Abstract
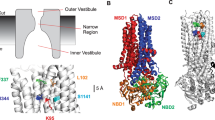
The cystic fibrosis transmembrane conductance regulator (CFTR) Cl− channel pore is thought to contain multiple binding sites for permeant and impermeant anions. Here, we investigate the effects of mutation of different positively charged residues in the pore on current inhibition by impermeant Pt(NO2) 2−4 and suramin anions. We show that mutations that remove positive charges (K95, R303) influence interactions with intracellular, but not extracellular, Pt(NO2) 2−4 ions, consistent with these residues being situated within the pore inner vestibule. In contrast, mutation of R334, supposedly located in the outer vestibule of the pore, affects block by both extracellular and intracellular Pt(NO2) 2−4 . Inhibition by extracellular Pt(NO2) 2−4 requires a positive charge at position 334, consistent with a direct electrostatic interaction resulting in either open channel block or surface charge screening. In contrast, inhibition by intracellular Pt(NO2) 2−4 is weakened in all R334-mutant forms of the channel studied, inconsistent with a direct interaction. Furthermore, mutation of R334 had similar effects on block by intracellular suramin, a large organic molecule that is apparently unable to enter deeply into the channel pore. Mutation of R334 altered interactions between intracellular Pt(NO2) 2−4 and extracellular Cl− but not those between intracellular Pt(NO2) 2−4 and extracellular Pt(NO2) 2−4 . We propose that while the positive charge of R334 interacts directly with extracellular anions, mutation of this residue also alters interactions with intracellular anions by an indirect mechanism, due to mutation-induced conformational changes in the protein that are propagated some distance from the site of the mutation in the outer mouth of the pore.











Similar content being viewed by others
References
Cheung M, Akabas MH (1996) Identification of cystic fibrosis transmembrane conductance regulator channel-lining residues in and flanking the M6 membrane-spanning segment. Biophys J 70:2688–2695
Fatehi M, St Aubin CN, Linsdell P (2007) On the origin of asymmetric interactions between permeant anions and the CFTR chloride channel pore. Biophys J 92:1241–1253
Ge N, Linsdell P (2006) Interactions between impermeant blocking ions in the cystic fibrosis transmembrane conductance regulator chloride channel pore: evidence for anion-induced conformational changes. J Membr Biol 210:31–42
Ge N, Muise CN, Gong X, Linsdell P (2004) Direct comparison of the functional roles played by different transmembrane regions in the cystic fibrosis transmembrane conductance regulator chloride channel pore. J Biol Chem 279:55283–55289
Gong X, Burbridge SM, Cowley EA, Linsdell P (2002a) Molecular determinants of Au(CN)2− binding and permeability within the cystic fibrosis transmembrane conductance regulator Cl– channel pore. J Physiol 540:39–47
Gong X, Burbridge SM, Lewis AC, Wong PYD, Linsdell P (2002b) Mechanism of lonidamine inhibition of the CFTR chloride channel. Br J Pharmacol 137:928–936
Gong X, Linsdell P (2003a) Molecular determinants and role of an anion binding site in the external mouth of the CFTR chloride channel pore. J Physiol 549:387–397
Gong X, Linsdell P (2003b) Mutation-induced blocker permeability and multiion block of the CFTR chloride channel pore. J Gen Physiol 122:673–687
Gong X, Linsdell P (2004) Maximization of the rate of chloride conduction in the CFTR channel pore by ion-ion interactions. Arch Biochem Biophys 426:78–82
Green WN, Andersen OS (1991) Surface charges and ion channel function. Annu Rev Physiol 53:341–359
Kidd JF, Kogan I, Bear CE (2004) Molecular basis for the chloride channel activity of cystic fibrosis transmembrane conductance regulator and the consequences of disease-causing mutations. Curr Top Dev Biol 60:215–249
Lewis HA, Buchanan SG, Burley SK, Conners K, Dickey M, Dorwart M, Fowler R, Gao X, Guggino WB, Hendrickson WA, Hunt JF, Kearins MC, Lorimer D, Maloney PC, Post KW, Rajashankar KR, Rutter ME, Sauder JM, Shriver S, Thibodeau PH, Thomas PJ, Zhang M, Zhao X, Emtage S (2004) Structure of nucleotide-binding domain 1 of the cystic fibrosis transmembrane conductance regulator. EMBO J 23:282–293
Linsdell P (2005) Location of a common inhibitor binding site in the cytoplasmic vestibule of the cystic fibrosis transmembrane conductance regulator chloride channel pore. J Biol Chem 280:8945–8950
Linsdell P (2006) Mechanism of chloride permeation in the cystic fibrosis transmembrane conductance regulator chloride channel. Exp Physiol 91:123–129
Linsdell P, Hanrahan JW (1996) Disulphonic stilbene block of cystic fibrosis transmembrane conductance regulator Cl− channels expressed in a mammalian cell line and its regulation by a critical pore residue. J Physiol 496:687–693
Linsdell P, Hanrahan JW (1998) Adenosine triphosphate-dependent asymmetry of anion permeation in the cystic fibrosis transmembrane conductance regulator chloride channel. J Gen Physiol 111:601–614
Liu X, Smith SS, Dawson DC (2003) CFTR: what’s it like inside the pore? J Exp Zool 300A:69–75
Rosenberg MF, Kamis AB, Aleksandrov LA, Ford RC, Riordan JR (2004) Purification and crystallization of the cystic fibrosis transmembrane conductance regulator (CFTR). J Biol Chem 279:39051–39057
Sheppard DN, Welsh MJ (1999) Structure and function of the CFTR chloride channel. Physiol Rev 79:S23–S45
Smith SS, Liu X, Zhang Z-R, Sun F, Kriewall TE, McCarty NA, Dawson DC (2001) CFTR: covalent and noncovalent modification suggests a role for fixed charges in anion conduction. J Gen Physiol 118:407–431
St Aubin CN, Linsdell P (2006) Positive charges at the intracellular mouth of the pore regulate anion conduction in the CFTR chloride channel. J Gen Physiol 128:535–545
St Aubin CN, Zhou J-J, Linsdell P (2007) Identification of a second blocker binding site at the cytoplasmic mouth of the CFTR chloride channel pore. Mol Pharmacol 71:1360–1368
Woodull AM (1973) Ionic blockage of sodium channels in nerve. J Gen Physiol 61:687–708
Zhang Z-R, Song B, McCarty NA (2005) State-dependent chemical reactivity of an engineered cysteine reveals conformational changes in the outer vestibule of the cystic fibrosis transmembrane conductance regulator. J Biol Chem 280:41997–42003
Acknowledgements
This work was supported by the Canadian Institutes of Health Research. We thank Elizabeth VandenBerg for technical assistance and Dr. Elizabeth Cowley for comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, JJ., Fatehi, M. & Linsdell, P. Direct and Indirect Effects of Mutations at the Outer Mouth of the Cystic Fibrosis Transmembrane Conductance Regulator Chloride Channel Pore. J Membrane Biol 216, 129–142 (2007). https://doi.org/10.1007/s00232-007-9056-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00232-007-9056-6