Abstract
Introduction
The purpose of this study was to examine the normal pituitary gland in male subjects with ultrashort echo time (TE) pulse sequences, describe its appearance and measure its signal intensity before and after contrast enhancement.
Methods
Eleven male volunteers (mean age 57.1 years; range 36–81 years) were examined with a fat-suppressed ultrashort TE (= 0.08 ms) pulse sequence. The studies were repeated after the administration of intravenous gadodiamide. The MR scans were examined for gland morphology and signal intensity before and after enhancement. Endocrinological evaluation included baseline pituitary function tests and a glucagon stimulatory test to assess pituitary cortisol and growth hormone reserve.
Results
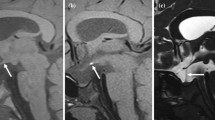
High signal intensity was observed in the anterior pituitary relative to the brain in nine of the 11 subjects. These regions involved the whole of the anterior pituitary in three subjects, were localised to one side in two examples and were seen inferiorly in three subjects. Signal intensities relative to the brain increased with age, with a peak around the sixth or seventh decade and decreasing thereafter. Overall, the pituitary function tests were considered to be within normal limits and did not correlate with pituitary gland signal intensity.
Conclusion
The anterior pituitary shows increased signal intensity in normal subjects when examined with T1-weighted ultrashort TE pulse sequences. The cause of this increased intensity is unknown, but fibrosis and iron deposition are possible candidates. The variation in signal intensity with age followed the temporal pattern of iron content observed at post mortem. No relationship with endocrine status was observed.








Similar content being viewed by others
References
Lum C, Kucharczyk W, Montanera WJ, Becker LE (2002) The sella turcica and parasellar region. In: Atlas SW (ed). Magnetic resonance imaging of the brain and spine. Lippincott, Williams and Wilkins, Philadelphia, pp 1283–1362
Sanders WP, Chundi VV (1998) Extra-axial tumors including pituitary and parasellar. In: Orrison WW (ed). Neuroimaging. Saunders, Philadelphia, pp 612–717
Bonneville F, Cattin F, Marsot-Dupuch K, Dormont D, Bonneville JF, Chiras J (2006) T1 signal hyperintensity in the sellar region: spectrum of findings. Radiographics 26:93–113
Rao VM, Vinitski S, Babaria A, Flanders A, Mishkin MM, Gonzalez C (1991) Enhanced resolution of pituitary fossa by three-dimensional fat-suppressed gradient-echo magnetic resonance: before and after gadolinium enhancement. J Neuroimaging 1:95–99
Stadnik T, Stevenaert A, Beckers A, Luypaert R, Buisseret T, Osteaux M (1990) Pituitary microadenomas: diagnosis with two-and three-dimensional MR imaging at 1.5 T before and after injection of gadolinium. Radiology 176:419–428
Girard N, Brue T, Chabert-Orsini V et al (1994) 3D-FT thin sections MRI of prolactin-secreting pituitary microadenomas. Neuroradiology 36:376–379
Stadnik T, D’Haens J, Luypaert R, Osteaux M (1994) The value of three-dimensional turbo-FLASH and spin-echo sequences in the detection of pituitary microadenomas following gadolinium administration. Neuroradiology 36:598–601
Bergin CJ, Pauly JM, Macovski A (1991) Lung parenchyma: projection reconstruction MR imaging. Radiology 179:777–781
Gatehouse PD, Bydder GM (2003) Magnetic resonance imaging of short T2 components in tissue. Clin Radiol 58:1–19
Mitchell ML, Byrne MJ, Sanchez Y, Sawin CT (1970) Detection of growth-hormone deficiency. N Engl J Med 282:539–541
Elster AD (1993) Sellar susceptibility artifacts: theory and implications. AJNR Am J Neuroradiol 14:129–136
