Abstract
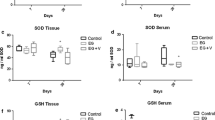
Hyperoxaluria and crystal deposition induce oxidative stress (OS) and renal epithelial cells injury, both mitochondria and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase are considered as the main sources of reactive oxygen species (ROS). Taurine is known to have antioxidant activity and shows renoprotective effect. We investigate the effect of taurine treatment on renal protection, and the putative source of ROS, in a rat model of calcium oxalate nephrolithiasis. Rats were administered with 2.5% (V/V) ethylene glycol + 2.5% (W/V) ammonium chloride (4 ml/day), with restriction on intake of drinking water (20 ml/day) for 4 weeks. Simultaneous treatment with taurine (2% W/W, mixed with the chow) was performed. At the end of the study, indexes of OS and renal injury were assessed. Renal tubular ultrastructure changes were analyzed under transmission electron microscopy. Crystal deposition in kidney was scored under light microscopy. Angiotensin II in kidney homogenates was determined by radioimmunoassay. Expression of NADPH oxidase subunits p47phox and Nox-4 mRNAs in kidney was evaluated by real time-polymerase chain reaction. The data showed that oxidative injury of the kidney occurred in nephrolithiasis-induced rats. Hyperplasia of mitochondria developed in renal tubular epithelium. The activities of superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) in mitochondria decreased and the mitochondrial membrane showed oxidative injury. Taurine treatment alleviated the oxidative injury of the kidney, improved SOD and GSH-Px activities, as well as the mitochondrial membrane injury, with lesser crystal depositions in the kidney. We could not detect statistical changes in the renal angiotensin II level, and the renal p47phox and Nox-4 mRNAs expression in those rats. The results suggest that mitochondria but not NADPH oxidase may account for the OS and taurine protected kidney from oxidative injury through mitochondrial-linked pathway in this rat model.








Similar content being viewed by others
References
Asplin JR (2002) Hyperoxaluric calcium nephrolithiasis. Endocrinol Metab Clin North Am 31:927–949. doi:10.1016/S0889-8529(02)00030-0
Khan SR (2006) Renal tubular damage/dysfunction: key to the formation of kidney stones. Urol Res 34:86–91. doi:10.1007/s00240-005-0016-2
Tsujihata M (2008) Mechanism of calcium oxalate renal stone formation and renal tubular cell injury. Int J Urol 15:115–120. doi:10.1111/j.1442-2042.2007.01953.x
Scheid C, Koul H, Hill WA, Luber-Narod J, Jonassen J, Honeyman T, Kennington L, Kohli R, Hodapp J, Ayvazian P, Menon M (1996) Oxalate toxicity in LLC-PK1 cells, a line of renal epithelial cells. J Urol 155:1112–1116. doi:10.1016/S0022-5347(01)66402-4
Khan SR (1995) calcium oxalate crystal interaction with renal tubular epithelium, mechanism of crystal adhesion and its impact on stone development. Urol Res 23:71–79. doi:10.1007/BF00307936
Khan SR (2004) Role of renal epithelial cells in the initiation of calcium oxalate stones. Nephron Exp Nephrol 98:e55–e60. doi:10.1159/000080257
Thamilselvan S, Byer KJ, Hackett RL, Khan SR (2000) Free radical scavengers, catalase and superoxide dismutase provide protection from oxalate-associated injury to LLC-PK1 and MDCK cells. J Urol 164:230–236. doi:10.1016/S0022-5347(05)67499-X
Thamilselvan S, Hackett RL, Khan SR (1997) Lipid peroxidation in ethylene glycol induced hyperoxaluria and calcium oxalate nephrolithiasis. J Urol 157:1059–1063. doi:10.1016/S0022-5347(01)65141-3
Rashed T, Menon M, Thamilselvan S (2004) Molecular mechanism of oxalate-induced free radical production and glutathione redox imbalance in renal epithelial cells: effect of antioxidants. Am J Nephrol 24:557–568. doi:10.1159/000082043
Thamilselvan S, Khan SR, Menon M (2003) Oxalate and calcium oxalate mediated free radical toxicity in renal epithelial cells: effect of antioxidants. Urol Res 31:3–9
