Abstract
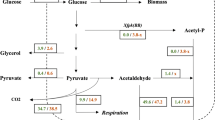
In Saccharomyces cerevisiae the export of 2-oxoglutarate from the mitochondria, catalyzed by Yhm2p, Odc1p and Odc2p or by at least one of these transporters, has recently been shown to be essential for glutamate biosynthesis in glucose-supplemented minimal synthetic (SM) medium without glutamate, because the triple mutant yhm2∆odc1∆odc2∆ displays a growth defect under these conditions. Surprisingly, in this study it was found that yhm2∆odc1∆odc2∆ cells grow like wild-type (WT) cells in the same medium supplemented with non-fermentable carbon sources. Direct transport assays of 2-oxoglutarate/2-oxoglutarate homoexchange activity in mitochondria from WT and yhm2∆odc1∆odc2∆ cells (solubilized and reconstituted into liposomes) showed that the mitochondrial extract from yhm2∆odc1∆odc2∆ was completely inactive at variance with that from WT cells, showing that S. cerevisiae mitochondria do not contain additional proteins capable of catalyzing 2-oxoglutarate transport efficiently besides Yhm2p, Odc1p and Odc2p. Furthermore, quantitative real-time PCR experiments showed that in both WT and yhm2∆odc1∆odc2∆ cells the expression of GDH1 is low on lactate and high on glucose and, vice versa, the expression of GDH3 is high on lactate and low on glucose. These results may be interpreted to indicate that in S. cerevisiae, grown in glucose-supplemented SM medium, glutamate is synthesized by Gdh1p in the cytosol, whereas in lactate-supplemented SM medium glutamate is synthesized by Gdh3p in the mitochondria; therefore, the pathway of ammonia assimilation under fermentative conditions requires export of 2-oxoglutarate from the mitochondria, whereas the alternative pathway under respiratory conditions does not.




Similar content being viewed by others
References
Agrimi G, Di Noia MA, Marobbio CM, Fiermonte G, Lasorsa FM, Palmieri F (2004) Identification of the human mitochondrial S-adenosylmethionine transporter: bacterial expression, reconstitution, functional characterization and tissue distribution. Biochem J 379:183–190. https://doi.org/10.1042/BJ20031664
Agrimi G, Russo A, Scarcia P, Palmieri F (2012) The human gene SLC25A17 encodes a peroxisomal transporter of coenzyme A, FAD and NAD(+). Biochem J 443:241–247. https://doi.org/10.1042/BJ20111420
Avendaño A, Deluna A, Olivera H, Valenzuela L, Gonzalez A (1997) GDH3 encodes a glutamate dehydrogenase isozyme, a previously unrecognized route for glutamate biosynthesis in Saccharomyces cerevisiae. J Bacteriol 179:5594–5597. https://doi.org/10.1128/jb.179.17.5594-5597.1997
Boles E, Lehnert W, Zimmermann FK (1993) The role of the NAD-dependent glutamate dehydrogenase in restoring growth on glucose of a Saccharomyces cerevisiae phosphoglucose isomerase mutant. Eur J Biochem 217:469–477. https://doi.org/10.1111/j.1432-1033.1993.tb18266.x
Castegna A, Scarcia P, Agrimi G, Palmieri L, Rottensteiner H, Spera I, Germinario L, Palmieri F (2010) Identification and functional characterization of a novel mitochondrial carrier for citrate and oxoglutarate in Saccharomyces cerevisiae. J Biol Chem 285:17359–17370. https://doi.org/10.1074/jbc.m109.097188
Cavero S, Vozza A, del Arco A, Palmieri L, Villa A, Blanco E, Runswick MJ, Walker JE, Cerdán S, Palmieri F, Satrústegui J (2003) Identification and metabolic role of the mitochondrial aspartate-glutamate transporter in Saccharomyces cerevisiae. Mol Microbiol 50:1257–1269. https://doi.org/10.1046/j.1365-2958.2003.03742.x
