Abstract
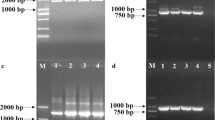
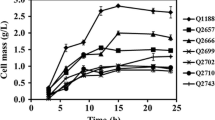
2,3-Butanediol is an important bio-based chemical product, because it can be converted into several C4 industrial chemicals. In this study, a lactate dehydrogenase-deleted mutant was constructed to improve 2,3-butanediol productivity in Enterobacter aerogenes. To delete the gene encoding lactate dehydrogenase, λ Red recombination method was successfully adapted for E. aerogenes. The resulting strain produced a very small amount of lactate and 16.7% more 2,3-butanediol than that of the wild-type strain in batch fermentation. The mutant and its parental strain were then cultured with six different carbon sources, and the mutant showed higher carbon source consumption and microbial growth rates in all media. The 2,3-butanediol titer reached 69.5 g/l in 54 h during fed-batch fermentation with the mutant, which was 27.4% higher than that with the parental strain. With further optimization of the medium and aeration conditions, 118.05 g/l 2,3-butanediol was produced in 54 h during fed-batch fermentation with the mutant. This is by far the highest titer of 2,3-butanediol with E. aerogenes achieved by metabolic pathway engineering.



Similar content being viewed by others
References
Booth IR (1985) Regulation of cytoplasmic pH in bacteria. Microbiol Rev 49:359–378
Celinska E, Grajek W (2009) Biotechnological production of 2,3-butanediol—current state and prospects. Biotechnol Adv 27:715–725
Converti A, Perego P (2002) Use of carbon and energy balances in the study of the anaerobic metabolism of Enterobacter aerogenes at variable starting glucose concentrations. Appl Microbiol Biotechnol 59:303–309
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci USA 97:6640–6645
Dharmadi Y, Murarka A, Gonzalez R (2006) Anaerobic fermentation of glycerol by Escherichia coli: a new platform for metabolic engineering. Biotechnol Bioeng 94:821–829
Gao JA, Xu H, Li QJ, Feng XH, Li S (2010) Optimization of medium for one-step fermentation of inulin extract from Jerusalem artichoke tubers using Paenibacillus polymyxa ZJ-9 to produce R, R-2,3-butanediol. Bioresource Technol 101:7076–7082
Geckil H, Barak Z, Chipman DM, Erenler SO, Webster DA, Stark BC (2004) Enhanced production of acetoin and butanediol in recombinant Enterobacter aerogenes carrying Vitreoscilla hemoglobin gene. Bioprocess Biosyst Eng 26:325–330
Ji XJ, Huang H, Ouyang PK (2011) Microbial 2,3-butanediol production: a state-of-the-art review. Biotechnol Adv 29:351–364
Ji XJ, Huang H, Zhu JG, Ren LJ, Nie ZK, Du J, Li S (2010) Engineering Klebsiella oxytoca for efficient 2,3-butanediol production through insertional inactivation of acetaldehyde dehydrogenase gene. Appl Microbiol Biotechnol 85:1751–1758
Lu Y, Zhao H, Zhang C, Lai Q, Wu X, Xing XH (2010) Alteration of hydrogen metabolism of ldh-deleted Enterobacter aerogenes by overexpression of NAD+ -dependent formate dehydrogenase. Appl Microbiol Biotechnol 86:255–262
Ma CQ, Wang AL, Qin JY, Li LX, Ai XL, Jiang TY, Tang HZ, Xu P (2009) Enhanced 2,3-butanediol production by Klebsiella pneumoniae SDM. Appl Microbiol Biotechnol 82:49–57
Murphy KC, Campellone KG (2003) Lambda Red-mediated recombinogenic engineering of enterohemorrhagic and enteropathogenic E. coli. BMC Mol Biol 4:11
Perego P, Converti A, Del Borghi A, Canepa P (2000) 2, 3-Butanediol production by Enterobacter aerogenes: selection of the optimal conditions and application to food industry residues. Bioprocess Biosyst Eng 23:613–620
Petrov K, Petrova P (2009) High production of 2,3-butanediol from glycerol by Klebsiella pneumoniae G31. Appl Microbiol Biotechnol 84:659–665
Petrov K, Petrova P (2010) Enhanced production of 2,3-butanediol from glycerol by forced pH fluctuations. Appl Microbiol Biotechnol 87:943–949
Yang T, Rao Z, Zhang X, Lin Q, Xia H, Xu Z, Yang S (2011) Production of 2,3-butanediol from glucose by GRAS microorganism Bacillus amyloliquefaciens. J Basic Microbiol. doi:10.1002/jobm.201100033
Zeng AP, Biebl H, Deckwer WD (1990a) 2, 3-Butanediol production by Enterobacter aerogenes in continuous culture: role of oxygen supply. Appl Microbiol Biotechnol 33:264–268
Zeng AP, Biebl H, Deckwer WD (1990b) Effect of pH and acetic acid on growth and 2,3-butanediol production of Enterobacter aerogenes in continuous culture. Appl Microbiol Biotechnol 33:485–489
Zeng AP, Biebl H, Deckwer WD (1991) Production of 2,3-butanediol in a membrane bioreactor with cell recycle. Appl Microbiol Biotechnol 34:463–468
Zhang LY, Sun JA, Hao YL, Zhu JW, Chu J, Wei DZ, Shen YL (2010) Microbial production of 2,3-butanediol by a surfactant (serrawettin)-deficient mutant of Serratia marcescens H30. J Ind Microbiol Biotechnol 37:857–862
Zhao HX, Ma K, Lu Y, Zhang C, Wang LY, Xing XH (2009a) Cloning and knockout of formate hydrogen lyase and H(2)-uptake hydrogenase genes in Enterobacter aerogenes for enhanced hydrogen production. Int J Hydrogen Energy 34:186–194
Zhao HX, Ma K, Lu Y, Zhang C, Wang LY, Xing XH (2009b) Perturbation of formate pathway for hydrogen production by expressions of formate hydrogen lyase and its transcriptional activator in wild Enterobacter aerogenes and its mutants. Int J Hydrogen Energy 34:5072–5079
Acknowledgements
This research was supported by the R&D Program of MKE/KEIT (No. 10035578, Development of 2,3-butanediol and derivative production technology for C-Zero bio-platform industry). The authors would also like to thank Macrogen, Inc. for providing the E. aerogenes KCTC2190 full genome sequence.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jung, MY., Ng, C.Y., Song, H. et al. Deletion of lactate dehydrogenase in Enterobacter aerogenes to enhance 2,3-butanediol production. Appl Microbiol Biotechnol 95, 461–469 (2012). https://doi.org/10.1007/s00253-012-3883-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-3883-9