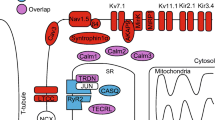
Abstract
Calcium (Ca2+) ions are a key second messenger involved in the rhythmic excitation and contraction of cardiomyocytes throughout the heart. Proper function of Ca2+-handling proteins is required for healthy cardiac function, whereas disruption in any of these can cause cardiac arrhythmias. This comprehensive review provides a broad overview of the roles of Ca2+-handling proteins and their regulators in healthy cardiac function and the mechanisms by which mutations in these proteins contribute to inherited arrhythmias. Major Ca2+ channels and Ca2+-sensitive regulatory proteins involved in cardiac excitation–contraction coupling are discussed, with special emphasis on the function of the RyR2 macromolecular complex. Inherited arrhythmia disorders including catecholaminergic polymorphic ventricular tachycardia, long QT syndrome, Brugada syndrome, short QT syndrome, and arrhythmogenic right-ventricular cardiomyopathy are discussed with particular emphasis on subtypes caused by mutations in Ca2+-handling proteins.





Similar content being viewed by others
Availability of data and materials
All the data are available in the text, tables, and figures of the review.
Code availability
Not applicable.
Abbreviations
- AF:
-
Atrial fibrillation
- APD:
-
Action potential duration
- Ca2+ :
-
Calcium ion
- CRU:
-
Calcium release unit
- CaMKII:
-
Calcium/calmodulin-dependent protein kinase II
- CICR:
-
Calcium-induced calcium release
- CPVT:
-
Catecholaminergic polymorphic ventricular tachycardia
- DAD:
-
Delayed after-depolarization
- EAD:
-
Early after-depolarization
- ECG:
-
Electrocardiogram
- EC:
-
Excitation–contraction
- FKBP 12.6:
-
FK-506-binding protein 12.6
- K+ :
-
Potassium ion
- LQTS:
-
Long QT syndrome
- LTCC:
-
Voltage-gated L-type calcium channel
- Na+ :
-
Sodium ion
- NCX:
-
Sodium-calcium exchanger
- PKA:
-
Protein kinase A
- PLN:
-
Phospholamban
- PP:
-
Protein phosphatase
- RyR2:
-
Ryanodine receptor type-2
- SERCA2a:
-
Sarco/endoplasmic reticulum Ca2+-ATPase-2a
- SPEG:
-
Striated preferentially expressed protein kinase
- SR:
-
Sarcoplasmic reticulum
- SOICR:
-
Store overload-induced calcium release
- VT:
-
Ventricular tachycardia
References
Algra A, Tijssen JG, Roelandt JR, Pool J, Lubsen J (1993) QT interval variables from 24 hour electrocardiography and the two year risk of sudden death. Br Heart J 70(1):43–48. https://doi.org/10.1136/hrt.70.1.43
Alsina KM, Hulsurkar M, Brandenburg S et al (2019) Loss of protein phosphatase 1 regulatory subunit PPP1R3A promotes atrial fibrillation. Circulation 140(8):681–693. https://doi.org/10.1161/CIRCULATIONAHA.119.039642
Altmann HM, Tester DJ, Will ML et al (2015) Homozygous/compound heterozygous triadin mutations associated with autosomal-recessive long-QT syndrome and pediatric sudden cardiac arrest: elucidation of the triadin knockout syndrome. Circulation 131(23):2051–2060. https://doi.org/10.1161/CIRCULATIONAHA.115.015397
Antzelevitch C, Pollevick GD, Cordeiro JM et al (2007) Loss-of-function mutations in the cardiac calcium channel underlie a new clinical entity characterized by ST-segment elevation, short QT intervals, and sudden cardiac death. Circulation 115(4):442–449. https://doi.org/10.1161/CIRCULATIONAHA.106.668392
Arvanitis DA, Vafiadaki E, Fan GC et al (2007) Histidine-rich Ca-binding protein interacts with sarcoplasmic reticulum Ca-ATPase. Am J Physiol Heart Circ Physiol 293(3):H1581–H1589. https://doi.org/10.1152/ajpheart.00278.2007
Asahi M, Kurzydlowski K, Tada M, MacLennan DH (2002) Sarcolipin inhibits polymerization of phospholamban to induce superinhibition of sarco(endo)plasmic reticulum Ca2+-ATPases (SERCAs). J Biol Chem 277(30):26725–26728. https://doi.org/10.1074/jbc.C200269200
Babu GJ, Bhupathy P, Timofeyev V et al (2007) Ablation of sarcolipin enhances sarcoplasmic reticulum calcium transport and atrial contractility. Proc Natl Acad Sci U S A 104(45):17867–17872. https://doi.org/10.1073/pnas.0707722104
Baddeley D, Jayasinghe ID, Lam L, Rossberger S, Cannell MB, Soeller C (2009) Optical single-channel resolution imaging of the ryanodine receptor distribution in rat cardiac myocytes. Proc Natl Acad Sci U S A 106(52):22275–22280. https://doi.org/10.1073/pnas.0908971106
Baig SM, Koschak A, Lieb A et al (2011) Loss of Ca(v)1.3 (CACNA1D) function in a human channelopathy with bradycardia and congenital deafness. Nat Neurosci 14(1):77–84. https://doi.org/10.1038/nn.2694
Balijepalli RC, Foell JD, Hall DD, Hell JW, Kamp TJ (2006) Localization of cardiac L-type Ca(2+) channels to a caveolar macromolecular signaling complex is required for beta(2)-adrenergic regulation. Proc Natl Acad Sci U S A 103(19):7500–7505. https://doi.org/10.1073/pnas.0503465103
Bandyopadhyay A, Shin DW, Ahn JO, Kim DH (2000) Calcineurin regulates ryanodine receptor/Ca(2+)-release channels in rat heart. Biochem J 352(Pt 1):61–70
Bassani JW, Yuan W, Bers DM (1995) Fractional SR Ca release is regulated by trigger Ca and SR Ca content in cardiac myocytes. Am J Physiol 268(5 Pt 1):C1313–C1319. https://doi.org/10.1152/ajpcell.1995.268.5.C1313
Baudot M, Torre E, Bidaud I et al (2020) Concomitant genetic ablation of L-type Cav1.3 (α1D) and T-type Ca v 3.1 (α1G) Ca2+ channels disrupts heart automaticity. Sci Rep 10(1):18906. https://doi.org/10.1038/s41598-020-76049-7
Bauer R, Timothy KW, Golden A (2021) Update on the molecular genetics of timothy syndrome. Front Pediatr 9:668546. https://doi.org/10.3389/fped.2021.668546
Beavers DL, Wang W, Ather S et al (2013) Mutation E169K in junctophilin-2 causes atrial fibrillation due to impaired RyR2 stabilization. J Am Coll Cardiol 62(21):2010–2019. https://doi.org/10.1016/j.jacc.2013.06.052
