Abstract
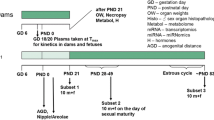
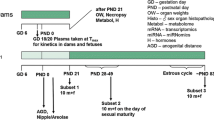
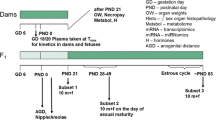
We performed a uterotrophic assay, the Hershberger assay, and the 28-day repeated-dose toxicity study (enhanced OECD test guideline no. 407) of 3-amino-1,2,4-triazole based on the OECD draft protocols. In the uterotrophic assay, female SD rats were subcutaneously injected with the chemical at doses of 0, 100, 300, and 1,000 mg/kg on each of 3 days from postnatal day 20 to day 22, and no changes were observed. In the Hershberger assay, the test chemical was orally administered at doses of 0, 40, 200, and 1,000 mg/kg/day to castrated male Wistar rats for 10 consecutive days beginning on postnatal day 56, and no androgen agonistic and antagonistic changes were observed. Alternatively, when the test chemical was orally administered at doses 0, 5, 25, and 125 mg/kg/day for at least 28 days in the subacute oral toxicity study, thyroid follicular epithelial cell hypertrophy with increased thyroid weights was detected in the male and female rats in 25 and/or 125 mg/kg groups, and hypertrophy of the anterior pituitary cells with increased pituitary weights in male and female rats was also observed in the 125 mg/kg group. Furthermore, serum T3 and T4 values decreased and serum TSH values increased in male and female rats in the 125 mg/kg group. Therefore, 3-amino-1,2,4-triazole was concluded to have anti-thyroid acting as endocrine-mediated effects, but no estrogenic or androgenic effects. In addition, decreased body weight, and abnormal biochemical parameters attributed to thyroid, liver or kidney dysfunction were observed in male and female rats in the 25 and/or 125 mg/kg groups.
Similar content being viewed by others
References
Alexander NM (1959) Antithyroid action of 3-anino-1, 2, 4-triazole. J Biol Chem 234:148–150
Andrews P, Freyberger A, Hartmann E, Eiben R, Loof I, Schmidt U, Temerowski M, Becka M (2001) Feasibility and potential gains of enhancing the subacute rat study protocol (OECD test guideline no. 407) by additional parameters selected to determine endocrine modulation A pre-validation study to determine endocrine-mediated effects of the antiandrogenic drug flutamide. Arch Toxicol 75:65–73
Capen CC (1994) Mechanisms of chemical injury of thyroid gland. Prog Clin Biol Res 387:173–191
Cartier LJ, Williams IK, Holloszy J, Premachandra BN (1985) Potentiation of thyroxine 5-deiodination by aminotriazole. Biochim Biophys Acta 843:68–72
Davis RG, Madsen KM, Fregly MJ, Tisher CC (1983) Kidney structure in hypothyroidism. Am J Pathol 113:41–49
Gelbke HP, Hofmann A, Owens JW, Freyberger A (2007) The enhancement of the subacute repeat dose toxicity test OECD TG 407 for the detection of endocrine active chemicals: comparison with toxicity tests of longer duration. Arch Toxicol 81:227–250
Higashihara N, Shiraishi K, Miyata K, Oshima Y, Minobe Y, Yamasaki K (2007) Subacute oral toxicity study of bisphenol F based on the draft protocol for the “Enhanced OECD Test Guideline no. 407”. Arch Toxicol 81:825–832
Kennel P, Pallen C, Barale-Thomas E, Espuna G, Bars R (2003) Tamoxifen: 28-day oral toxicity study in the rat based on the enhanced OECD test guideline 407 to detect endocrine effects. Arch Toxicol 77:487–489
Kunimatsu T, Yamada T, Miyata K, Yabushita S, Seki T, Okuno Y, Matsuo M (2004) Evaluation for reliability and feasibility of the draft protocol for the enhanced rat 28-day subacute study (OECD guideline 407) using androgen antagonist flutamide. Toxicology 2000:77–89
Mayberry WE (1968) Antithyroid effect of 3-amono-1,2,4-triazole. Proc Soc Exp Biol Med 129:551–556
Mellert W, Deckardt K, Walter J, Gfatter S, van Ravenzwaay B (2003) Detection of endocrine-modulating effects of the antithyroid acting drug 6-propyl-2-thiouracil in rats, based on the “enhanced OECD test guideline 407”. Regul Toxicol Pharmacol 38:368–377
Miyata K, Shiraishi S, Houshuyama S, Imatanaka N, Umano T, Minobe Y, Yamasaki K (2006) Subacute oral toxicity study of di(2-ethylhexyl)adipate based on the draft protocol for the “Enhanced OECD Test Guideline no. 407”. Arch Toxicol 80:181–186
OECD (1999) EMSG OECD draft proposal for testing of adequacy of an enhanced OECD protocol, Repeated dose (28 days) toxicity (oral) study, based on OECD 407, OECD, Paris
OECD (2000) OECD protocol for investigating the efficacy of the enhanced TG407 test guideline (phase 2), rationale for the investigation, and description of the protocol, OECD, Paris
OECD (2001) Third meeting of the validation management group for the screening and testing of endocrine disrupters (mammalian effects), OECD, Paris