Khand FD, Gordge MP, Robertson WG, Noronha-Dutra AA, Hothersall JS (2002) Mitochondrial superoxide production during oxalate mediated oxidative stress in renal epithelial cells. Free Radic Biol Med 32:1339–1350. doi:10.1016/S0891-5849(02)00846-8
Cao LC, Honeyman TW, Cooney R, Kennington L, Scheid CR, Jonassen JA (2004) Mitochondrial dysfunction is a primary event in renal cell oxalate toxicity. Kidney Int 66:1890–1900. doi:10.1111/j.1523-1755.2004.00963.x
Umekawa T, Byer K, Uemura H, Khan SR (2005) Diphenyleneiodium (DPI) reduces oxalate ion- and calcium oxalate monohydrate and brushite crystal-induced upregulation of MCP-1 in NRK 52E cells. Nephrol Dial Transplant 20:870–878. doi:10.1093/ndt/gfh750
Thamilselvan S, Menon M (2005) Vitamin E therapy prevents hyperoxaluria-induced calcium oxalate crystal deposition in the kidney by improving renal tissue antioxidant status. BJU Int 96:117–126. doi:10.1111/j.1464-410X.2005.05579.x
Selvam R, Ravichandran V (1993) Restoration of tissue antioxidants and prevention of renal stone deposition in vitamin B6 deficient rats fed with vitamin E or methionine. Indian J Exp Biol 31:882–887
Muthukumar A, Selvam R (1998) Role of glutathione on renal mitochondrial status in hyperoxaluria. Mol Cell Biochem 185:77–84. doi:10.1023/A:1006817319876
Itoh Y, Yasui T, Okada A, Tozawa K, Hayashi Y, Kohri K (2005) Preventive effects of green tea on renal stone formation and the role of oxidative stress in nephrolithiasis. J Urol 173:271–275
Khan SR (2005) Hyperoxaluria-induced oxidative stress and antioxidants for renal protection. Urol Res 33:349–357. doi:10.1007/s00240-005-0492-4
Selvam R (2002) Calcium oxalate stone disease: role of lipid peroxidation and antioxidants. Urol Res 30:35–47. doi:10.1007/s00240-001-0228-z
Tungsanga K, Sriboonlue P, Futrakul P, Yachantha C, Tosukhowong P (2005) Renal tubular cell damage and oxidative stress in renal stone patients and the effect of potassium citrate treatment. Urol Res 33:65–69. doi:10.1007/s00240-004-0444-4
Schwille PO, Manoharan M, Schmiedl A (2005) Is idiopathic recurrent calcium urolithiasis in males a cellular disease? Laboratory findings in plasma, urine and erythrocytes, emphasizing the absence and presence of stones, oxidative and mineral metabolism: an observational study. Clin Chem Lab Med 43:590–600. doi:10.1515/CCLM.2005.103
Erdem A, Gündoğan NU, Usubütün A, Kilinç K, Erdem SR, Kara A, Bozkurt A (2000) The protective effect of taurine against gentamicin-induced acute tubular necrosis in rats. Nephrol Dial Transplant 15:1175–1182. doi:10.1093/ndt/15.8.1175
Wang L, Zhang L, Yu Y, Wang Y, Niu N (2008) The protective effects of taurine against early renal injury in STZ-induced diabetic rats, correlated with inhibition of renal LOX-1-mediated ICAM-1 expression. Ren Fail 30(8):763–771. doi:10.1080/08860220802272563
Manna P, Sinha M, Sil PC (2008) Taurine plays a beneficial role against cadmium-induced oxidative renal dysfunction. Amino Acids [Epub ahead of print]
Sener G, Sehirli O, Ipçi Y, Cetinel S, Cikler E, Gedik N, Alican I (2005) Protective effects of taurine against nicotine-induced oxidative damage of rat urinary bladder and kidney. Pharmacology 74:37–44. doi:10.1159/000083245
Schaffer S, Azuma J, Takahashi K, Mozaffari M (2003) Why is taurine cytoprotective? Adv Exp Med Biol 526:307–321
Parvez S, Tabassum H, Banerjee BD, Raisuddin S (2008) Taurine prevents tamoxifen-induced mitochondrial oxidative damage in mice. Basic Clin Pharmacol Toxicol 102:382–387. doi:10.1111/j.1742-7843.2008.00208.x
Takahahsi K, Azuma M, Baba A, Schaffer S, Azuma J (1998) Taurine improves angiotensin II-induced hypertrophy of cultured neonatal rat heart cells. Adv Exp Med Biol 442:129–135
Umekawa T, Hatanaka YJ, Kurita T, Khan SR (2004) Effect of Angiotensin II receptor blockage on osteopontin expression and calcium oxalate crystal deposition in rat kidneys. J Am Soc Nephrol 15:635–644. doi:10.1097/01.ASN.0000113321.49771.2D