Beca S, Helli PB, Simpson JA et al (2011) Phosphodiesterase 4D regulates baseline sarcoplasmic reticulum Ca2+ release and cardiac contractility, independently of L-type Ca2+ current. Circ Res 109(9):1024–1030. https://doi.org/10.1161/CIRCRESAHA.111.250464
Benitah JP, Alvarez JL, Gomez AM (2010) L-type Ca(2+) current in ventricular cardiomyocytes. J Mol Cell Cardiol 48(1):26–36. https://doi.org/10.1016/j.yjmcc.2009.07.026
Bennett V, Baines AJ (2001) Spectrin and ankyrin-based pathways: metazoan inventions for integrating cells into tissues. Physiol Rev 81(3):1353–1392. https://doi.org/10.1152/physrev.2001.81.3.1353
Berlin JR, Bassani JW, Bers DM (1994) Intrinsic cytosolic calcium buffering properties of single rat cardiac myocytes. Biophys J 67(4):1775–1787. https://doi.org/10.1016/S0006-3495(94)80652-6
Bers DM (2002) Cardiac excitation-contraction coupling. Nature 415(6868):198–205. https://doi.org/10.1038/415198a
Bers DM, Bassani JW, Bassani RA (1993) Competition and redistribution among calcium transport systems in rabbit cardiac myocytes. Cardiovasc Res 27(10):1772–1777. https://doi.org/10.1093/cvr/27.10.1772
Boczek NJ, Best JM, Tester DJ et al (2013) Exome sequencing and systems biology converge to identify novel mutations in the L-type calcium channel, CACNA1C, linked to autosomal dominant long QT syndrome. Circ Cardiovasc Genet 6(3):279–289. https://doi.org/10.1161/CIRCGENETICS.113.000138
Boczek NJ, Ye D, Jin F et al (2015) Identification and functional characterization of a novel CACNA1C-mediated cardiac disorder characterized by prolonged qt intervals with hypertrophic cardiomyopathy, congenital heart defects, and sudden cardiac death. Circ Arrhythm Electrophysiol 8(5):1122–1132. https://doi.org/10.1161/CIRCEP.115.002745
Boczek NJ, Gomez-Hurtado N, Ye D et al (2016) Spectrum and prevalence of CALM1-, CALM2-, and CALM3-encoded calmodulin variants in long QT syndrome and functional characterization of a novel long QT syndrome-associated calmodulin missense variant, E141G. Circ Cardiovasc Genet 9(2):136–146. https://doi.org/10.1161/CIRCGENETICS.115.001323
Bodi I, Mikala G, Koch SE, Akhter SA, Schwartz A (2005) The L-type calcium channel in the heart: the beat goes on. J Clin Invest 115(12):3306–3317. https://doi.org/10.1172/JCI27167
Bottinelli R, Coviello DA, Redwood CS et al (1998) A mutant tropomyosin that causes hypertrophic cardiomyopathy is expressed in vivo and associated with an increased calcium sensitivity. Circ Res 82(1):106–115. https://doi.org/10.1161/01.res.82.1.106
Brandenburg S, Kohl T, Williams GS et al (2016) Axial tubule junctions control rapid calcium signaling in atria. J Clin Invest 126(10):3999–4015. https://doi.org/10.1172/JCI88241
Brautigan DL, Shenolikar S (2018) Protein serine/threonine phosphatases: keys to unlocking regulators and substrates. Annu Rev Biochem 87:921–964. https://doi.org/10.1146/annurev-biochem-062917-012332
Brugada P, Brugada J (1992) Right bundle branch block, persistent ST segment elevation and sudden cardiac death: a distinct clinical and electrocardiographic syndrome. A multicenter report. J Am Coll Cardiol 20(6):1391–1396. https://doi.org/10.1016/0735-1097(92)90253-j
Brugada R, Hong K, Dumaine R et al (2004) Sudden death associated with short-QT syndrome linked to mutations in HERG. Circulation 109(1):30–35. https://doi.org/10.1161/01.CIR.0000109482.92774.3A
Brunner F, Andrew P, Wölkart G, Zechner R, Mayer B (2001) Myocardial contractile function and heart rate in mice with myocyte-specific overexpression of endothelial nitric oxide synthase. Circulation 104(25):3097–3102. https://doi.org/10.1161/hc5001.101966
Bryant S, Kimura TE, Kong CH et al (2014) Stimulation of ICa by basal PKA activity is facilitated by caveolin-3 in cardiac ventricular myocytes. J Mol Cell Cardiol 68:47–55. https://doi.org/10.1016/j.yjmcc.2013.12.026
Burashnikov E, Pfeiffer R, Barajas-Martinez H et al (2010) Mutations in the cardiac L-type calcium channel associated with inherited J-wave syndromes and sudden cardiac death. Heart Rhythm 7(12):1872–1882. https://doi.org/10.1016/j.hrthm.2010.08.026
Busquet P, Nguyen NK, Schmid E et al (2010) CaV1.3 L-type Ca2+ channels modulate depression-like behaviour in mice independent of deaf phenotype. Int J Neuropsychopharmacol 13(4):499–513. https://doi.org/10.1017/S1461145709990368
Camors E, Mohler PJ, Bers DM, Despa S (2012) Ankyrin-B reduction enhances Ca spark-mediated SR Ca release promoting cardiac myocyte arrhythmic activity. J Mol Cell Cardiol 52(6):1240–1248. https://doi.org/10.1016/j.yjmcc.2012.02.010
Campbell HM, Quick AP, Abu-Taha I et al (2020) Loss of SPEG inhibitory phosphorylation of ryanodine receptor type-2 promotes atrial fibrillation. Circulation 142(12):1159–1172. https://doi.org/10.1161/CIRCULATIONAHA.120.045791
Catterall WA (2000) Structure and regulation of voltage-gated Ca2+ channels. Annu Rev Cell Dev Biol 16:521–555. https://doi.org/10.1146/annurev.cellbio.16.1.521
Chelu MG, Sarma S, Sood S et al (2009) Calmodulin kinase II-mediated sarcoplasmic reticulum Ca2+ leak promotes atrial fibrillation in mice. J Clin Invest 119(7):1940–1951. https://doi.org/10.1172/jci37059
Chen Q, Kirsch GE, Zhang D et al (1998) Genetic basis and molecular mechanism for idiopathic ventricular fibrillation. Nature 392(6673):293–296. https://doi.org/10.1038/32675
Chen PP, Patel JR, Rybakova IN, Walker JW, Moss RL (2010) Protein kinase A-induced myofilament desensitization to Ca(2+) as a result of phosphorylation of cardiac myosin-binding protein C. J Gen Physiol 136(6):615–627. https://doi.org/10.1085/jgp.201010448
Cheng J, Valdivia CR, Vaidyanathan R, Balijepalli RC, Ackerman MJ, Makielski JC (2013) Caveolin-3 suppresses late sodium current by inhibiting nNOS-dependent S-nitrosylation of SCN5A. J Mol Cell Cardiol 61:102–110. https://doi.org/10.1016/j.yjmcc.2013.03.013
