Abstract
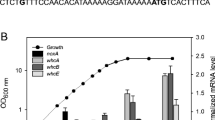
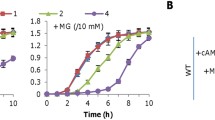
The ethanol-producing bacterium Zymomonas mobilis is of great interest from a bioenergetic perspective because, although it has a very high respiratory capacity, the respiratory system does not appear to be primarily required for energy conservation. To investigate the regulation of respiratory genes and function of electron transport branches in Z. mobilis, several mutants of the common wild-type strain Zm6 (ATCC 29191) were constructed and analyzed. Mutant strains with a chloramphenicol-resistance determinant inserted in the genes encoding the cytochrome b subunit of the bc 1 complex (Zm6-cytB), subunit II of the cytochrome bd terminal oxidase (Zm6-cydB), and in the catalase gene (Zm6-kat) were constructed. The cytB and cydB mutants had low respiration capacity when cultivated anaerobically. Zm6-cydB lacked the cytochrome d absorbance at 630 nm, while Zm6-cytB had very low spectral signals of all cytochromes and low catalase activity. However, under aerobic growth conditions, the respiration capacity of the mutant cells was comparable to that of the parent strain. The catalase mutation did not affect aerobic growth, but rendered cells sensitive to hydrogen peroxide. Cytochrome c peroxidase activity could not be detected. An upregulation of several thiol-dependent oxidative stress-protective systems was observed in an aerobically growing ndh mutant deficient in type II NADH dehydrogenase (Zm6-ndh). It is concluded that the electron transport chain in Z. mobilis contains at least two electron pathways to oxygen and that one of its functions might be to prevent endogenous oxidative stress.





Similar content being viewed by others
References
Atack JM, Kelly DJ (2006) Structure, mechanism and physiological roles of bacterial cytochrome c peroxidases. Adv Microb Physiol 52:73–106
Belaich JP, Senez JC (1965) Influence of aeration and pantothenate on growth yields of Zymomonas mobilis. J Bacteriol 89:1195–1200
Bergmeyer HU, Gawehn K, Grassl M (1974) Glutathione reductase. In: Bergmeyer HU (ed) Methods of enzymatic analysis, vol 1. Academic Press, New York, pp 465–466
Bringer S, Finn RK, Sahm H (1984) Effect of oxygen on the metabolism of Zymomonas mobilis. Arch Microbiol 139:376–381
Charoensuk K, Irie A, Lertwattanasakul N, Sootsuwan K, Thanonkeo P, Yamada M (2011) Physiological importance of cytochrome c peroxidase in ethanologenic thermotolerant Zymomonas mobilis. J Mol Microbiol Biotechnol 20:70–82
Cosgrove K, Coutts G, Jonsson I-M, Tarkowski A, Kokai-Kun JF, Mond JJ, Foster SJ (2007) Catalase (KatA) and alkyl hydroperoxide reductase (AhpC) have compensatory roles in peroxide stress resistance and are required for survival, persistance, and nasal colonization in Staphylococcus aureus. J Bacteriol 189:1025–1035
Echave P, Tamarit J, Cabiscol E, Ros J (2003) Novel antioxidant role of alcohol dehydrogenase E from Escherichia coli. J Biol Chem 278:30193–30198
Ellfolk N, Soininen R (1970) Pseudomonas cytochrome c peroxidase. Acta Chem Scand 24:2126–2136
Gonzalez-Flecha B, Demple B (1994) Intracellular generation of superoxide as a by-product of Vibrio harveyi luciferase expressed in Escherichia coli. J Bacteriol 176:2293–2299
Gonzalez-Flecha B, Demple B (1995) Metabolic sources of hydrogen peroxide in aerobically growing Escherichia coli. J Biol Chem 270:13681–13687
Goodhew CF, Wilson IBH, Hunter DJB, Pettigrew GW (1990) The cellular location and specificity of bacterial cytochrome c peroxidases. Biochem J 271:707–712
Kalnenieks U (2006) Physiology of Zymomonas mobilis: some unanswered questions. Adv Microb Physiol 51:73–117
Kalnenieks U, de Graaf AA, Bringer-Meyer S, Sahm H (1993) Oxidative phosphorylation in Zymomonas mobilis. Arch Microbiol 160:74–79
Kalnenieks U, Galinina N, Bringer-Meyer S, Poole RK (1998) Membrane D-lactate oxidase in Zymomonas mobilis: evidence for a branched respiratory chain. FEMS Microbiol Lett 168:91–97
Kalnenieks U, Galinina N, Toma MM, Poole RK (2000) Cyanide inhibits respiration yet stimulates aerobic growth of Zymomonas mobilis. Microbiology 146:1259–1266
Kalnenieks U, Galinina N, Toma MM (2005) Physiological regulation of the properties of alcohol dehydrogenase II (ADH II) of Zymomonas mobilis: NADH renders ADH II resistant to cyanide and aeration. Arch Microbiol 183:450–455
Kalnenieks U, Galinina N, Toma MM, Pickford JL, Rutkis R, Poole RK (2006) Respiratory behaviour of a Zymomonas mobilis adhB:kanr mutant supports the hypothesis of two alcohol dehydrogenase isoenzymes catalysing opposite reactions. FEBS Lett 580:5084–5088
Kalnenieks U, Galinina N, Strazdina I, Kravale Z, Pickford JL, Rutkis R, Poole RK (2008) NADH dehydrogenase deficiency results in low respiration rate and improved aerobic growth of Zymomonas mobilis. Microbiology 154:989–994