Toblli JE, Ferder L, Stella I, Angerosa M, Inserra F (2001) Protective role of enalapril for chronic tubulointerstitial lesions of hyperoxaluria. J Urol 166:275–280. doi:10.1016/S0022-5347(05)66144-7
Toblli JE, Ferder L, Stella I, De Cavanagh MVE, Angerosa M, Inserra F (2002) Effects of angiotensin II subtype 1 receptor blockade by losartan on tubulointerstitial lesions caused by hyperoxaluria. J Urol 168:1550–1555. doi:10.1016/S0022-5347(05)64519-3
Hanna IR, Taniyama Y, Szocs K, Rocic P, Griendling KK (2002) NAD(P)H oxidase derived reactive oxygen species as mediators of angiotensin II signaling. Antioxid Redox Signal 4:899–914. doi:10.1089/152308602762197443
Aihara K, Byer KJ, Khan SR (2003) Calcium phosphate-induced renal epithelial injury and stone formation: involvement of reactive oxygen species. Kidney Int 64:1283–1291. doi:10.1046/j.1523-1755.2003.00226.x
Gambaro G, Valente ML, Zanetti E, Della Barbera M, Del Prete D, D’Angelo A, Trevisan A (2006) Mild tubular damage induces calcium oxalate crystalluria in a model of subtle hyperoxaluria: evidence that a second hit is necessary for renal lithogenesis. J Am Soc Nephrol 17:2213–2219. doi:10.1681/ASN.2005121282
Meimaridou E, Lobos E, Hothersall JS (2006) Renal oxidative vulnerability due to changes in mitochondrial-glutathione and energy homeostasis in a rat model of calcium oxalate urolithiasis. Am J Physiol Renal Physiol 291:731–740. doi:10.1152/ajprenal.00024.2006
Huang H-S, Ma M-C, Chen C-F, Chen J (2003) Lipid peroxidation and its correlations with urinary levels of oxalate, citric acid, and osteopontin in patients with renal calcium oxalate stones. Urology 62:1123–1128. doi:10.1016/S0090-4295(03)00764-7
Veena CK, Josephine A, Preetha SP, Rajesh NG, Varalakshmi P (2008) Mitochondrial dysfunction in an animal model of hyperoxaluria: a prophylactic approach with fucoidan. Eur J Pharmacol 579(1–3):330–336
Babior BM, Lambeth JD, Nauseef W (2002) The neutrophil NADPH oxidase. Arch Biochem Biophys 397:342–344. doi:10.1006/abbi.2001.2642
Chabrashvili T, Tojo A, Onozato ML, Kitiyakara C, Quinn MT, Fujita T, Welch WJ, Wilcox CS (2002) Expression and cellular localization of classic NADPH oxidase subunits in the spontaneously hypertensive rat kidney. Hypertension 39:269–274. doi:10.1161/hy0202.103264
Shiose A, Kuroda J, Tsuruya K (2001) A novel superoxide producing NADPH oxidase in kidney. J Biol Chem 276:1417–1423. doi:10.1074/jbc.M007597200
Li JM, Shah AM (2003) ROS generation by nonphagocytic NADPH oxidase: potential relevance in diabetic nephropathy. J Am Soc Nephrol 14:S221–S226. doi:10.1097/01.ASN.0000077406.67663.E7
de Water R, Noordermeer C, van der Kwast TH, Nizze H, Boevé ER, Kok DJ, Schröder FH (1999) Calcium oxalate nephrolithiasis: effect of renal crystal deposition on the cellular composition of the renal interstitium. Am J Kidney Dis 33(4):761–771. doi:10.1016/S0272-6386(99)70231-3
Khan SR, Glenton P (1995) Deposition of calcium phosphate and calcium oxalate crystals in the kidneys. J Urol 153:811–817. doi:10.1016/S0022-5347(01)67728-0
Hansen SH, Andersen ML, Birkedal H, Cornett C, Wibrand F (2006) The important role of taurine in oxidative metabolism. Adv Exp Med Biol 583:129–135. doi:10.1007/978-0-387-33504-9_13
Li Y, Arnold JM, Pampillo M, Babwah AV, Peng T (2009) Taurine prevents cardiomyocyte death by inhibiting NADPH oxidase-mediated calpain activation. Free Radic Biol Med 46:51–61. doi:10.1016/j.freeradbiomed.2008.09.025
Acknowledgments
This study was supported by the Major Program of Guangxi Zhuang Autonomous Region Bureau of Health (No. 200729), the Guangxi Science and Technology Development Program (No. 0816004-4), and the Guangxi Postgraduate Innovation Program (No. 2008105981002D31).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, C.Y., Deng, Y.L. & Sun, B.H. Taurine protected kidney from oxidative injury through mitochondrial-linked pathway in a rat model of nephrolithiasis. Urol Res 37, 211–220 (2009). https://doi.org/10.1007/s00240-009-0197-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00240-009-0197-1