Chiang DY, Li N, Wang Q et al (2014) Impaired local regulation of ryanodine receptor type 2 by protein phosphatase 1 promotes atrial fibrillation. Cardiovasc Res 103(1):178–187. https://doi.org/10.1093/cvr/cvu123
Chiang DY, Lahiri S, Wang G et al (2021) Phosphorylation-dependent interactome of ryanodine receptor type 2 in the heart. Proteomes. https://doi.org/10.3390/proteomes9020027
Chin D, Means AR (2000) Calmodulin: a prototypical calcium sensor. Trends Cell Biol 10(8):322–328. https://doi.org/10.1016/s0962-8924(00)01800-6
Chopra N, Kannankeril PJ, Yang T et al (2007) Modest reductions of cardiac calsequestrin increase sarcoplasmic reticulum Ca2+ leak independent of luminal Ca2+ and trigger ventricular arrhythmias in mice. Circ Res 101(6):617–626. https://doi.org/10.1161/CIRCRESAHA.107.157552
Chopra N, Yang T, Asghari P et al (2009) Ablation of triadin causes loss of cardiac Ca2+ release units, impaired excitation-contraction coupling, and cardiac arrhythmias. Proc Natl Acad Sci USA 106(18):7636–7641. https://doi.org/10.1073/pnas.0902919106
Clarke JD, Caldwell JL, Horn MA et al (2015) Perturbed atrial calcium handling in an ovine model of heart failure: potential roles for reductions in the L-type calcium current. J Mol Cell Cardiol 79:169–179. https://doi.org/10.1016/j.yjmcc.2014.11.017
Colbran RJ, Carmody LC, Bauman PA, Wadzinski BE, Bass MA (2003) Analysis of specific interactions of native protein phosphatase 1 isoforms with targeting subunits. Methods Enzymol 366:156–175. https://doi.org/10.1016/s0076-6879(03)66014-3
Connell P, Word TA, Wehrens XHT (2020) Targeting pathological leak of ryanodine receptors: preclinical progress and the potential impact on treatments for cardiac arrhythmias and heart failure. Expert Opin Ther Targets 24(1):25–36. https://doi.org/10.1080/14728222.2020.1708326
Cordeiro JM, Brugada R, Wu YS, Hong K, Dumaine R (2005) Modulation of I(Kr) inactivation by mutation N588K in KCNH2: a link to arrhythmogenesis in short QT syndrome. Cardiovasc Res 67(3):498–509. https://doi.org/10.1016/j.cardiores.2005.02.018
Coronel R, Janse MJ, Opthof T, Wilde AA, Taggart P (2012) Postrepolarization refractoriness in acute ischemia and after antiarrhythmic drug administration: action potential duration is not always an index of the refractory period. Heart Rhythm 9(6):977–982. https://doi.org/10.1016/j.hrthm.2012.01.021
Crotti L, Johnson CN, Graf E et al (2013) Calmodulin mutations associated with recurrent cardiac arrest in infants. Circulation 127(9):1009–1017. https://doi.org/10.1161/CIRCULATIONAHA.112.001216
Cunha SR, Bhasin N, Mohler PJ (2007) Targeting and stability of Na/Ca exchanger 1 in cardiomyocytes requires direct interaction with the membrane adaptor ankyrin-B. J Biol Chem 282(7):4875–4883. https://doi.org/10.1074/jbc.M607096200
DeGrande S, Nixon D, Koval O et al (2012) CaMKII inhibition rescues proarrhythmic phenotypes in the model of human ankyrin-B syndrome. Heart Rhythm 9(12):2034–2041. https://doi.org/10.1016/j.hrthm.2012.08.026
Dipla K, Mattiello JA, Margulies KB, Jeevanandam V, Houser SR (1999) The sarcoplasmic reticulum and the Na+/Ca2+ exchanger both contribute to the Ca2+ transient of failing human ventricular myocytes. Circ Res 84(4):435–444. https://doi.org/10.1161/01.res.84.4.435
Dobrev D, Wehrens XH (2014) Role of RyR2 phosphorylation in heart failure and arrhythmias: controversies around ryanodine receptor phosphorylation in cardiac disease. Circ Res 114(8):1311–1319. https://doi.org/10.1161/CIRCRESAHA.114.300568
Eisner DA, Caldwell JL, Kistamas K, Trafford AW (2017) Calcium and excitation-contraction coupling in the heart. Circ Res 121(2):181–195. https://doi.org/10.1161/CIRCRESAHA.117.310230
England PJ (1975) Correlation between contraction and phosphorylation of the inhibitory subunit of troponin in perfused rat heart. FEBS Lett 50(1):57–60. https://doi.org/10.1016/0014-5793(75)81040-4
Eschenhagen T (2013) PDE4 in the human heart-major player or little helper? Br J Pharmacol 169(3):524–527. https://doi.org/10.1111/bph.12168
Fan GC, Yuan Q, Zhao W, Chu G, Kranias EG (2007) Junctin is a prominent regulator of contractility in cardiomyocytes. Biochem Biophys Res Commun 352(3):617–622. https://doi.org/10.1016/j.bbrc.2006.11.093
Ferrantini C, Crocini C, Coppini R et al (2013) The transverse-axial tubular system of cardiomyocytes. Cell Mol Life Sci 70(24):4695–4710. https://doi.org/10.1007/s00018-013-1410-5
Franzini-Armstrong C, Protasi F, Ramesh V (1999) Shape, size, and distribution of Ca(2+) release units and couplons in skeletal and cardiac muscles. Biophys J 77(3):1528–1539. https://doi.org/10.1016/S0006-3495(99)77000-1
Franzini-Armstrong C, Protasi F, Tijskens P (2005) The assembly of calcium release units in cardiac muscle. Ann N Y Acad Sci 1047:76–85. https://doi.org/10.1196/annals.1341.007
Fukuda M, Yamamoto T, Nishimura S et al (2014) Enhanced binding of calmodulin to RyR2 corrects arrhythmogenic channel disorder in CPVT-associated myocytes. Biochem Biophys Res Commun 448(1):1–7. https://doi.org/10.1016/j.bbrc.2014.03.152
Gaita F, Giustetto C, Bianchi F et al (2003) Short QT Syndrome: a familial cause of sudden death. Circulation 108(8):965–970. https://doi.org/10.1161/01.CIR.0000085071.28695.C4
Gilbert G, Demydenko K, Dries E et al (2020) Calcium signaling in cardiomyocyte function. Cold Spring Harb Perspect Biol. https://doi.org/10.1101/cshperspect.a035428
Giustetto C, Di Monte F, Wolpert C et al (2006) Short QT syndrome: clinical findings and diagnostic-therapeutic implications. Eur Heart J 27(20):2440–2447. https://doi.org/10.1093/eurheartj/ehl185
Gray B, Bagnall RD, Lam L et al (2016) A novel heterozygous mutation in cardiac calsequestrin causes autosomal dominant catecholaminergic polymorphic ventricular tachycardia. Heart Rhythm 13(8):1652–1660. https://doi.org/10.1016/j.hrthm.2016.05.004
