Abstract
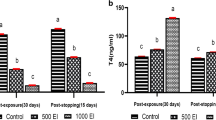
The long-lasting consequence of a new iodine thyroid blocking strategy (ITB) to be used in case of nuclear accident is evaluated in male Wistar rats using a metabolomics approach applied 30 days after ITB completion. The design used 1 mg/kg/day of KI over 8 days. Thyroid hormones remained unchanged, but there was a metabolic shift measured mainly in thyroid then in plasma and urine. In the thyroid, tyrosine metabolism associated to catecholamine metabolism was more clearly impacted than thyroid hormones pathway. It was accompanied by a peripheral metabolic shift including metabolic regulators, branched-chain amino acids, oxidant stress and inflammation-associated response. Our results suggested that iodide intake can impact gut microbiota metabolism, which was related to host metabolic regulations including in the thyroid. As there were no clear clinical signs of dysfunction or toxicity, we concluded that the measured metabolomics response to the new ITB strategy, especially in thyroid, is unlikely to reveal a pathological condition but a shift towards a new adaptive homeostatic state, called ‘allostatic regulation’. The question now is whether or not the shift is permanent and if so at what cost for long-term health. We anticipate our data as a start point for further regulatory toxicity studies.



Similar content being viewed by others
Abbreviations
- CV-ANOVA:
-
Cross-validation analysis of variance
- FT3:
-
Free triiodothyronine
- FT4:
-
Free thyroxine
- FWHM:
-
Full width at half maximum
- ITB:
-
Iodine thyroid blocking
- KI:
-
Potassium iodide
- LC/MS:
-
Liquid chromatography coupled with mass spectrometry
- m/z :
-
Mass-to-charge ratio
- NOAEL:
-
No-observed adverse effect limit
- PLS-DA:
-
Partial least squares–discriminant analysis
- PRIODAC:
-
Repeated stable iodide prophylaxis in accidental radioactive releases
- TSH:
-
Thyroid-stimulating hormone
- VIP:
-
Variable importance in projection
- WHO:
-
World Health Organization
References
Benderitter M, Pourcher T, Martin J-C et al (2018) Do multiple administrations of stable iodine protect population chronically exposed to radioactive iodine: what is PRIODAC research program (2014–22) teaching us? Radiat Prot Dosim 182(1):67–79
Cahoon EK, Nadyrov EA, Polyanskaya ON et al (2017) Risk of Thyroid Nodules in Residents of Belarus Exposed to Chernobyl Fallout as Children and Adolescents. J Clin Endocrinol Metab 102(7):2207–2217. https://doi.org/10.1210/jc.2016-3842
Corvilain B, Collyn L, van Sande J, Dumont JE (2000) Stimulation by iodide of H(2)O(2) generation in thyroid slices from several species. Am J Physiol Endocrinol Metab 278(4):E692–E699. https://doi.org/10.1152/ajpendo.2000.278.4.E692
Dreger S, Pfinder M, Christianson L, Lhachimi SK, Zeeb H (2015) The effects of iodine blocking following nuclear accidents on thyroid cancer, hypothyroidism, and benign thyroid nodules: design of a systematic review. Syst Rev 4:126. https://doi.org/10.1186/s13643-015-0106-3
Fiamoncini J, Rundle M, Gibbons H et al (2018) Plasma metabolome analysis identifies distinct human metabotypes in the postprandial state with different susceptibility to weight loss-mediated metabolic improvements. FASEB J 32(10):5447–5458. https://doi.org/10.1096/fj.201800330R
Giacomoni F, Le Corguille G, Monsoor M et al (2015) Workflow4Metabolomics: a collaborative research infrastructure for computational metabolomics. Bioinformatics 31(9):1493–1495. https://doi.org/10.1093/bioinformatics/btu813
Gieger C, Geistlinger L, Altmaier E et al (2008) Genetics meets metabolomics: a genome-wide association study of metabolite profiles in human serum. PLoS Genet 4(11):e1000282
Grison S, Fave G, Maillot M et al (2013) Metabolomics identifies a biological response to chronic low-dose natural uranium contamination in urine samples. Metabolomics 9(6):1168–1180
Grison S, Martin JC, Grandcolas L et al (2012) The metabolomic approach identifies a biological signature of low-dose chronic exposure to cesium 137. J Radiat Res (Tokyo) 53(1):33–43
Hendrikx T, Schnabl B (2019) Indoles: metabolites produced by intestinal bacteria capable of controlling liver disease manifestation. J Intern Med. https://doi.org/10.1111/joim.12892
