17:103–109
Zhao S, Fan C, Hu X, Chen J, Feng H (1993) The microbial production of polyhydroxybutyrate from methanol. Appl Biochem Biotechnol 39(40):191–199
Zinn M, Witholt B, Egli T (2001) Occurrence, synthesis and medical application of bacterial polyhydroxyalkanoate. Adv Drug Deliv Rev 53:5–21
Zuniga C, Morales M, Le Borgne S, Revah S (2011) Production of poly-β-hydroxybutyrate (PHB) by Methylobacterium organophilum isolated from a methanotrophic consortium in a two-phase partition bioreactor. J Hazard Mater 190:876–882
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khosravi-Darani, K., Mokhtari, ZB., Amai, T. et al. Microbial production of poly(hydroxybutyrate) from C1 carbon sources. Appl Microbiol Biotechnol 97, 1407–1424 (2013). https://doi.org/10.1007/s00253-012-4649-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4649-0