Greiser M, Kerfant BG, Williams GS et al (2014) Tachycardia-induced silencing of subcellular Ca2+ signaling in atrial myocytes. J Clin Invest 124(11):4759–4772. https://doi.org/10.1172/JCI70102
Gussak I, Brugada P, Brugada J et al (2000) Idiopathic short QT interval: a new clinical syndrome? Cardiology 94(2):99–102. https://doi.org/10.1159/000047299
Hain J, Onoue H, Mayrleitner M, Fleischer S, Schindler H (1995) Phosphorylation modulates the function of the calcium release channel of sarcoplasmic reticulum from cardiac muscle. J Biol Chem 270(5):2074–2081. https://doi.org/10.1074/jbc.270.5.2074
Hamilton S, Veress R, Belevych A, Terentyev D (2021) The role of calcium homeostasis remodeling in inherited cardiac arrhythmia syndromes. Pflugers Arch 473(3):377–387. https://doi.org/10.1007/s00424-020-02505-y
Hamilton S, Terentyeva R, Bogdanov V et al (2022) Ero1alpha-dependent ERp44 dissociation from RyR2 contributes to cardiac arrhythmia. Circ Res 130(5):711–724. https://doi.org/10.1161/CIRCRESAHA.121.320531
Hasenfuss G (1998) Alterations of calcium-regulatory proteins in heart failure. Cardiovasc Res 37(2):279–289. https://doi.org/10.1016/s0008-6363(97)00277-0
Higgins ER, Cannell MB, Sneyd J (2006) A buffering SERCA pump in models of calcium dynamics. Biophys J 91(1):151–163. https://doi.org/10.1529/biophysj.105.075747
Hobai IA, O’Rourke B (2000) Enhanced Ca(2+)-activated Na(+)-Ca(2+) exchange activity in canine pacing-induced heart failure. Circ Res 87(8):690–698. https://doi.org/10.1161/01.res.87.8.690
Horn T, Ullrich ND, Egger M (2013) ‘Eventless’ InsP3-dependent SR-Ca2+ release affecting atrial Ca2+ sparks. J Physiol 591(8):2103–2111. https://doi.org/10.1113/jphysiol.2012.247288
Hu D, Li Y, Zhang J et al (2017) The phenotypic spectrum of a mutation hotspot responsible for the short QT syndrome. JACC Clin Electrophysiol 3(7):727–743. https://doi.org/10.1016/j.jacep.2016.11.013
Hulsurkar MM, Lahiri SK, Karch J, Wang MC, Wehrens XHT (2022) Targeting calcium-mediated inter-organellar crosstalk in cardiac diseases. Expert Opin Ther Targets 26(4):303–317. https://doi.org/10.1080/14728222.2022.2067479
Ichikawa M, Aiba T, Ohno S et al (2016) Phenotypic variability of ANK2 mutations in patients with inherited primary arrhythmia syndromes. Circ J 80(12):2435–2442. https://doi.org/10.1253/circj.CJ-16-0486
Ihara Y, Kageyama K, Kondo T (2005) Overexpression of calreticulin sensitizes SERCA2a to oxidative stress. Biochem Biophys Res Commun 329(4):1343–1349. https://doi.org/10.1016/j.bbrc.2005.02.112
Irisawa H, Brown HF, Giles W (1993) Cardiac pacemaking in the sinoatrial node. Physiol Rev 73(1):197–227. https://doi.org/10.1152/physrev.1993.73.1.197
Juang JJ, Horie M (2016) Genetics of brugada syndrome. J Arrhythm 32(5):418–425. https://doi.org/10.1016/j.joa.2016.07.012
Kiewitz R, Acklin C, Schafer BW et al (2003) Ca2+ dependent interaction of S100A1 with the sarcoplasmic reticulum Ca2+ ATPase2a and phospholamban in the human heart. Biochem Biophys Res Commun 306(2):550–557. https://doi.org/10.1016/s0006-291x(03)00987-2
Kirchhefer U, Brekle C, Eskandar J et al (2014) Cardiac function is regulated by B56alpha-mediated targeting of protein phosphatase 2A (PP2A) to contractile relevant substrates. J Biol Chem 289(49):33862–33873. https://doi.org/10.1074/jbc.M114.598938
Kistamás K, Veress R, Horváth B, Bányász T, Nánási PP, Eisner DA (2020) Calcium handling defects and cardiac arrhythmia syndromes. Front Pharmacol 11:72. https://doi.org/10.3389/fphar.2020.00072
Kline CF, Scott J, Curran J, Hund TJ, Mohler PJ (2014) Ankyrin-B regulates Cav2.1 and Cav2.2 channel expression and targeting. J Biol Chem 289(8):5285–5295. https://doi.org/10.1074/jbc.M113.523639
Knollmann BC (2009) New roles of calsequestrin and triadin in cardiac muscle. J Physiol 587(Pt 13):3081–3087. https://doi.org/10.1113/jphysiol.2009.172098
Knollmann BC, Chopra N, Hlaing T et al (2006) Casq2 deletion causes sarcoplasmic reticulum volume increase, premature Ca2+ release, and catecholaminergic polymorphic ventricular tachycardia. J Clin Invest 116(9):2510–2520. https://doi.org/10.1172/JCI29128
Kobayashi YM, Jones LR (1999) Identification of triadin 1 as the predominant triadin isoform expressed in mammalian myocardium. J Biol Chem 274(40):28660–28668. https://doi.org/10.1074/jbc.274.40.28660
Lahat H, Eldar M, Levy-Nissenbaum E et al (2001) Autosomal recessive catecholamine or exercise-induced polymorphic ventricular tachycardia: clinical features and assignment of the disease gene to chromosome 1p13-21. Circulation 103(23):2822–2827. https://doi.org/10.1161/01.cir.103.23.2822
Lahiri SK, Aguilar-Sanchez Y, Wehrens XHT (2021a) Mechanisms underlying pathological Ca2+ handling in diseases of the heart. Pflugers Arch 473(3):331–347. https://doi.org/10.1007/s00424-020-02504-z
Lahiri SK, Aguilar-Sanchez Y, Wehrens XHT (2021b) Mechanisms underlying pathological Ca(2+) handling in diseases of the heart. Pflugers Arch 473(3):331–347. https://doi.org/10.1007/s00424-020-02504-z
Landstrom AP, Kellen CA, Dixit SS et al (2011) Junctophilin-2 expression silencing causes cardiocyte hypertrophy and abnormal intracellular calcium-handling. Circ Heart Fail 4(2):214–223. https://doi.org/10.1161/CIRCHEARTFAILURE.110.958694
Landstrom AP, Dobrev D, Wehrens XHT (2017) Calcium signaling and cardiac arrhythmias. Circ Res 120(12):1969–1993. https://doi.org/10.1161/CIRCRESAHA.117.310083
Lao QZ, Kobrinsky E, Harry JB, Ravindran A, Soldatov NM (2008) New determinant for the CaVbeta2 subunit modulation of the CaV1.2 calcium channel. J Biol Chem 283(23):15577–15588. https://doi.org/10.1074/jbc.M802035200
Lehnart SE, Wehrens XHT (2022) The role of junctophilin proteins in cellular function. Physiol Rev 102(3):1211–1261. https://doi.org/10.1152/physrev.00024.2021