Daum G, Gasser SM, Schatz G (1982) Import of proteins into mitochondria. Energy-dependent, two-step processing of the intermembrane space enzyme cytochrome b2 by isolated yeast mitochondria. J Biol Chem 257:13075–13080
DeLuna A, Avendaño A, Riego L, González A (2001) NADP-glutamate dehydrogenase isoenzymes of Saccharomyces cerevisiae. J Biol Chem 276:43775–43783. https://doi.org/10.1074/jbc.m107986200
Di Noia MA, Todisco S, Cirigliano A, Rinaldi T, Agrimi G, Iacobazzi V, Palmieri F (2014) The human SLC25A33 and SLC25A36 genes of solute carrier family 25 encode two mitochondrial pyrimidine nucleotide transporters. J Biol Chem 289:33137–33148. https://doi.org/10.1074/jbc.M114.610808
Fiermonte G, Dolce V, Palmieri L, Ventura M, Runswick MJ, Palmieri F, Walker JE (2001) Identification of the human mitochondrial oxodicarboxylate carrier. Bacterial expression, reconstitution, functional characterization, tissue distribution, and chromosomal location. J Biol Chem 276:8225–8230. https://doi.org/10.1074/jbc.M009607200
Fiermonte G, Dolce V, David L, Santorelli FM, Dionisi-Vici C, Palmieri F, Walker JE (2003) The mitochondrial ornithine transporter. Bacterial expression, reconstitution, functional characterization, and tissue distribution of two human isoforms. J Biol Chem 278:32778–32783. https://doi.org/10.1074/jbc.M302317200
Fiermonte G, De Leonardis F, Todisco S, Palmieri L, Lasorsa FM, Palmieri F (2004) Identification of the mitochondrial ATP-Mg/Pi transporter. Bacterial expression, reconstitution, functional characterization, and tissue distribution. J Biol Chem 279:30722–30730. https://doi.org/10.1074/jbc.M400445200
Fiermonte G, Paradies E, Todisco S, Marobbio CM, Palmieri F (2009) A novel member of solute carrier family 25 (SLC25A42) is a transporter of coenzyme A and adenosine 3′,5′-diphosphate in human mitochondria. J Biol Chem 284:18152–18159. https://doi.org/10.1074/jbc.M109.014118
Filetici P, Martegani MP, Valenzuela L, González A, Ballario P (1996) Sequence of the GLT1 gene from Saccharomyces cerevisiae reveals the domain structure of yeast glutamate synthase Yeast 12:1359–1366. https://doi.org/10.1002/(sici)1097-0061(199610)12:13<1359::aid-yea3>3.0.co;2-5
Hoyos ME, Palmieri L, Wertin T, Arrigoni R, Polacco JC, Palmieri F (2003) Identification of a mitochondrial transporter for basic amino acids in Arabidopsis thaliana by functional reconstitution into liposomes and complementation in yeast. Plant J 33:1027–1035
Huh W-K, Falvo JV, Gerke LC, Carroll AS, Howson RW, Weissman JS, O’Shea EK (2003) Global analysis of protein localization in budding yeast. Nature 425:686–691. https://doi.org/10.1038/nature02026
Indiveri C, Tonazzi A, Palmieri F (1994) The reconstituted carnitine carrier from rat liver mitochondria: evidence for a transport mechanism different from that of the other mitochondrial translocators. Biochim Biophys Acta 1189:65–73. https://doi.org/10.1016/0005-2736(94)90281-x
Krämer R, Palmieri F (1992) Chapter 16 Metabolite carriers in mitochondria. In: Ernster L (ed) Molecular mechanisms in bioenergetics. Elsevier Science Publishers B. V., Amsterdam. https://doi.org/10.1016/s0167-7306(08)60184-2
Marobbio CMT, Vozza A, Harding M, Bisaccia F, Palmieri F, Walker JE (2002) Identification and reconstitution of the yeast mitochondrial transporter for thiamine pyrophosphate. EMBO J 21:5653–5661. https://doi.org/10.1093/emboj/cdf583