Kelly MJ, Poole RK, Yates MG, Kennedy C (1990) Cloning and mutagenesis of genes encoding the cytochrome bd terminal oxidase complex in Azotobacter vinelandii: mutants deficient in the cytochrome d complex are unable to fix nitrogen in air. J Bacteriol 172:6010–6019
Korshunov S, Imlay JA (2010) Two sources of endogenous hydrogen peroxide in Escherichia coli. Mol Microbiol 75:1389–1401
Kouvelis VN, Saunders E, Brettin TS, Bruce D, Detter C, Han C, Typas MA, Pappas KM (2009) Complete genome sequence of the ethanol producer Zymomonas mobilis NCIMB 11163. J Bacteriol 191:7140–7141
Liang C–C, Lee W-C (1998) Characteristics and transformation of Zymomonas mobilis with plasmid pKT230 by electroporation. Bioprocess Eng 19:81–85
Markwell MAK, Haas SM, Bieber LL, Talbert NE (1978) A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Anal Biochem 87:206–210
Osman YA, Conway T, Bonetti SJ, Ingram LO (1987) Glycolytic flux in Zymomonas mobilis: enzyme and metabolite levels during batch fermentation. J Bacteriol 169:3726–3736
Panek HR, O’Brian MR (2004) KatG is the primary detoxifier of hydrogen peroxide produced by aerobic metabolism in Bradyrhizobium japonicum. J Bacteriol 186:7874–7880
Poole RK, Cook GM (2000) Redundancy of aerobic respiratory chains in bacteria? Routes, reasons and regulation. Adv Microb Physiol 43:165–224
Reyes L, Scopes RK (1991) Membrane-associated ATPase from Zymomonas mobilis; purification and characterization. BBA 1068:174–178
Rogers PL, Lee KJ, Skotnicki ML, Tribe DE (1982) Ethanol production by Zymomonas mobilis. Adv Biochem Eng 23:37–84
Rogers PL, Jeon YJ, Lee KJ, Lawford HG (2007) Zymomonas mobilis for fuel ethanol and higher value products. Adv Biochem Eng Biotechnol 108:263–288
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor
Schmehl M, Jahn A, Meyer zu Vilsendorf A, Hennecke S, Masepohl B, Schuppler M, Marxer M, Oelze J, Klipp W (1993) Identification of a new class of nitrogen fixation genes in Rhodobacter capsulatus: a putative membrane complex involved in electron transport to nitrogenase. Mol Gen Genet 241:602–615
Seaver LC, Imlay JA (2001) Hydrogen peroxide fluxes and compartmentalization inside growing Escherichia coli. J Bacteriol 183:7182–7189
Seo JS, Chong H, Park HS, Yoon KO, Jung C, Kim JJ, Hong JH, Kim H, Kim JH et al (2005) The genome sequence of the ethanologenic bacterium Zymomonas mobilis ZM4. Nat Biotechnol 23:63–68
Sootsuwan K, Lertwattanasakul N, Thanonkeo P, Matsushita K, Yamada M (2008) Analysis of the respiratory chain in ethanologenic Zymomonas mobilis with a cyanide-resistant bd-type ubiquinol oxidase as the only terminal oxidase and its possible physiological roles. J Mol Microbiol Biotechnol 14:163–175
Sprenger G (1996) Carbohydrate metabolism in Zymomonas mobilis: a catabolic highway with some scenic routes. FEMS Microbiol Lett 145:301–307
Strohdeicher M, Neuß B, Bringer-Meyer S, Sahm H (1990) Electron transport chain of Zymomonas mobilis. Interaction with the membrane-bound glucose dehydrogenase and identification of ubiquinone 10. Arch Microbiol 154:536–543
Swings J, DeLey J (1977) The biology of Zymomonas. Bacteriol Rev 41:1–46
Trumpower BL, Gennis RB (1994) Energy transduction by cytochrome complexes in mitochondrial and bacterial respiration: the enzymology of coupling electron transfer reactions to transmembrane proton translocation. Annu Rev Biochem 63:675–716
Viikari L (1986) By-product formation in ethanol fermentation by Zymomonas mobilis. Technical Research Centre of Finland. Publication 27
Viikari L, Berry DR (1988) Carbohydrate metabolism in Zymomonas. Crit Rev Biotechnol 7:237–261
Yang S, Pappas KM, Hauser LJ, Land ML, Chen G-L, Hurst GB, Pan C, Kouvelis VN, Typas MA, Pelletier DA, Klingeman DL, Chang YJ, Samatova NF, Brown SD (2009a) Improved genome annotation for Zymomonas mobilis. Nat Biotechnol 27:893–894
Yang S, Tschaplinski TJ, Engle NL, Carroll SL, Martin SL, Davison BH, Palumbo AV, Rodriguez M Jr, Brown SD (2009b) Transcriptomic and metabolomic profiling of Zymomonas mobilis during aerobic and anaerobic fermentations. BMC Genomics 10:34
Acknowledgments
This work was funded by grant 09.1306 of Latvian Council of Science, The Royal Society Travel Grant TG 102318 (for UK), and by Latvian ESF project 2009/0207/1DP/1.1.1.2.0/09/APIA/VIAA/128.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Gregory Cook.
Rights and permissions
About this article
Cite this article
Strazdina, I., Kravale, Z., Galinina, N. et al. Electron transport and oxidative stress in Zymomonas mobilis respiratory mutants. Arch Microbiol 194, 461–471 (2012). https://doi.org/10.1007/s00203-011-0785-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-011-0785-7