Abstract
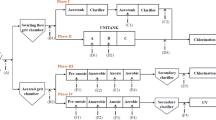
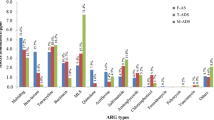
In this study, the abilities of two anaerobic digestion processes used for sewage sludge stabilization were compared for their ability to reduce the quantities of three genes that encode resistance to tetracycline (tet(A), tet(O), and tet(X)) and one gene involved with integrons (intI1). A two-stage, thermophilic/mesophilic digestion process always resulted in significant decreases in the quantities of tet(X) and intI1, less frequently in decreases of tet(O), and no net decrease in tet(A). The thermophilic stage was primarily responsible for reducing the quantities of these genes, while the subsequent mesophilic stage sometimes caused a rebound in their quantities. In contrast, a conventional anaerobic digestion process rarely caused a significant decrease in the quantities of any of these genes, with significant increases occurring more frequently. Our results demonstrate that anaerobic thermophilic treatment was more efficient in reducing quantities of genes associated with the spread of antibiotic resistance compared to mesophilic digestion.


Similar content being viewed by others
References
Aitken MD, Sobsey MD, Shehee M, Blauth KE, Hill VR, Farrell JB, Nappier SP, Walters GW, Crunk PL, van Abel N (2005) Laboratory evaluation of thermophilic-anaerobic digestion to produce class A biosolids. 2. Inactivation of pathogens and indicator organisms in a continuous flow reactor followed by batch treatment. Water Environ Res 77:3028–3036
Alanis AJ (2005) Resistance to antibiotics: are we in the post-antibiotic era? Arch Med Res 36:697–705
Amann RI, Ludwig W, Schleifer K-H (1995) Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiol Rev 59:143–169
Aminov RI, Chee-Sanford JC, Garrigues N, Teferedegne B, Krapac IJ, White BA, Mackie RI (2002) Development, validation, and application of PCR primers for detection of tetracycline efflux genes of gram-negative bacteria. Appl Environ Microbiol 68:1786–1793
Auerbach EA, Seyfried EE, McMahon KD (2007) Tetracycline resistance genes in activated sludge wastewater treatment plants. Water Res 41:1143–1151
Berg G, Berman D (1980) Destruction by anaerobic mesophilic and thermophilic digestion of viruses and indicator bacteria indigenous to domestic sludges. Appl Environ Microbiol 39:361–368
Chopra I, Roberts M (2001) Tetracycline antibiotics: mode of action, applications, molecular biology, and epidemiology of bacterial resistance. Microbiol Mol Biol Rev 65:232–260
Davelos AL, Kinkel LL, Samac DA (2004) Spatial variation in frequency and intensity of antibiotic interactions among Streptomycetes from prairie soil. Appl Environ Microbiol 70:1051–1058
Falagas ME, Bliziotis IA (2007) Pandrug-resistant gram-bacteria in an urban wastewater treatment plant. FEMS Microbiol Ecol 55:322–329
Firl SJ (2006) Prevalence, fate, and characterization of antibiotic-resistant bacteria in municipal wastewater treatment plants. MS Thesis, University of Minnesota, Minneapolis, MN
Ghosh S (2007) Characterization of antibiotic resistance in soils exposed to manure from farms using subtherapeutic antibiotics for growth promotion. Ph. D Thesis, University of Minnesota, Minneapolis, MN
Grewal SK, Rajeev S, Sreevatsan S, Michel FC (2006) Persistence of Mycobacterium avium subsp. paratuberculosis and other zoonotic pathogens during simulated compositing, manure packing, and liquid storage of dairy manure. Appl Environ Microbiol 72:565–574
Han I, Congeevaram S, Park J (2009) Improved control of multiple-antibiotic-resistance-related microbial risk in swine manure wastes by autothermal thermophilic aerobic digestion. Water Sci Technol 59:267–271
Iranpour R, Cox HHJ (2006) Recurrence of fecal coliforms and Salmonella species in biosolids following thermophilic anaerobic digestion. Water Environ Res 78:1005–1012
Lang NL, Smith SR (2008) Time and temperature inactivation kinetics of enteric bacteria relevant to sewage sludge processes for agricultural use. Water Res 42:2229–2241
Livermore DM (2003) Bacterial resistance: origins, epidemiology, and impact. Clin Infect Dis 26:S11–S23
Mazel D (2006) Integrons: agents of bacterial evolution. Nat Rev Microbiol 4:608–620
Muyzer G, Dewaal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Ng L-K, Martin I, Alfa M, Mulvey MR (2001) Multiplex PCR for the detection of tetracycline resistant genes. Mol Cell Probes 15:209–215
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Schluter A, Szczepanowski R, Pühler A, Top EM (2007) Genomics of IncP-1 antibiotic resistance plasmids isolated from wastewater treatment plants provides evidence for a widely accessible drug resistance gene pool. FEMS Microbiol Rev 31:449–477
Smith DL, Dushoff J, Morris JG (2005) Agricultural antibiotics and human health. PLoS Med 2:e232
Snyder L, Champness W (2007) Molecular genetics of bacteria, 3rd edn. ASM Press, Washington DC
Summers AO (2002) Generally overlooked fundamentals of bacterial genetics and ecology. Clin Infect Dis 34:S85–S92
Tchobanoglous G, Burton FL, Stensel HD (2003) Wastewater engineering: treatment and reuse, 4th edn. Metcalf and Eddy, Inc, McGraw-Hill, Boston
US EPA (1994) Standards for the use or disposal of sewage sludge. Code of Federal Regulations, Title 40, Chapter 503
Wagner AO, Gstraunthaler G, Illmer P (2008) Survival of bacterial pathogens during the thermophilic anaerobic digestion of biowaste: laboratory experiments and in situ validation. Anaerobe 14:181–183
Wright GD (2007) The antibiotic resistome: the nexus of chemical and genetic diversity. Nat Rev Microbiol 5:175–186
Zhang T, Zhang M, Zhang X, Fang HHP (2009a) Tetracycline resistance genes and tetracycline resistant lactose-fermenting Enterobacteriaceae in activated sludge of sewage treatment plants. Environ Sci Technol 43:3455–3460
Zhang X, Zhang T, Zhang M, Fang HHP, Cheng S-P (2009b) Characterization and quantification of class 1 integrons and associated gene cassettes in sewage treatment plants. Appl Microbiol Biotechnol 82:1169–1177
Acknowledgements
This research was supported by a grant from the Center for Urban and Regional Affairs to TML and a fellowship from Geomatrix Consultants to SJR.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghosh, S., Ramsden, S.J. & LaPara, T.M. The role of anaerobic digestion in controlling the release of tetracycline resistance genes and class 1 integrons from municipal wastewater treatment plants. Appl Microbiol Biotechnol 84, 791–796 (2009). https://doi.org/10.1007/s00253-009-2125-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-2125-2