OECD (2006) OECD 5th meeting of the validation management group for mammalian effects testing (VMG-Mammalian) of the task force on endocrine disrupters testing and assessment. OECD, Washington
OECD (2007a) OECD 6th meeting of the validation management group for mammalian effects testing (VMG-Mammalian) of the task force on endocrine disrupters testing and assessment. OECD, Ljubljana
OECD (2007b) OECD 7th meeting of the validation management group for mammalian effects testing (VMG-Mammalian) of the task force on endocrine disrupters testing and assessment. OECD, Paris
Okazaki K, Okazaki S, Nishimura S, Nakamura H, Kitamura Y, Hatayama K, Nakamura A, Tsuda T, Katsumata T, Nishikawa A, Hirose M (2001) A repeated 28-day oral dose toxicity study of methoxychlor in rats, based on the ‘enhanced OECD test guideline 407’ for screening endocrine-disrupting chemicals. Arch Toxicol 75:513–521
Okazaki K, Imazawa T, Nakamura H, Furukawa F, Nishikawa A, Hirose M (2002a) A repeated 28-day oral dose toxicity study of 17α-methyltestosterone in rats, based on the ‘enhanced OECD test guideline 407’ for screening endocrine-disrupting chemicals. Arch Toxicol 75:635–642
Okazaki K, Okazaki S, Nakamura H, Kitamura Y, Hatayama K, Wakabayashi S, Tsuda T, Katsumata T, Nishikawa A, Hirose M (2002b) A repeated 28-day oral dose toxicity study of genisein in rats, based on the ‘enhanced OECD test guideline 407’ for screening endocrine-disrupting chemicals. Arch Toxicol 76:553–559
Shin JH, Moon HJ, Kim TS, Kang IH, Kim HY, Choi KS, Han SY (2006a) Repeated 28-day oral toxicity study of vinclozolin in rats based on the draft protocol for the “Enhanced OECD Test Guideline No. 407” to detect endocrine effects. Arch Toxicol 80:547–554
Shin JH, Moon HJ, Kang IH, Kim TS, Kim IY, Park IS, Kim HS, Jeung EB, Han SY (2006b) Repeated 28-day oral toxicity study of ketoconazole in rats based on the draft protocol for the “Enhanced OECD Test Guideline No. 407” to detect endocrine effects. Arch Toxicol 80:797–803
Shiraishi K, Miyata K, Houshuyama S, Imatanaka N, Umano T, Minobe Y, Yamasaki K (2006) Subacute oral toxicity study of diethylphthalate based on the draft protocol for “Enhanced OECD Test Guideline no. 407”. Arch Toxicol 80:10–16
Takaoka M, Manabe S, Yamato T, Teranishi M, Matsunuma N, Masuda H, Goto N (1994) Comparative study of goitrogenic actions of 3-substituted 1, 2, 4-triazoles in rats. J Vet Med Sci 56:341–346
Toyoda K, Shibuntani M, Tamaru T, Koujitani T, Uneyama C, Hirose M (2000) Repeated dose (28 days) oral toxicity study of flutamide in rats, based on the draft protocol for the ‘enhanced OECD test guideline 407’ for screening for endocrine-disrupting chemicals. Arch Toxicol 74:127–132
Wason S, Pohlmeyer-Esch G, Pallen C, Palazzi X, Espina G, Bars R (2003) 17α-methyltestosterone: 28-day oral toxicity study in the rat based on the “enhanced OECD test guideline 407” to detect endocrine effects. Toxicology 192:119–137
Woo GH, Shibutani M, Ichiki T, Hamamura M, Lee KY, Inoue K, Hirose MA (2007) Repeated 28-day oral dose toxicity study of nonylphenol in rats, based on the ‘Enhanced OECD Test Guideline 407’ for screening of endocrine-disrupting chemicals. Arch Toxicol 81:77–88
Yamasaki K, Tago Y, Nagai K, Sawaki M, Noda S, Takatsuki M (2002a) Comparison of toxicity studies based on the draft protocol for the “enhanced OECD test guideline no. 407” and the research protocol of “pubertal development and thyroid function in immature male rats” with 6-n-propyl-2-thiouracil. Arch Toxicol 76:495–501
Yamasaki K, Sawaki M, Noda S, Imatanaka M, Takatsuki M (2002b) Subacute oral toxicity study of ethynyl estradiol and bisphenol A based on the draft protocol for the “enhanced OECD test guideline no. 407”. Arch Toxicol 76:65–74
Yamasaki K, Shiraishi K, Miyata K, Muroi T, Higashihara N, Oshima Y, Minobe Y (2008) Uterotrophic assay, Hershberger assay, and subacute oral toxicity study of 4,4′-butylidenebis(2-tert-butyl-5-methylphenol) and 3-(dibutylamino)phenol, based on the OECD draft protocols. Arch Toxicol (in press)
Acknowledgments
This work was supported by a grant from the Ministry of Economical Trade and Industry.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Umano, T., Shiraishi, K., Minobe, Y. et al. Uterotrophic assay, Hershberger assay, and subacute oral toxicity study of 3-amino-1,2,4-triazole based on the Organization for Economic Co-operation and Development draft protocols. Arch Toxicol 83, 459–467 (2009). https://doi.org/10.1007/s00204-008-0357-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-008-0357-z