Lehnart SE, Wehrens XH, Reiken S et al (2005) Phosphodiesterase 4D deficiency in the ryanodine-receptor complex promotes heart failure and arrhythmias. Cell 123(1):25–35. https://doi.org/10.1016/j.cell.2005.07.030
Levitan BM, Ahern BM, Aloysius A et al (2021) Rad-GTPase contributes to heart rate via L-type calcium channel regulation. J Mol Cell Cardiol 154:60–69. https://doi.org/10.1016/j.yjmcc.2021.01.005
Li L, Chu G, Kranias EG, Bers DM (1998) Cardiac myocyte calcium transport in phospholamban knockout mouse: relaxation and endogenous CaMKII effects. Am J Physiol 274(4):H1335–H1347. https://doi.org/10.1152/ajpheart.1998.274.4.H1335
Lieve KVV, Verhagen JMA, Wei J et al (2019) Linking the heart and the brain: Neurodevelopmental disorders in patients with catecholaminergic polymorphic ventricular tachycardia. Heart Rhythm 16(2):220–228. https://doi.org/10.1016/j.hrthm.2018.08.025
Limpitikul WB, Dick IE, Joshi-Mukherjee R, Overgaard MT, George AL Jr, Yue DT (2014) Calmodulin mutations associated with long QT syndrome prevent inactivation of cardiac L-type Ca(2+) currents and promote proarrhythmic behavior in ventricular myocytes. J Mol Cell Cardiol 74:115–124. https://doi.org/10.1016/j.yjmcc.2014.04.022
Louradour J, Bortolotti O, Torre E et al (2022) L-Type Cav1.3 calcium channels are required for beta-adrenergic triggered automaticity in dormant mouse sinoatrial pacemaker cells. Cells. https://doi.org/10.3390/cells11071114
Lu X, Ginsburg KS, Kettlewell S, Bossuyt J, Smith GL, Bers DM (2013) Measuring local gradients of intramitochondrial [Ca(2+)] in cardiac myocytes during sarcoplasmic reticulum Ca(2+) release. Circ Res 112(3):424–431. https://doi.org/10.1161/CIRCRESAHA.111.300501
Lucet V, Grau F, Denjoy I et al (1994) Long term course of catecholaminergic polymorphic ventricular tachycardia in children. Apropos of 20 cases with an 8 year-follow-up. Arch Pediatr 1(1):26–32
Maltsev AV, Evdokimovskii EV, Kokoz YM (2021) Protein kinase C-mediated calcium signaling as the basis for cardiomyocyte plasticity. Arch Biochem Biophys 701:108817. https://doi.org/10.1016/j.abb.2021.108817
Marian AJ, Asatryan B, Wehrens XHT (2020) Genetic basis and molecular biology of cardiac arrhythmias in cardiomyopathies. Cardiovasc Res 116(9):1600–1619. https://doi.org/10.1093/cvr/cvaa116
Marks ML, Whisler SL, Clericuzio C, Keating M (1995) A new form of long QT syndrome associated with syndactyly. J Am Coll Cardiol 25(1):59–64. https://doi.org/10.1016/0735-1097(94)00318-k
Maruyama M, Lin SF, Xie Y et al (2011) Genesis of phase 3 early afterdepolarizations and triggered activity in acquired long-QT syndrome. Circ Arrhythm Electrophysiol 4(1):103–111. https://doi.org/10.1161/CIRCEP.110.959064
Marx SO, Reiken S, Hisamatsu Y et al (2000) PKA phosphorylation dissociates FKBP12.6 from the calcium release channel (ryanodine receptor): defective regulation in failing hearts. Cell 101(4):365–376. https://doi.org/10.1016/s0092-8674(00)80847-8
Marx SO, Reiken S, Hisamatsu Y et al (2001) Phosphorylation-dependent regulation of ryanodine receptors: a novel role for leucine/isoleucine zippers. J Cell Biol 153(4):699–708. https://doi.org/10.1083/jcb.153.4.699
McHugh D, Sharp EM, Scheuer T, Catterall WA (2000) Inhibition of cardiac L-type calcium channels by protein kinase C phosphorylation of two sites in the N-terminal domain. Proc Natl Acad Sci U S A 97(22):12334–12338. https://doi.org/10.1073/pnas.210384297
McKinney BC, Murphy GG (2006) The L-Type voltage-gated calcium channel Cav1.3 mediates consolidation, but not extinction, of contextually conditioned fear in mice. Learn Mem 13(5):584–589. https://doi.org/10.1101/lm.279006
Miyata K, Ohno S, Itoh H, Horie M (2018) Bradycardia is a specific phenotype of catecholaminergic polymorphic ventricular tachycardia induced by RYR2 mutations. Intern Med 57(13):1813–1817. https://doi.org/10.2169/internalmedicine.9843-17
Mohler PJ, Gramolini AO, Bennett V (2002) Ankyrins. J Cell Sci 115(8):1565–1566. https://doi.org/10.1242/jcs.115.8.1565
Mohler PJ, Schott JJ, Gramolini AO et al (2003) Ankyrin-B mutation causes type 4 long-QT cardiac arrhythmia and sudden cardiac death. Nature 421(6923):634–639. https://doi.org/10.1038/nature01335
Mohler PJ, Splawski I, Napolitano C et al (2004) A cardiac arrhythmia syndrome caused by loss of ankyrin-B function. Proc Natl Acad Sci U S A 101(24):9137–9142. https://doi.org/10.1073/pnas.0402546101
Mohler PJ, Davis JQ, Bennett V (2005) Ankyrin-B coordinates the Na/K ATPase, Na/Ca exchanger, and InsP3 receptor in a cardiac T-tubule/SR microdomain. PLoS Biol 3(12):e423. https://doi.org/10.1371/journal.pbio.0030423
Negretti N, O’Neill SC, Eisner DA (1993) The relative contributions of different intracellular and sarcolemmal systems to relaxation in rat ventricular myocytes. Cardiovasc Res 27(10):1826–1830. https://doi.org/10.1093/cvr/27.10.1826
Njegic A, Wilson C, Cartwright EJ (2020) Targeting Ca2+ handling proteins for the treatment of heart failure and arrhythmias. Front Physiol 11:1068. https://doi.org/10.3389/fphys.2020.01068
Nyegaard M, Overgaard MT, Sondergaard MT et al (2012) Mutations in calmodulin cause ventricular tachycardia and sudden cardiac death. Am J Hum Genet 91(4):703–712. https://doi.org/10.1016/j.ajhg.2012.08.015
Ono K, Fozzard HA (1992) Phosphorylation restores activity of L-type calcium channels after rundown in inside-out patches from rabbit cardiac cells. J Physiol 454:673–688. https://doi.org/10.1113/jphysiol.1992.sp019286
Ortner NJ, Kaserer T, Copeland JN, Striessnig J (2020) De novo CACNA1D Ca(2+) channelopathies: clinical phenotypes and molecular mechanism. Pflugers Arch 472(7):755–773. https://doi.org/10.1007/s00424-020-02418-w
Paludan-Muller C, Ahlberg G, Ghouse J et al (2017) Integration of 60,000 exomes and ACMG guidelines question the role of catecholaminergic polymorphic ventricular tachycardia-associated variants. Clin Genet 91(1):63–72. https://doi.org/10.1111/cge.12847
