Abstract
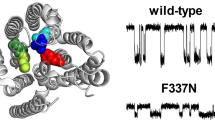
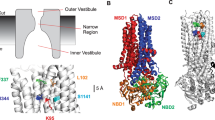
Our molecular understanding of the cystic fibrosis transmembrane conductance regulator (CFTR)—the chloride channel that is mutated in cystic fibrosis—has been greatly enhanced by a number of recent atomic-level structures of the protein in different conformations. One surprising aspect of these structures was the finding that the eighth of CFTR’s 12 membrane-spanning segments (TM8) appeared close to the channel pore. Although functional evidence supports a role for other TMs in forming the pore, such a role for TM8 has not previously been reported. Here, we use patch-clamp recording to investigate the functional role of TM8. Using substituted cysteine accessibility mutagenesis, we find that three amino acid side-chains in TM8 (Y913, Y914, and Y917) are exposed to the extracellular, but not the intracellular, solution. Cysteine cross-linking experiments suggest that Y914 and Y917 are in close proximity to L102 (TM1) and F337 (TM6), respectively, suggesting that TM8 contributes to the narrow selectivity filter region of the pore. Different amino acid substitutions suggest that Y914, and to a lesser extent Y917, play important roles in controlling anion flux through the open channel. Furthermore, substitutions that reduce side-chain volume at Y917 severely affect channel gating, resulting in a channel with an extremely unstable open state. Our results suggest that pore-lining TM8 is among the most important TMs controlling the permeation phenotype of the CFTR channel, and also that movement of TM8 may be critically involved in channel gating.







Similar content being viewed by others
Abbreviations
- BHK:
-
Baby hamster kidney
- CF:
-
Cystic fibrosis
- CFTR:
-
CF transmembrane conductance regulator
- CHO:
-
Chinese hamster ovary
- Cryo-EM:
-
Electron cryo-microscopy
- CuPhe:
-
Copper(II)-o-phenanthroline
- DTT:
-
Dithiothreitol
- MSD:
-
Membrane-spanning domain
- MTS:
-
Methanethiosulfonate
- MTSES:
-
[2-Sulfonatoethyl] MTS
- MTSET:
-
[2-(Trimethylammonium)ethyl] MTS
- NBD:
-
Nucleotide-binding domain
- PKA:
-
Protein kinase A
- SCAM:
-
Substituted cysteine accessibility mutagenesis
- TM:
-
Transmembrane helix
References
Wang Y, Wrennall JA, Cai Z, Li H, Sheppard DN (2014) Understanding how cystic fibrosis mutations disrupt CFTR function: from single molecules to animal models. Int J Biochem Cell Biol 52:47–57
Csanády L, Vergani P, Gadsby DC (2019) Structure, gating, and regulation of the CFTR anion channel. Physiol Rev 99:707–738
Moran O (2017) The gating of the CFTR channel. Cell Mol Life Sci 74:85–92
Hwang T-C, Yeh J-T, Zhang J, Yu Y-C, Yeh H-I, Destefano S (2018) Structural mechanisms of CFTR function and dysfunction. J Gen Physiol 150:539–570
Zhang Z, Chen J (2016) Atomic structure of the cystic fibrosis transmembrane conductance regulator. Cell 167:1586–1597
Liu F, Zhang Z, Csanády L, Gadsby DC, Chen J (2017) Molecular structure of the human CFTR ion channel. Cell 169:85–95
Zhang Z, Liu F, Chen J (2017) Conformational changes of CFTR upon phosphorylation and ATP binding. Cell 170:483–491
Zhang Z, Liu F, Chen J (2018) Molecular structure of the ATP-bound, phosphorylated human CFTR. Proc Natl Acad Sci USA 115:12757–12762
Callebaut I, Hoffmann B, Lehn P, Mornon J-P (2017) Molecular modelling and molecular dynamics of CFTR. Cell Mol Life Sci 74:3–22
Linsdell P (2017) Architecture and functional properties of the CFTR channel pore. Cell Mol Life Sci 74:67–83
