Key message
The elongated hypocotyl1 (elh1) mutant in cucumber is due to a mutation in CsHY2, which is a homolog of the Arabidopsis HY2 encoding the phytochromobilin (PΦB) synthase for phytochrome biosynthesis
Abstract
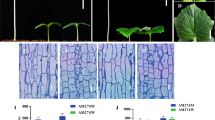
Hypocotyl length is a critical determinant in establishing high quality seedlings for successful cucumber production, but knowledge on the molecular regulation of hypocotyl growth in cucumber is very limited. Here, we reported identification and characterization of a cucumber elongated hypocotyl 1 (elh1) mutant. We found that the longer hypocotyl in elh1 was due to longitudinal growth of hypocotyl cells. With fine mapping, the elh1 locus was delimited to a 20.9-kb region containing three annotated genes; only one polymorphism was identified in this region between two parental lines, which was a non-synonymous SNP (G28153633A) in the third exon of CsHY2 (CsGy1G030000) that encodes a phytochromobilin (PΦB) synthase. Uniqueness of the mutant allele at CsHY2 was verified in natural cucumber populations. Ectopic expression of CsHY2 in Arabidopsis hy2-1 long-hypocotyl mutant led to reduced hypocotyl length. The PΦB protein was targeted to the chloroplast. The expression levels of CsHY2 and five phytochrome genes CsPHYA1, CsPHYA2, CsPHYB, CsPHYC and CsPHYE were all significantly down-regulated while several cell elongation related genes were up-regulated in elh1 mutant compared to wild-type cucumber, which are correlated with dynamic hypocotyl elongation in the mutant. RNA-seq analysis in the WT and mutant revealed differentially expressed genes involved in porphyrin and chlorophyll metabolisms, cell elongation and plant hormone signal transduction pathways. This is the first report to characterize and clone the CsHY2 gene in cucumber. This work reveals the important of CsHY2 in regulating hypocotyl length and extends our understanding of the roles of CsHY2 in cucumber.







Similar content being viewed by others
References
Bawa G, Feng LY, Chen GP, Chen H, Hu Y et al (2020) Gibberellins and auxin regulate soybean hypocotyl elongation under low light and high-temperature interaction. Physiol Plant 170:345–356
Bo KL, Wang H, Pan YP, Behera TK, Pandey S, Wen CL, Wang YH, Simon PW, Li YH, Chen JF, Weng YQ (2016) SHORT HYPOCOTYL1 encodes a SMARCA3-like chromatin remodeling factor regulating elongation. Plant Physiol 172(2):1273–1292
Boron AK, Vissenberg K (2014) The Arabidopsis thaliana hypocotyl, a model to identify and study control mechanisms of cellular expansion. Plant Cell Rep 33:697–706
Briesemeister S, Rahnenführer J, Kohlbacher O (2010) Going from where to why-interpretable prediction of protein subcellular localization. Bioinformatics 26:1232–1238
Castillon A, Shen H, Huq E (2007) Phytochrome interacting factors: central players in phytochrome-mediated light signaling networks. Trends Plant Sci 12:514–521
Cavagnaro PF, Senalik DA, Yang LM, Simon PW, Harkins TT, Kodira CD, Huang SW, Weng Y (2010) Genome-wide characterization of simple sequence repeats in cucumber (Cucumis sativus L). BMC Genomics 11:569
Chen FF, Fu BB, Pan YP, Zhang CW, Wen HF, Weng YQ, Chen P, Li YH (2017) Fine mapping identifies CsGCN5 encoding a histone acetyltransferase as putative candidate gene for tendril-less1 mutation (td-1) in cucumber. Theor Appl Genet 130:1549–1558
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Derbyshire P, McCann MC, Roberts K (2007) Restricted cell elongation in Arabidopsis hypocotyls is associated with a reduced average pectin esterification level. BMC Plant Biology 7:31