Park SJ, Zhang D, Qi Y et al (2019) Insights into the pathogenesis of catecholaminergic polymorphic ventricular tachycardia from engineered human heart tissue. Circulation 140(5):390–404. https://doi.org/10.1161/CIRCULATIONAHA.119.039711
Pereira L, Rehmann H, Lao DH et al (2015) Novel epac fluorescent ligand reveals distinct Epac1 vs. Epac2 distribution and function in cardiomyocytes. Proc Natl Acad Sci USA 112(13):3991–3996. https://doi.org/10.1073/pnas.1416163112
Picht E, Zima AV, Shannon TR, Duncan AM, Blatter LA, Bers DM (2011) Dynamic calcium movement inside cardiac sarcoplasmic reticulum during release. Circ Res 108(7):847–856. https://doi.org/10.1161/CIRCRESAHA.111.240234
Platzer J, Engel J, Schrott-Fischer A et al (2000) Congenital deafness and sinoatrial node dysfunction in mice lacking class D L-type Ca2+ channels. Cell 102(1):89–97. https://doi.org/10.1016/s0092-8674(00)00013-1
Popescu I, Galice S, Mohler PJ, Despa S (2016) Elevated local [Ca2+] and CaMKII promote spontaneous Ca2+ release in ankyrin-B-deficient hearts. Cardiovasc Res 111(3):287–294. https://doi.org/10.1093/cvr/cvw093
Potenza DM, Janicek R, Fernandez-Tenorio M et al (2019) Phosphorylation of the ryanodine receptor 2 at serine 2030 is required for a complete beta-adrenergic response. J Gen Physiol 151(2):131–145. https://doi.org/10.1085/jgp.201812155
Prakriya M, Lewis RS (2015) Store-operated calcium channels. Physiol Rev 95(4):1383–1436. https://doi.org/10.1152/physrev.00020.2014
Priori SG, Chen SR (2011) Inherited dysfunction of sarcoplasmic reticulum Ca2+ handling and arrhythmogenesis. Circ Res 108(7):871–883. https://doi.org/10.1161/CIRCRESAHA.110.226845
Priori SG, Napolitano C, Tiso N et al (2001) Mutations in the cardiac ryanodine receptor gene (hRyR2) underlie catecholaminergic polymorphic ventricular tachycardia. Circulation 103(2):196–200. https://doi.org/10.1161/01.cir.103.2.196
Priori SG, Wilde AA, Horie M et al (2013) Executive summary: HRS/EHRA/APHRS expert consensus statement on the diagnosis and management of patients with inherited primary arrhythmia syndromes. Heart Rhythm 10(12):e85-108. https://doi.org/10.1016/j.hrthm.2013.07.021
Quick AP, Wang Q, Philippen LE et al (2017) SPEG (striated muscle preferentially expressed protein kinase) is essential for cardiac function by regulating junctional membrane complex activity. Circ Res 120(1):110–119. https://doi.org/10.1161/CIRCRESAHA.116.309977
Reed GJ, Boczek NJ, Etheridge SP, Ackerman MJ (2015) CALM3 mutation associated with long QT syndrome. Heart Rhythm 12(2):419–422. https://doi.org/10.1016/j.hrthm.2014.10.035
Reid DS, Tynan M, Braidwood L, Fitzgerald GR (1975) Bidirectional tachycardia in a child. A study using His bundle electrography. Br Heart J 37(3):339–344. https://doi.org/10.1136/hrt.37.3.339
Rizzi N, Liu N, Napolitano C et al (2008) Unexpected structural and functional consequences of the R33Q homozygous mutation in cardiac calsequestrin: a complex arrhythmogenic cascade in a knock in mouse model. Circ Res 103(3):298–306. https://doi.org/10.1161/CIRCRESAHA.108.171660
Rohr S (2004) Role of gap junctions in the propagation of the cardiac action potential. Cardiovasc Res 62(2):309–322. https://doi.org/10.1016/j.cardiores.2003.11.035
Roux-Buisson N, Cacheux M, Fourest-Lieuvin A et al (2012) Absence of triadin, a protein of the calcium release complex, is responsible for cardiac arrhythmia with sudden death in human. Hum Mol Genet 21(12):2759–2767. https://doi.org/10.1093/hmg/dds104
Sandow A (1952) Excitation-contraction coupling in muscular response. Yale J Biol Med 25(3):176–201
Schwartz PJ, Crotti L, Insolia R (2012) Long-QT syndrome: from genetics to management. Circ Arrhythm Electrophysiol 5(4):868–877. https://doi.org/10.1161/CIRCEP.111.962019
Shannon TR, Guo T, Bers DM (2003) Ca2+ scraps: local depletions of free [Ca2+] in cardiac sarcoplasmic reticulum during contractions leave substantial Ca2+ reserve. Circ Res 93(1):40–45. https://doi.org/10.1161/01.RES.0000079967.11815.19
Sherman J, Tester DJ, Ackerman MJ (2005) Targeted mutational analysis of ankyrin-B in 541 consecutive, unrelated patients referred for long QT syndrome genetic testing and 200 healthy subjects. Heart Rhythm 2(11):1218–1223. https://doi.org/10.1016/j.hrthm.2005.07.026
Skogestad J, Aronsen JM, Tovsrud N et al (2020) Coupling of the Na+/K+-ATPase to ankyrin B controls Na+/Ca2+ exchanger activity in cardiomyocytes. Cardiovasc Res 116(1):78–90. https://doi.org/10.1093/cvr/cvz087
Smith GL, Eisner DA (2019) Calcium buffering in the heart in health and disease. Circulation 139(20):2358–2371. https://doi.org/10.1161/CIRCULATIONAHA.118.039329
Solaro RJ, Moir AJ, Perry SV (1976) Phosphorylation of troponin I and the inotropic effect of adrenaline in the perfused rabbit heart. Nature 262(5569):615–617. https://doi.org/10.1038/262615a0
Sondergaard MT, Tian X, Liu Y et al (2015) Arrhythmogenic calmodulin mutations affect the activation and termination of cardiac ryanodine receptor-mediated Ca2+ release. J Biol Chem 290(43):26151–26162. https://doi.org/10.1074/jbc.M115.676627
Sondergaard MT, Liu Y, Larsen KT et al (2017) The arrhythmogenic calmodulin p.Phe142Leu mutation impairs C-domain Ca2+ binding but not calmodulin-dependent inhibition of the cardiac ryanodine receptor. J Biol Chem 292(4):1385–1395. https://doi.org/10.1074/jbc.M116.766253
Splawski I, Timothy KW, Sharpe LM et al (2004) Ca(V)1.2 calcium channel dysfunction causes a multisystem disorder including arrhythmia and autism. Cell 119(1):19–31. https://doi.org/10.1016/j.cell.2004.09.011
Striessnig J, Koschak A (2008) Exploring the function and pharmacotherapeutic potential of voltage-gated Ca2+ channels with gene knockout models. Channels 2(4):233–251. https://doi.org/10.4161/chan.2.4.5847