Karnovsky A, Weymouth T, Hull T et al (2011) Metscape 2 bioinformatics tool for the analysis and visualization of metabolomics and gene expression data. Bioinformatics 28(3):373–380
Lebsir D, Cohen D, Manens L, et al. (2018a) Toxicology of repeated iodine thyroid blocking in adult rat. J Pharm Res3(1):1–8. https://opastonline.com/wp-content/uploads/2018/04/toxicology-of-repeated-iodine-thyroid-blocking-in-adult-rat-jpr-18-002.pdf. (ISSN: 2573-962X)
Lebsir D, Manens L, Grison S et al (2018) Effects of repeated potassium iodide administration on genes involved in synthesis and secretion of thyroid hormone in adult male rat. Mol Cell Endocrinol 474:119–126. https://doi.org/10.1016/j.mce.2018.02.017
Lestaevel P, Grison S, Favé G, Elie C, Dhieux B, Martin JC, Tack K, Souidi M (2016) Assessment of the central effects of natural uranium viabehavioural performances and the cerebrospinal fluid metabolome. Neural Plast. 2016:9740353. https://doi.org/10.1155/2016/9740353
Maayan ML, Sellitto RV, Volpert EM (1986) Dopamine and L-dopa: inhibition of thyrotropin-stimulated thyroidal thyroxine release. Endocrinology 118(2):632–636. https://doi.org/10.1210/endo-118-2-632
Martin J-C, Maillot M, Mazerolles G et al (2015a) Can we trust untargeted metabolomics? Results of the metabo-ring initiative, a large-scale, multi-instrument inter-laboratory study. Metabolomics 11(4):807–821
Martin JC, Berton A, Ginies C et al (2015b) Multi-level systems biology modeling characterized the atheroprotective efficiencies of modified dairy fats in a hamster model. Am J Physiol Heart Circ Physiol 309:H935–H945. https://doi.org/10.1152/ajpheart.00032.2015
Melander A, Sundler F, Westgren U (1973) Intrathyroidal amines and the synthesis of thyroid hormone. Endocrinology 93(1):193–200. https://doi.org/10.1210/endo-93-1-193
Mullur R, Liu YY, Brent GA (2014) Thyroid hormone regulation of metabolism. Physiol Rev 94(2):355–382. https://doi.org/10.1152/physrev.00030.2013
Nicola JP, Velez ML, Lucero AM, Fozzatti L, Pellizas CG, Masini-Repiso AM (2009) Functional toll-like receptor 4 conferring lipopolysaccharide responsiveness is expressed in thyroid cells. Endocrinology 150(1):500–508. https://doi.org/10.1210/en.2008-0345
O'Sullivan A, Gibney MJ, Connor AO et al (2011) Biochemical and metabolomic phenotyping in the identification of a vitamin D responsive metabotype for markers of the metabolic syndrome. Mol Nutr Food Res 55(5):679–690. https://doi.org/10.1002/mnfr.201000458
Phan G, Rebiere F, Suhard D et al (2017) Optimal KI prophylactic dose determination for thyroid radiation protection after a single administration in adult rats. Dose Response 15(4):1559325817746558. https://doi.org/10.1177/1559325817746558
Pietzner M, Engelmann B, Kacprowski T et al (2017) Plasma proteome and metabolome characterization of an experimental human thyrotoxicosis model. BMC Med 15(1):6. https://doi.org/10.1186/s12916-016-0770-8
Pietzner M, Kacprowski T, Friedrich N (2018) Empowering thyroid hormone research in human subjects using OMICs technologies. J Endocrinol 238(1):R13–R29. https://doi.org/10.1530/JOE-18-0117
Piras C, Arisci N, Poddighe S, Liggi S, Mariotti S, Atzori L (2017) Metabolomic profile in hyperthyroid patients before and after antithyroid drug treatment: Correlation with thyroid hormone and TSH concentration. Int J Biochem Cell Biol 93:119–128. https://doi.org/10.1016/j.biocel.2017.07.024
Ramsay DS, Woods SC (2014) Clarifying the roles of homeostasis and allostasis in physiological regulation. Psychol Rev 121(2):225–247. https://doi.org/10.1037/a0035942
Ramsay DS, Woods SC (2016) Physiological regulation: how it really works. Cell Metab 24(3):361–364. https://doi.org/10.1016/j.cmet.2016.08.004
Rosique C, Lebsir D, Lestaevel P et al (2019) Assessment of the effects of repeated doses of potassium iodide intake during pregnancy on male and female rat offspring using metabolomics and lipidomics. J Toxicol Environ Health A. https://doi.org/10.1080/15287394.2019.1625474
Shannon P, Markiel A, Ozier O et al (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13(11):2498–2504
Shen H, Han J, Li Y et al (2019) Different host-specific responses in thyroid function and gut microbiota modulation between diet-induced obese and normal mice given the same dose of iodine. Appl Microbiol Biotechnol. https://doi.org/10.1007/s00253-019-09687-1