Marobbio CMT, Agrimi G, Lasorsa FM, Palmieri F (2003) Identification and functional reconstitution of yeast mitochondrial carrier for S-adenosylmethionine The. EMBO J 22:5975–5982. https://doi.org/10.1093/emboj/cdg574
Marobbio CMT, Di Noia MA, Palmieri F (2006) Identification of a mitochondrial transporter for pyrimidine nucleotides in Saccharomyces cerevisiae: bacterial expression, reconstitution and functional characterization. Biochem J 393:441–446. https://doi.org/10.1042/bj20051284
Marobbio CMT, Giannuzzi G, Paradies E, Pierri CL, Palmieri F (2008) alpha-Isopropylmalate, a leucine biosynthesis intermediate in yeast, is transported by the mitochondrial oxalacetate carrier. J Biol Chem 283:28445–28453. https://doi.org/10.1074/jbc.M804637200
Miller SM, Magasanik B (1990) Role of NAD-linked glutamate dehydrogenase in nitrogen metabolism in Saccharomyces cerevisiae. J Bacteriol 172:4927–4935. https://doi.org/10.1128/jb.172.9.4927-4935.1990
Minard KI, McAlister-Henn L (2005) Sources of NADPH in yeast vary with carbon source. J Biol Chem 280:39890–39896. https://doi.org/10.1074/jbc.m509461200
Monné M, Miniero DV, Obata T, Daddabbo L, Palmieri L, Vozza A, Nicolardi MC, Fernie AR, Palmieri F (2015) Functional characterization and organ distribution of three mitochondrial ATP–Mg/Pi carriers in Arabidopsis thaliana. Biochim Biophys Acta 1847:1220–1230. https://doi.org/10.1016/j.bbabio.2015.06.015
Moye WS, Amuro N, Rao JK, Zalkin H (1985) Nucleotide sequence of yeast GDH1 encoding nicotinamide adenine dinucleotide phosphate-dependent glutamate dehydrogenase. J Biol Chem 260:8502–8508
Nogae I, Johnston M (1990) Isolation and characterization of the ZWF1 gene of Saccharomyces cerevisiae, encoding glucose-6-phosphate dehydrogenase. Gene 96:161–169. https://doi.org/10.1016/0378-1119(90)90248-p
Palmieri F (2013) The mitochondrial transporter family SLC25: identification, properties and physiopathology. Mol Aspects Med 34:465–484. https://doi.org/10.1016/j.mam.2012.05.005
Palmieri F (2014) Mitochondrial transporters of the SLC25 family and associated diseases: a review. J Inherit Metab Dis 37:565–575. https://doi.org/10.1007/s10545-014-9708-5
Palmieri F, Monné M (2016) Discoveries, metabolic roles and diseases of mitochondrial carriers: a review. Biochim Biophys Acta 1863:2362–2378. https://doi.org/10.1016/j.bbamcr.2016.03.007
Palmieri F, Indiveri C, Bisaccia F, Iacobazzi V (1995) Mitochondrial metabolite carrier proteins: purification, reconstitution, and transport studies. Methods Enzymol 260:349–369. https://doi.org/10.1016/0076-6879(95)60150-3
Palmieri L, Lasorsa FM, Iacobazzi V, Runswick MJ, Palmieri F, Walker JE (1999a) Identification of the mitochondrial carnitine carrier in Saccharomyces cerevisiae. FEBS Lett 462:472–476
Palmieri L, Vozza A, Honlinger A, Dietmeier K, Palmisano A, Zara V, Palmieri F (1999b) The mitochondrial dicarboxylate carrier is essential for the growth of Saccharomyces cerevisiae on ethanol or acetate as the sole carbon source. Mol Microbiol 31:569–577. https://doi.org/10.1046/j.1365-2958.1999.01197.x
Palmieri L, Lasorsa FM, Vozza A, Agrimi G, Fiermonte G, Runswick MJ, Walker JE, Palmieri F (2000) Identification and functions of new transporters in yeast mitochondria. Biochim Biophys Acta 1459:363–369. https://doi.org/10.1016/S0005-2728(00)00173-0