Linsdell P, Evagelidis A, Hanrahan JW (2000) Molecular determinants of anion selectivity in the cystic fibrosis transmembrane conductance regulator chloride channel pore. Biophys J 78:2973–2982
McCarty NA, Zhang Z-R (2001) Identification of a region of strong discrimination in the pore of CFTR. Am J Physiol 281:L852–L867
Linsdell P (2016) Anion conductance selectivity mechanism of the CFTR chloride channel. Biochim Biophys Acta 1858:740–747
Negoda A, El Hiani Y, Cowley EA, Linsdell P (2017) Contribution of a leucine residue in the first transmembrane segment to the selectivity filter region in the CFTR chloride channel. Biochim Biophys Acta 1859:1049–1058
Corradi V, Vergani P, Tieleman DP (2015) Cystic fibrosis transmembrane conductance regulator (CFTR): closed and open state channel models. J Biol Chem 290:22891–22906
Gao X, Hwang T-C (2015) Localizing a gate in CFTR. Proc Natl Acad Sci USA 112:2461–2466
Wei S, Roessler BC, Icyuz M, Chauvet S, Tao B, Hartman JL, Kirk KL (2016) Long-range coupling between the extracellular gates and the intracellular ATP binding domains of multidrug resistance protein pumps and cystic fibrosis transmembrane conductance regulator channels. FASEB J 30:1247–1262
Fay JF, Aleksandrov LA, Jensen T, Cui L, Kousouros JN, He L, Aleksandrov AA, Gingerich D, Riordan JR, Chen J (2018) Cryo-EM visualization of an active high open probability CFTR anion channel. Biochemistry 57:6234–6246
Corradi V, Gu R-X, Vergani P, Tieleman DP (2018) Structure of transmembrane helix 8 and possible membrane defects in CFTR. Biophys J 114:1751–1754
Zhou J-J, Li M-S, Qi J, Linsdell P (2010) Regulation of conductance by the number of fixed positive charges in the intracellular vestibule of the CFTR chloride channel pore. J Gen Physiol 135:229–245
Mense M, Vergani P, White DM, Altberg G, Nairn AC, Gadsby DC (2006) In vivo phosphorylation of CFTR promotes formation of a nucleotide-binding domain heterodimer. EMBO J 25:4728–4739
Li M-S, Demsey AFA, Qi J, Linsdell P (2009) Cysteine-independent inhibition of the CFTR chloride channel by the cysteine-reactive reagent sodium (2-sulphonatoethyl) methanethiosulphonate. Br J Pharmacol 157:1065–1071
Negoda A, Cowley EA, El Hiani Y, Linsdell P (2018) Conformational change of the extracellular parts of the CFTR protein during channel gating. Cell Mol Life Sci 75:3027–3038
Broadbent SD, Wang W, Linsdell P (2014) Interaction between two extracellular loops influences the activity of the cystic fibrosis transmembrane conductance regulator chloride channel. Biochem Cell Biol 92:390–396
Wang W, El Hiani Y, Rubaiy HN, Linsdell P (2014) Relative contribution of different transmembrane segments to the CFTR chloride channel pore. Pflügers Arch 466:477–490
El Hiani Y, Linsdell P (2015) Functional architecture of the cytoplasmic entrance to the cystic fibrosis transmembrane conductance regulator chloride channel pore. J Biol Chem 290:15855–15865
El Hiani Y, Negoda A, Linsdell P (2016) Cytoplasmic pathway followed by chloride ions to enter the CFTR channel pore. Cell Mol Life Sci 73:1917–1925
Linsdell P, Hanrahan JW (1998) Adenosine triphosphate-dependent asymmetry of anion permeation in the cystic fibrosis transmembrane conductance regulator chloride channel. J Gen Physiol 111:601–614
Smith SS, Steinle ED, Meyerhoff ME, Dawson DC (1999) Cystic fibrosis transmembrane conductance regulator. Physical basis for lyotropic anion selectivity patterns. J Gen Physiol 114:799–818
Gong X, Burbridge SM, Cowley EA, Linsdell P (2002) Molecular determinants of Au(CN) −2 binding and permeability within the cystic fibrosis transmembrane conductance regulator Cl− channel pore. J Physiol 540:39–47