Sterling P, Eyer J (1988) Allostasis: a new paradigm to explain arousal pathology. In: Fisher S, Reason J (eds) Handbook of life stress, cognition and health. Wiley, New York, pp 629–649. https://retina.anatomy.upenn.edu/pdfiles/5446.pdf
Thabuis C, Destaillats F, Lambert D et al (2011) Lipid transport function is the main target of oral oleylethanolamide to reduce adiposity in high-fat fed mice. J Lipid Res 7(52):1373–1382
Virili C, Centanni M (2015) Does microbiota composition affect thyroid homeostasis? Endocrine 49(3):583–587. https://doi.org/10.1007/s12020-014-0509-2
Want EJ, Masson P, Michopoulos F et al (2012) Global metabolic profiling of animal and human tissues via UPLC-MS. Nat Protoc 8(1):17–32
WHO (2017) Iodine thyroid blocking: Guidelines for use in planning for and responding to radiological and nuclear emergencies. World Health Organization. https://www.who.int/iris/handle/10665/259510. Licence: CC BY-NC-SA 3.0 IGO
Wikoff WR, Anfora AT, Liu J et al (2009) Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proc Natl Acad Sci USA 106(10):3698–3703
Wolff J, Chaikoff IL (1948) Plasma inorganic iodide as a homeostatic regulator of thyroid function. J Biol Chem 174(2):555–564
Wu S, Tan G, Dong X et al (2013) Metabolic profiling provides a system understanding of hypothyroidism in rats and its application. PLoS ONE 8(2):e55599. https://doi.org/10.1371/journal.pone.0055599
Wurtz P, Soininen P, Kangas AJ et al (2011) Characterization of systemic metabolic phenotypes associated with subclinical atherosclerosis. Mol Biosyst 7(2):385–393
Xia J, Psychogios N, Young N, Wishart DS (2009) MetaboAnalyst: a web server for metabolomic data analysis and interpretation. Nucl Acids Res 37(suppl_2):W652–W660 doi:10.1093/nar/gkp356. https://academic.oup.com/nar/article/37/suppl_2/W652/1137529
Yamazaki K, Tanigawa K, Suzuki K et al (2010) Iodide-induced chemokines and genes related to immunological function in cultured human thyroid follicles in the presence of thyrotropin. Thyroid 20(1):67–76. https://doi.org/10.1089/thy.2009.0242
Yamazaki K, Yamada E, Kanaji Y et al (2003) Genes regulated by thyrotropin and iodide in cultured human thyroid follicles: analysis by cDNA microarray. Thyroid 13(2):149–158. https://doi.org/10.1089/105072503321319459
Zandkarimi F, Vanegas J, Fern X, Maier CS, Bobe G (2018) Metabotypes with elevated protein and lipid catabolism and inflammation precede clinical mastitis in prepartal transition dairy cows. J Dairy Sci 101(6):5531–5548. https://doi.org/10.3168/jds.2017-13977
Acknowledgements
This study is a part of the PRIODAC research program supported by the French National Research Agency (ANR) and the Investing for the Future program (Grant #11-RSNR-0019, 2014). The authors thank the Pharmacie Centrale des Armées—French Armed Forces Central Pharmacy—for providing the KI solution and F. Voyer, A. Sache and R. Granger for animal care.
Author information
Authors and Affiliations
Contributions
CR performed the metabolomics analyses and contribute to data analyses and manuscript writing, DL contributed to study design and manuscript writing, SB performed analyses, PG and FC-M contributed to manuscript writing, MB and MS designed the study and contributed to manuscript writing, J-CM contributed to study design, data analyses and manuscript writing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Fig. 1
Thyroid YWHAH or tyrosine hydroxylase or tyrosine 3-monoxygenase (A) and thyroid peroxidase (B) gene expression in the thyroid of male rats collected 30 days after completion of the ITB strategy using 1 mg/kg/day over 8 days. Microarray data (published in Lebsir, D., Cohen, D., Manens, L., Grison, S., Tack, K., Benderitter, M., Pech, A., Lestaevel, P., and Souidi, M. (2018). Toxicology of repeated iodine thyroid blocking in adult rat. Journal of Pharmaceutical Research3(1), 1–8). P value after t-test, n=5 rats per group (PDF 508 kb)
Rights and permissions
About this article
Cite this article
Rosique, C., Lebsir, D., Benatia, S. et al. Metabolomics evaluation of repeated administration of potassium iodide on adult male rats. Arch Toxicol 94, 803–812 (2020). https://doi.org/10.1007/s00204-020-02666-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-020-02666-w