Palmieri L, Agrimi G, Runswick MJ, Fearnley IM, Palmieri F, Walker JE (2001) Identification in Saccharomyces cerevisiae of two isoforms of a novel mitochondrial transporter for 2-oxoadipate and 2-oxoglutarate. J Biol Chem 276:1916–1922. https://doi.org/10.1074/jbc.M004332200
Palmieri F, Agrimi G, Blanco E, Castegna A, Di Noia MA, Iacobazzi V, Lasorsa FM, Marobbio CM, Palmieri L, Scarcia P, Todisco S, Vozza A, Walker J (2006a) Identification of mitochondrial carriers in Saccharomyces cerevisiae by transport assay of reconstituted recombinant proteins. Biochim Biophys Acta 1757:1249–1262. https://doi.org/10.1016/j.bbabio.2006.05.023
Palmieri L, Arrigoni R, Blanco E, Carrari F, Zanor MI, Studart-Guimaraes C, Fernie AR, Palmieri F (2006b) Molecular identification of an Arabidopsis S-adenosylmethionine transporter. Analysis of organ distribution, bacterial expression, reconstitution into liposomes, and functional characterization. Plant Physiol 142:855–865. https://doi.org/10.1104/pp.106.086975
Perlman PS, Mahler HR (1970) Intracellular localization of enzymes in yeast. Arch Biochem Biophys 136:245–259. https://doi.org/10.1016/0003-9861(70)90348-6
Porcelli V, Fiermonte G, Longo A, Palmieri F (2014) The human gene SLC25A29, of solute carrier family 25, encodes a mitochondrial transporter of basic amino acids. J Biol Chem 289:13374–13384. https://doi.org/10.1074/jbc.M114.547448
Punzi G, Porcelli V, Ruggiu M, Hossain MF, Menga A, Scarcia P, Castegna A, Gorgoglione R, Pierri CL, Laera L, Lasorsa FM, Paradies E, Pisano I, Marobbio CMT, Lamantea E, Ghezzi D, Tiranti V, Giannattasio S, Donati MA, Guerrini R, Palmieri L, Palmieri F, De Grassi A (2018) SLC25A10 biallelic mutations in intractable epileptic encephalopathy with complex I deficiency. Hum Mol Genet 27:499–504. https://doi.org/10.1093/hmg/ddx419
Sambrook J, Russell DW (2006) Separation of RNA according to size: electrophoresis of RNA through agarose gels containing formaldehyde. Cold Spring Harbor Protoc. https://doi.org/10.1101/pdb.prot4050
Scarcia P, Palmieri L, Agrimi G, Palmieri F, Rottensteiner H (2017) Three mitochondrial transporters of Saccharomyces cerevisiae are essential for ammonium fixation and lysine biosynthesis in synthetic minimal medium. Mol Genet Metab 122:54–60. https://doi.org/10.1016/j.ymgme.2017.07.004
Sherman F (1991) Getting started with yeast. Methods Enzymol 194:3–21. https://doi.org/10.1016/0076-6879(91)94004-v
Sickmann A, Reinders J, Wagner Y, Joppich C, Zahedi R, Meyer HE, Schönfisch B, Perschil I, Chacinska A, Guiard B, Rehling P, Pfanner N, Meisinger C (2003) The proteome of Saccharomyces cerevisiae mitochondria. Proc Natl Acad Sci USA 100:13207–13212. https://doi.org/10.1073/pnas.2135385100
Sikorski RS, Hieter P (1989) A system of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae. Genetics 122:19–27
Acknowledgements
This work was supported by Grants from the Ministero dell’Istruzione, dell’Università e della Ricerca (MIUR) and the Center of Excellence on Comparative Genomics (CEGBA).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scarcia, P., Agrimi, G., Germinario, L. et al. In Saccharomyces cerevisiae grown in synthetic minimal medium supplemented with non-fermentable carbon sources glutamate is synthesized within mitochondria. Rend. Fis. Acc. Lincei 29, 483–490 (2018). https://doi.org/10.1007/s12210-018-0687-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12210-018-0687-6