Greenberg SR (1975) The pathogenesis of hypophyseal fibrosis in aging: its relationship to tissue iron deposition. J Gerontol 30:531–538
Sano T, Kovacs KT, Scheithauer BW, Young WF Jr (1993) Aging and the human pituitary gland. Mayo Clin Proc 68:971–977
Christoforidis A, Haritandi A, Perifanis V, Tsatra I, Athanassiou-Metaxa M, Dimitriadis AS (2007) MRI for the determination of pituitary iron overload in children and young adults with beta-thalassaemia major. Eur J Radiol 62:138–142
Argyropoulou MI, Metafratzi Z, Kiortsis DN, Bitsis S, Tsatsoulis A, Efremidis S (2000) T2 relaxation rate as an index of pituitary iron overload in patients with beta-thalassemia major. AJR Am J Roentgenol 175:1567–1569
Tien RD, Kucharczyk J, Bessette J, Middleton M (1992) MR imaging of the pituitary gland in infants and children: changes in size, shape, and MR signal with growth and development. AJR Am J Roentgenol 158:1151–1154
Argyropoulou MI, Xydis V, Kiortsis DN et al (2004) Pituitary gland signal in pre-term infants during the first year of life: an MRI study. Neuroradiology 46:1031–1035
Holder CA, Elster AD (1997) Magnetization transfer imaging of the pituitary: further insights into the nature of the posterior “bright spot”. J Comput Assist Tomogr 21:171–174
Argyropoulou MI, Kiortsis DN, Metafratzi Z, Efremidis SC (2001) Magnetisation transfer imaging of the normal adenohypophysis: the effect of sex and age. Neuroradiology 43:305–308
Sato N, Tanaka S, Tateno M, Ohya N, Takata K, Endo K (1995) Origin of posterior pituitary high intensity on T1-weighted magnetic resonance imaging. Immunohistochemical, electron microscopic, and magnetic resonance studies of posterior pituitary lobe of dehydrated rabbits. Invest Radiol 30:567–571
Caruso RD, Rosenbaum AE, Sherry RG et al (1998) Pituitary gland. Variable signal intensities on MRI. A pictorial essay. Clin Imaging 22:327–332
Yousem DM, Arrington JA, Kumar AJ, Bryan RN (1990) Bright lesions on sellar/parasellar T1-weighted scans. Clin Imaging 14:99–105
Bonneville F, Cattin F, Barrali E, Lucas X, Narboux Y, Bonneville J (2001) Increased T1 signal of the residual normal anterior pituitary gland following medical treatment of pituitary prolactinoma. J Radiol 82:501–505
Landolt AM, Osterwalder V (1984) Perivascular fibrosis in prolactinomas: is it increased by bromocriptine? J Clin Endocrinol Metab 58:1179–1183
Nishioka H, Ito H, Haraoka J, Hirano A (2001) Histological changes in the hypofunctional pituitary gland following conventional radiotherapy for adenoma. Histopathology 38:561–566
Anniko M, Wersall J (1982) Morphological effects in pituitary tumours following radiotherapy. Virchows Arch A Pathol Anat Histol 395:45–58
Ishii K, Ikeda H, Takahashi S, Matsumoto K, Ishibashi T, Tazawa S (1996) MR imaging of pituitary adenomas with sphenoid sinus invasion: characteristic MR findings indicating fibrosis. Radiat Med 14:173–178
Diniz RL, Reimund JM, Duclos B, Reis M Jr, Baumann R, Dietemann JL (1998) Spontaneous hyperintensity of the anterior pituitary gland in MRI T1-weighted images related to manganese deposits in a patient undergoing prolonged parenteral nutrition. J Radiol 79:345–347
Dietemann JL, Reimund JM, Diniz RL et al (1998) High signal in the adenohypophysis on T1-weighted images presumably due to manganese deposits in patients on long-term parenteral nutrition. Neuroradiology 40:793–796
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Portman, O., Flemming, S., Cox, J.P.D. et al. Magnetic resonance imaging of the normal pituitary gland using ultrashort TE (UTE) pulse sequences (REV 1.0). Neuroradiology 50, 213–220 (2008). https://doi.org/10.1007/s00234-007-0329-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-007-0329-7