Dong J, Chen HD, Deng XW, Irish VF, Wei N (2020) Phytochrome B induces intro retention and translational inhibition of phytochrome-interacting factor3. Plant Physiol 182:159–166
Du JB, Jiang HK, Sun X, Li Y, Liu Y, Sun MY, Fan Z et al (2018) Auxin and Gibberellins are required for the receptor-like kinase ERECTA regulated hypocotyl elongation in shade avoidance in Arabidopsis. Front Plant Sci 9:124
Eckardt NA (2007) GA perception and signal transduction: molecular interactions of the GA receptor GID1 with GA and the DELLA protein SLR1 in rice. Plant Cell 19:2095–2097
Fantini E, Sulli M, Zhang L, Aprea G, Jiménez-Gómez JM, Bendahmane A, Perrotta G, Giuliano G, Facella P (2019) Pivotal roles of cryptochromes 1a and 2 in tomato development and physiology. Plant Physiol 179:732–748
Ferrero LV, ViolaAriel ILFD, Gonzalez DH (2019) Class I TCP transcription factors target the gibberellin biosynthesis gene GA20ox1 and the growth-promoting genes HBI1 and PRE6 during thermomorphogenic growth in Arabidopsis. Plant Cell Physiol 60:1633–1645
Ferrero LV, Gastaldi V, Ariel FD, Viola IL, Gonzalez DH (2021) Class I TCP proteins TCP14 and TCP15 are required for elongation and gene expression responses to auxin. Plant Mol Biol 105:147–159
Foreman J, White JN, Graham IA, Halliday KJ, Josse EM (2011) Shedding light on flower development: Phytochrome B regulates gynoecium formation in association with the transcription factor spatula. Plant Signal Behav 6:471–476
Frankenberg N, Mukougawa K, Kohchi T, Lagarias JC (2001) Functional genomic analysis of the HY2 family of ferredoxin-dependent bilin reductases from oxygenic photosynthetic organisms. Plant Cell 13:965–978
Huai JL, Jing YJ, Lin RC (2020) Functional analysis of ZmCOP1 and ZmHY5 reveals conserved light signaling mechanism in maize and Arabidopsis. Physiol Plant 169:369–379
Izawa T, Oikawa T, Tokutomi S, Okuno K, Shimamoto K (2000) Phytochromes confer the photoperiodic control of flowering in rice (a short-day plant). Plant J 22:391–399
Jang IC, Henriques R, Chua NH (2013) Three transcription factors, HFR1, LAF1 and HY5, regulate largely independent signaling pathways downstream of phytochrome A. Plant Cell Physiol 54:907–916
Jiang HK, Shui ZW, Xu L, Yang YH, Li Y et al (2020) Gibberellins modulate shade-induced soybean hypocotyl elongation downstream of the mutual promotion of auxin and brassinosteroids. Plant Physiol Bioch 150:209–221
Josse EM, Halliday KJ (2008) Skotomorphogenesis: the dark side of light signalling. Curr Biol 18:R1144–R1146
Koornneef M, van der Knaap BJ (1983) A second long hypocotyl mutant at the lh Locus. Cucurbit Genet Coop Rpt 6:13
Kahle N, Sheerin DJ, Fischbach P, Koch LA, Schwenk P, Lambert D, Rodriguez R, Kerner K, Hoecker U, Zurbriggen MD, Hiltbrunner A (2020) COLD REGULATED 27 and 28 are targets of CONSTITUTIVELY PHOTOMORPHOGENIC 1 and negatively affect phytochrome B signalling. Plant J 104:1038–1053
Kohchi T, Mukougawa K, Frankenberg N, Masuda M, Yokota A, Lagarias J (2001) The arabidopsis HY2 gene encodes phytochromobilin synthase, a ferredoxin-dependent biliverdin reductase. Plant Cell 13:425–436
Kraepiel Y, Jullien M, Cordonnier-Pratt MM, Pratt L (1994) Identification of two loci involved in phytochrome expression in Nicotiana plumbaginifolia and lethality of the corresponding double mutant. Mol Gen Genet 242:559–565
López-Juez E, Nagatani A, Tomizawa KI, Deak M, Kern R, Kendrick RE, Furuya M (1992) The cucumber long hypocotyl mutant lacks a light-stable PHYB-like phytochrome. Plant Cell 4:241–251