Sugawara K, Shibasaki T, Takahashi H, Seino S (2016) Structure and functional roles of Epac2 (Rapgef4). Gene 575(2 Pt 3):577–583. https://doi.org/10.1016/j.gene.2015.09.029
Sumitomo N (2016) Current topics in catecholaminergic polymorphic ventricular tachycardia. J Arrhythm 32(5):344–351. https://doi.org/10.1016/j.joa.2015.09.008
Sun B, Yao J, Ni M et al (2021) Cardiac ryanodine receptor calcium release deficiency syndrome. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aba7287
Swan H, Piippo K, Viitasalo M et al (1999) Arrhythmic disorder mapped to chromosome 1q42-q43 causes malignant polymorphic ventricular tachycardia in structurally normal hearts. J Am Coll Cardiol 34(7):2035–2042. https://doi.org/10.1016/s0735-1097(99)00461-1
Tadross MR, Dick IE, Yue DT (2008) Mechanism of local and global Ca2+ sensing by calmodulin in complex with a Ca2+ channel. Cell 133(7):1228–1240. https://doi.org/10.1016/j.cell.2008.05.025
Takasago T, Imagawa T, Shigekawa M (1989) Phosphorylation of the cardiac ryanodine receptor by cAMP-dependent protein kinase. J Biochem 106(5):872–877. https://doi.org/10.1093/oxfordjournals.jbchem.a122945
Takeshima H, Komazaki S, Nishi M, Iino M, Kangawa K (2000) Junctophilins: a novel family of junctional membrane complex proteins. Mol Cell 6(1):11–22. https://doi.org/10.1016/s1097-2765(00)00003-4
Templin C, Ghadri JR, Rougier JS et al (2011) Identification of a novel loss-of-function calcium channel gene mutation in short QT syndrome (SQTS6). Eur Heart J 32(9):1077–1088. https://doi.org/10.1093/eurheartj/ehr076
Tester DJ, Kim CSJ, Hamrick SK et al (2020) Molecular characterization of the calcium release channel deficiency syndrome. JCI Insight. https://doi.org/10.1172/jci.insight.135952
Thevenon D, Smida-Rezgui S, Chevessier F et al (2003) Human skeletal muscle triadin: gene organization and cloning of the major isoform, Trisk 51. Biochem Biophys Res Commun 303(2):669–675. https://doi.org/10.1016/s0006-291x(03)00406-6
Tiso N, Stephan DA, Nava A et al (2001) Identification of mutations in the cardiac ryanodine receptor gene in families affected with arrhythmogenic right ventricular cardiomyopathy type 2 (ARVD2). Hum Mol Genet 10(3):189–194. https://doi.org/10.1093/hmg/10.3.189
Tiso N, Salamon M, Bagattin A, Danieli GA, Argenton F, Bortolussi M (2002) The binding of the RyR2 calcium channel to its gating protein FKBP12.6 is oppositely affected by ARVD2 and VTSIP mutations. Biochem Biophys Res Commun 299(4):594–598. https://doi.org/10.1016/s0006-291x(02)02689-x
Torrente AG, Mesirca P, Neco P et al (2016) L-type Cav1.3 channels regulate ryanodine receptor-dependent Ca2+ release during sino-atrial node pacemaker activity. Cardiovasc Res 109(3):451–461. https://doi.org/10.1093/cvr/cvw006
Torrente AG, Mesirca P, Bidaud I, Mangoni ME (2020) Channelopathies of voltage-gated L-type Cav1.3/α1D and T-type Cav3.1/α1G Ca2+ channels in dysfunction of heart automaticity. Pflugers Arch 472(7):817–830. https://doi.org/10.1007/s00424-020-02421-1
Towbin JA, McKenna WJ, Abrams DJ et al (2019) 2019 HRS expert consensus statement on evaluation, risk stratification, and management of arrhythmogenic cardiomyopathy. Heart Rhythm 16(11):e301–e372. https://doi.org/10.1016/j.hrthm.2019.05.007
Trafford AW, Diaz ME, Eisner DA (1999) A novel, rapid and reversible method to measure Ca buffering and time-course of total sarcoplasmic reticulum Ca content in cardiac ventricular myocytes. Pflugers Arch 437(3):501–503. https://doi.org/10.1007/s004240050808
Trafford AW, Diaz ME, Eisner DA (2001) Coordinated control of cell Ca(2+) loading and triggered release from the sarcoplasmic reticulum underlies the rapid inotropic response to increased L-type Ca(2+) current. Circ Res 88(2):195–201. https://doi.org/10.1161/01.res.88.2.195
Tristani-Firouzi M, Jensen JL, Donaldson MR et al (2002) Functional and clinical characterization of KCNJ2 mutations associated with LQT7 (Andersen syndrome). J Clin Invest 110(3):381–388. https://doi.org/10.1172/JCI15183
Tse G, Sun B, Wong ST, Tse V, Yeo JM (2016) Anti-arrhythmic effects of hypercalcemia in hyperkalemic. Langendorff-Perfused Mouse Hearts Biomed Rep 5(3):301–310. https://doi.org/10.3892/br.2016.735
Tyan L, Foell JD, Vincent KP et al (2019a) Long QT syndrome caveolin-3 mutations differentially modulate Kv4 and Cav1.2 channels to contribute to action potential prolongation. J Physiol 597(6):1531–1551. https://doi.org/10.1113/JP276014
Tyan L, Foell JD, Vincent KP et al (2019b) Long QT syndrome caveolin-3 mutations differentially modulate Kv 4 and Cav 1.2 channels to contribute to action potential prolongation. J Physiol 597(6):1531–1551. https://doi.org/10.1113/JP276014
Vaidyanathan R, Vega AL, Song C et al (2013) The interaction of caveolin 3 protein with the potassium inward rectifier channel Kir2.1: physiology and pathology related to long qt syndrome 9 (LQT9). J Biol Chem 288(24):17472–17480. https://doi.org/10.1074/jbc.M112.435370
van der Werf C, Nederend I, Hofman N et al (2012) Familial evaluation in catecholaminergic polymorphic ventricular tachycardia: disease penetrance and expression in cardiac ryanodine receptor mutation-carrying relatives. Circ Arrhythm Electrophysiol 5(4):748–756. https://doi.org/10.1161/CIRCEP.112.970517
van Oort RJ, McCauley MD, Dixit SS et al (2010) Ryanodine receptor phosphorylation by calcium/calmodulin-dependent protein kinase II promotes life-threatening ventricular arrhythmias in mice with heart failure. Circulation 122(25):2669–2679. https://doi.org/10.1161/CIRCULATIONAHA.110.982298
Vatta M, Ackerman MJ, Ye B et al (2006) Mutant caveolin-3 induces persistent late sodium current and is associated with long-QT syndrome. Circulation 114(20):2104–2112. https://doi.org/10.1161/CIRCULATIONAHA.106.635268
