Abstract
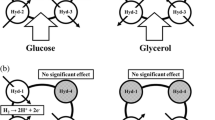
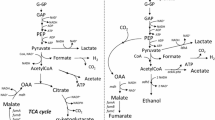
We improved the hydrogen yield from glucose using a genetically modified Escherichia coli. E. coli strain SR15 (ΔldhA, ΔfrdBC), in which glucose metabolism was directed to pyruvate formate lyase (PFL), was constructed. The hydrogen yield of wild-type strain of 1.08 mol/mol glucose, was enhanced to 1.82 mol/mol glucose in strain SR15. This figure is greater than 90 % of the theoretical hydrogen yield of facultative anaerobes (2.0 mol/mol glucose). Moreover, the specific hydrogen production rate of strain SR15 (13.4 mmol h−1 g−1 dry cell) was 1.4-fold higher than that of wild-type strain. In addition, the volumetric hydrogen production rate increased using the process where cells behaved as an effective catalyst. At 94.3 g dry cell/l, a productivity of 793 mmol h−1 l−1 (20.2 l h−1 l−1 at 37 °C) was achieved using SR15. The reported productivity substantially surpasses that of conventional biological hydrogen production processes and can be a trigger for practical applications.



Similar content being viewed by others
References
Alam KY, Clark DP (1989) Anaerobic fermentation balance of Escherichia coli as observed by in vivo nuclear magnetic resonance spectroscopy. J Bacteriol 171:6213–6217
Benemann J (1996) Hydrogen biotechnology: progress and prospects. Nat Biotechnol 14:1101–1103
Chin HL, Chen ZS, Chou CP (2003) Fedbatch operation using Clostridium acetobutylicum suspension culture as biocatalyst for enhancing hydrogen production. Biotechnol Prog 19:383–388
Clark DP (1989) The fermentation pathways of Escherichia coli. FEMS Microbiol Rev 5:223–234
Inui M, Kawaguchi H, Murakami S, Vertès AA, Yukawa H (2004) Metabolic engineering of Corynebacterium glutamicum for fuel ethanol production under oxygen-deprivation conditions. J Mol Microbiol Biotechnol 8:243–254
Kawaguchi H, Hashimoto K, Hirata K, Miyamoto K (2001) H2 production from algal biomass by a mixed culture of Rhodobium marinum A-501 and Lactobacillus amylovorus 91:277–282
Kim SH, Han SK, Shin HS (2004) Feasibility of biohydrogen production by anaerobic co-digestion of food waste and sewage sludge. Int J Hydrogen Energy 29:1607–1616
Kumar N, Das D (2001) Continuous hydrogen production by immobilized Enterobacter cloacae IIT-BT 08 using lignocellulosic materials as solid matrices. Enzyme Microb Technol 29:280–287
Lee KS, Lo YS, Lo YC, Lin PJ, Chang JS (2003) H2 production with anaerobic sludge using activated-carbon supported packed-bed bioreactors. Biotechnol Lett 25:133–138
Levin D, Pitt L, Love M (2004) Biohydrogen production: prospects and limitations to practical application. Int J Hydrogen Energy 29:173–185
Nandi R, Sengupta S (1998) Microbial production of hydrogen: an overview. Crit Rev Microbiol 24:61–84
Nath K, Das D (2004) Improvement of fermentative hydrogen production: various approaches. Appl Microbiol Biotechnol 65:520–529
Oh Y-K, Seol E-H, Yeol Lee E, Park S (2002) Fermentative hydrogen production by a new chemolithotrophic bacterium Rhodopseudomonas palustris P4. Int J Hydrogen Energy 27:1373–1379
Penfold DW, Macaskie LE (2004) Production of H2 from sucrose by Escherichia coli strains carrying the pUR400 plasmid, which encodes invertase activity. Biotechnol Lett 26:1879–1883
Rachman MA, Nakashimada Y, Kakizono T, Nishio N (1998) Hydrogen production with high yield and high evolution rate by self-flocculated cells of Enterobacter aerogenes in a packed-bed reactor. Appl Microbiol Biotechnol 49:450–454
Sauter M, Böhm R, Böck A (1992) Mutational analysis of the operon (hyc) determining hydrogenase 3 formation in Escherichia coli. Mol Microbiol 6:1523–1532
Sawers G (1994) The hydrogenases and formate dehydrogenases of Escherichia coli. Antonie Van Leeuwenhoek 66:57–88
Sode K, Watanabe M, Makimoto H, Tomiyama M (1998) Effect of hydrogenase 3 overexpression and disruption of nitrate reductase on fermamentive hydrogen production in Escherichia coli. In: Zaborsky OR, Benemann JR, Matsunaga T, Miyake J, San Pietro A (eds) Biohydrogen. Plenum, New York, pp 73–79
Sode K, Watanabe M, Makimoto H, Tomiyama M (1999) Construction and characterization of fermentative lactate dehydrogenase Escherichia coli mutant and its potential for bacterial hydrogen production. Appl Biochem Biotechnol 77–79:317–323
Wu SY, Hung CH, Lin CN, Chen HW, Lee AS, Chang JS (2006) Fermentative hydrogen production and bacterial community structure in high-rate anaerobic bioreactors containing silicone-immobilized and self-flocculated sludge. Biotechnol Bioeng 93:934–946
Yoshida A, Nishimura T, Kawaguchi H, Inui M, Yukawa H (2005) Enhanced hydrogen production from formic acid by formate hydrogen lyase-overexpressing Escherichia coli strains. Appl Environ Microbiol 71:6762–6768
Acknowledgements
We thank R. H. Doi (University of California, Davis) and C. A. Omumasaba (Research Institute of Innovative Technology for the Earth) for helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshida, A., Nishimura, T., Kawaguchi, H. et al. Enhanced hydrogen production from glucose using ldh- and frd-inactivated Escherichiacoli strains. Appl Microbiol Biotechnol 73, 67–72 (2006). https://doi.org/10.1007/s00253-006-0456-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-006-0456-9