Ge N, Muise CN, Gong X, Linsdell P (2004) Direct comparison of the functional roles played by different transmembrane regions in the cystic fibrosis transmembrane conductance regulator chloride channel pore. J Biol Chem 279:55283–55289
Fatehi M, Linsdell P (2009) Novel residues lining the CFTR chloride channel pore identified by functional modification of introduced cysteines. J Membr Biol 228:151–164
Zhou J-J, Fatehi M, Linsdell P (2008) Identification of positive charges situated at the outer mouth of the CFTR chloride channel pore. Pflügers Arch 457:351–360
Wang W, El Hiani Y, Linsdell P (2011) Alignment of transmembrane regions in the cystic fibrosis transmembrane conductance regulator chloride channel pore. J Gen Physiol 138:165–178
Gao X, Bai Y, Hwang T-C (2013) Cysteine scanning of CFTR’s first transmembrane segment reveals its plausible roles in gating and permeation. Biophys J 104:786–797
Beck EJ, Yang Y, Yaemsiri S, Raghuram V (2008) Conformational changes in a pore-lining helix coupled to cystic fibrosis transmembrane conductance regulator channel gating. J Biol Chem 283:4957–4966
Bai Y, Li M, Hwang T-C (2010) Dual roles of the sixth transmembrane segment of the CFTR chloride channel in gating and permeation. J Gen Physiol 136:293–309
El Hiani Y, Linsdell P (2010) Changes in accessibility of cytoplasmic substances to the pore associated with activation of the cystic fibrosis transmembrane conductance regulator chloride channel. J Biol Chem 285:32126–32140
Norimatsu Y, Ivetac A, Alexander C, Kirkham J, O’Donnell N, Dawson DC, Sansom MSP (2012) Cystic fibrosis transmembrane conductance regulator: a molecular model defines the architecture of the anion conduction path and locates a “bottleneck” in the pore. Biochemistry 51:2199–2212
Bai Y, Li M, Hwang T-C (2011) Structural basis for the channel function of a degraded ABC transporter, CFTR (ABCC7). J Gen Physiol 138:495–507
Qian F, El Hiani Y, Linsdell P (2011) Functional arrangement of the 12th transmembrane region in the CFTR chloride channel based on functional investigation of a cysteine-less variant. Pflügers Arch 462:559–571
Zhang J, Hwang T-C (2015) The fifth transmembrane segment of cystic fibrosis transmembrane conductance regulator contributes to its anion permeation pathway. Biochemistry 54:3839–3850
Holstead RG, Li M-S, Linsdell P (2011) Functional differences in pore properties between wild-type and cysteine-less forms of the CFTR chloride channel. J Membr Biol 243:15–23
Linsdell P, Zheng S-X, Hanrahan JW (1998) Non-pore lining amino acid side chains influence anion selectivity of the human CFTR Cl− channel expressed in mammalian cell lines. J Physiol 512:1–16
McDonough S, Davidson N, Lester HA, McCarty NA (1994) Novel pore-lining residues in CFTR that govern permeation and open-channel block. Neuron 13:623–634
Gupta J, Linsdell P (2003) Extent of the selectivity filter conferred by the sixth transmembrane region in the CFTR chloride channel pore. Mol Membr Biol 20:45–52
Linsdell P (2015) Metal bridges to probe membrane ion channel structure and function. Biomol Concepts 6:191–203
Acknowledgements
We would like to thank Christina Irving for technical assistance. This work was supported by Cystic Fibrosis Canada.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Negoda, A., Hogan, M.S., Cowley, E.A. et al. Contribution of the eighth transmembrane segment to the function of the CFTR chloride channel pore. Cell. Mol. Life Sci. 76, 2411–2423 (2019). https://doi.org/10.1007/s00018-019-03043-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-019-03043-2