Viatchenko-Karpinski S, Terentyev D, Gyorke I et al (2004) Abnormal calcium signaling and sudden cardiac death associated with mutation of calsequestrin. Circ Res 94(4):471–477. https://doi.org/10.1161/01.RES.0000115944.10681.EB
Walweel K, Gomez-Hurtado N, Rebbeck RT et al (2019) Calmodulin inhibition of human RyR2 channels requires phosphorylation of RyR2-S2808 or RyR2-S2814. J Mol Cell Cardiol 130:96–106. https://doi.org/10.1016/j.yjmcc.2019.03.018
Wang G, Zhu X, Xie W et al (2010) Rad as a novel regulator of excitation-contraction coupling and beta-adrenergic signaling in heart. Circ Res 106(2):317–327. https://doi.org/10.1161/CIRCRESAHA.109.208272
Wang G, Zhao N, Zhong S, Wang Y, Li J (2019) Safety and efficacy of flecainide for patients with catecholaminergic polymorphic ventricular tachycardia: a systematic review and meta-analysis. Medicine (baltimore) 98(34):e16961. https://doi.org/10.1097/MD.0000000000016961
Wang Y, Li C, Shi L et al (2020) Integrin beta1D deficiency-mediated RyR2 dysfunction contributes to catecholamine-sensitive ventricular tachycardia in arrhythmogenic right ventricular cardiomyopathy. Circulation 141(18):1477–1493. https://doi.org/10.1161/CIRCULATIONAHA.119.043504
Watanabe H, Knollmann BC (2011) Mechanism underlying catecholaminergic polymorphic ventricular tachycardia and approaches to therapy. J Electrocardiol 44(6):650–655. https://doi.org/10.1016/j.jelectrocard.2011.07.025
Watanabe H, Chopra N, Laver D et al (2009) Flecainide prevents catecholaminergic polymorphic ventricular tachycardia in mice and humans. Nat Med 15(4):380–383. https://doi.org/10.1038/nm.1942
Wehrens XH, Vos MA, Doevendans PA, Wellens HJ (2002) Novel insights in the congenital long QT syndrome. Ann Intern Med 137(12):981–992. https://doi.org/10.7326/0003-4819-137-12-200212170-00012
Wehrens XH, Lehnart SE, Huang F et al (2003) FKBP12.6 deficiency and defective calcium release channel (ryanodine receptor) function linked to exercise-induced sudden cardiac death. Cell 113(7):829–840. https://doi.org/10.1016/s0092-8674(03)00434-3
Wehrens XH, Lehnart SE, Reiken SR et al (2004a) Protection from cardiac arrhythmia through ryanodine receptor-stabilizing protein calstabin2. Science 304(5668):292–296. https://doi.org/10.1126/science.1094301
Wehrens XH, Lehnart SE, Reiken SR, Marks AR (2004b) Ca2+/calmodulin-dependent protein kinase II phosphorylation regulates the cardiac ryanodine receptor. Circ Res 94(6):e61-70. https://doi.org/10.1161/01.RES.0000125626.33738.E2
Wehrens XH, Lehnart SE, Marks AR (2005a) Intracellular calcium release and cardiac disease. Annu Rev Physiol 67:69–98. https://doi.org/10.1146/annurev.physiol.67.040403.114521
Wehrens XH, Lehnart SE, Reiken S et al (2005b) Enhancing calstabin binding to ryanodine receptors improves cardiac and skeletal muscle function in heart failure. Proc Natl Acad Sci U S A 102(27):9607–9612. https://doi.org/10.1073/pnas.0500353102
Wehrens XH, Lehnart SE, Reiken S, Vest JA, Wronska A, Marks AR (2006) Ryanodine receptor/calcium release channel PKA phosphorylation: a critical mediator of heart failure progression. Proc Natl Acad Sci U S A 103(3):511–518. https://doi.org/10.1073/pnas.0510113103
Wleklinski MJ, Kannankeril PJ, Knollmann BC (2020) Molecular and tissue mechanisms of catecholaminergic polymorphic ventricular tachycardia. J Physiol 598(14):2817–2834. https://doi.org/10.1113/JP276757
Yamaguchi H, Hara M, Strobeck M, Fukasawa K, Schwartz A, Varadi G (1998) Multiple modulation pathways of calcium channel activity by a beta subunit. Direct evidence of beta subunit participation in membrane trafficking of the alpha1C subunit. J Biol Chem 273(30):19348–19356. https://doi.org/10.1074/jbc.273.30.19348
Yang Y, Guo T, Oda T et al (2014) Cardiac myocyte Z-line calmodulin is mainly RyR2-bound, and reduction is arrhythmogenic and occurs in heart failure. Circ Res 114(2):295–306. https://doi.org/10.1161/CIRCRESAHA.114.302857
Yoshida A, Takahashi M, Imagawa T, Shigekawa M, Takisawa H, Nakamura T (1992) Phosphorylation of ryanodine receptors in rat myocytes during beta-adrenergic stimulation. J Biochem 111(2):186–190. https://doi.org/10.1093/oxfordjournals.jbchem.a123735
Zhang R, Zhao J, Potter JD (1995) Phosphorylation of both serine residues in cardiac troponin I is required to decrease the Ca2+ affinity of cardiac troponin C. J Biol Chem 270(51):30773–30780. https://doi.org/10.1074/jbc.270.51.30773
Zhang Q, Chen J, Qin Y, Wang J, Zhou L (2018) Mutations in voltage-gated L-type calcium channel: implications in cardiac arrhythmia. Channels (austin) 12(1):201–218. https://doi.org/10.1080/19336950.2018.1499368
Zhong X, Guo W, Wei J et al (2021) Identification of loss-of-function RyR2 mutations associated with idiopathic ventricular fibrillation and sudden death. Biosci Rep. https://doi.org/10.1042/BSR20210209
Zhu W, Wang C, Hu J et al (2018) Ankyrin-B Q1283H variant linked to arrhythmias via loss of local protein phosphatase 2A activity causes ryanodine receptor hyperphosphorylation. Circulation 138(23):2682–2697. https://doi.org/10.1161/CIRCULATIONAHA.118.034541
Funding
This work was supported by National Institutes of Health grants R01-HL147108, R01-HL153350, and R01-HL089598 (to X.H.T.W.), the Robert and Janice McNair Foundation McNair MD/PhD Scholars Program (J.K.), and the Baylor College of Medicine Medical Scientist Training Program (T32-GM136611; J.K., O.M., K.H.).
Author information
Authors and Affiliations
Contributions
All authors participated in writing of this manuscript.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Keefe, J.A., Moore, O.M., Ho, K.S. et al. Role of Ca2+ in healthy and pathologic cardiac function: from normal excitation–contraction coupling to mutations that cause inherited arrhythmia. Arch Toxicol 97, 73–92 (2023). https://doi.org/10.1007/s00204-022-03385-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-022-03385-0