Lai YS, Shen D, Zhang W, Zhang XH, Qiu Y, Wang HP et al (2018) Temperature and photoperiod changes affect cucumber sex expression by different epigenetic regulations. BMC Plant Biol 18:268
Li XY (2011) Infiltration of Nicotiana benthamiana protocol for transient expression via Agrobacterium. Bio-Protocol 1:1–3
Li JG, Li G, Wang HY, Deng XW (2011) Phytochrome signaling mechanisms. Arabidopsis Book 9:e0148
LiWen YHC, Weng Y (2013) Fine mapping of the pleiotropic locus B for black spine and orange mature fruit color in cucumber identifies a 50 kb region containing a R2R3-MYB transcription factor. Theor Appl Genet 126:2187–2196
Lin YJ, Chen YC, Tseng KC, Chang WC, Ko SS (2019) Phototropins mediate chloroplast movement in phalaenopsis aphrodite (Moth Orchid). Plant Cell Physiol 60:2243–2254
Linley PJ, Landsberger M, Kohchi T, Cooper JB, Terry MJ (2006) The molecular basis of heme oxyngenase deficiency in the pcd1 mutant of pea. FEBS J 273:2594–2606
Liu K, Li YH, Chen XN, Li LJ, Liu K, Zhao HP, Wang YD, Han SC (2018) ERF72 interacts with ARF6 and BZR1 to regulate hypocotyl elongation in Arabidopsis. J Exp Bot 69:3933–3947
Liu B, Long H, Yan J, Ye LL, Zhang Q, Chen HM et al (2021) A HY5-COL3-COL13 regulatory chain for controlling hypocotyl elongation in Arabidopsis. Plant Cell Environ 44:130–142
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCt method. Methods 25:402–408
Ming CH, Jiang FL, Hu HM, Zhou XC, Zhan FH, Wu Z (2011) Effects of different leggy extent seedling on cucumber growth, yield and quality. China Vegetables 4:29–34 ((In Chinese))
Montgomery BL (2008) Right place, right time: Spatiotemporal light regulation of plant growth and development. Plant Signal Behav 3:1053–1060
Montgomery BL (2009) Spatial-specific phytochrome responses during de-etiolation in Arabidopsis thaliana. Plant Signal Behav 4:47–49
Muramoto T, Kami C, Kataoka H, Iwata N, Linley PJ, Mukougawa K, Yokota A, Kohchi T (2005) The tomato photomorphogenetic mutant, aurea, is defect in phytochromobilin synthase for phytochrome chromophore biosynthesis. Plant Cell Physiol 46:661–665
Oh J, Park E, Song K, Bae G, Choi G (2020) Phytochrome interacting factor8 inhibits phytochrome a-mediated far-red light responses in Arabidopsis. The Plant Cell 32:186–205
Parks BM, Quail PH (1991) Phytochrome-defect hy1 and hy2 long hypocotyl mutants of Arabidopsis are defective in phytochrome chromophore biosynthesis. Plant Cell 3:1177–1186
Pham VN, Kathare PK, Huq E (2018) Phytochromes and phytochrome interacting factors. Plant Physiol 176:1025–1038
Qi JJ, Liu X, Shen D, Miao H, Xie BY, Li XX, Zeng P et al (2013) A genomic variation map provides insights into the genetic basis of cucumber domestication and diversity. Nat Genet 45(12):1510–1515
Qu XY, Zhao Z, Tian ZX (2017) ERECTA regulates cell elongation by activating auxin biosynthesis in Arabidopsis thaliana. Front Plant Sci 8:1688
Quail PH (2002) Phytochrome photosensory signalling networks. Nat Rev Mol Cell Biol 3:85–93
Ren Y, Zhang Z, Liu J, Staub JE, Han Y, Cheng Z, Li X et al (2009) An integrated genetic and cytogenetic map of the cucumber genome. PLoS ONE 4:e5795
Reig-valiente J, Borredá C, Talón M, Domingo C (2020) The G123 rice mutant, carrying a mutation in SE13, presents alterations in the expression patterns of photosynthetic and major flowering regulatory. PLoS One 15:e0233120
Robinson RW, Shail JW (1981) A cucumber mutant with increased hypocotyl and internode length. Cucurbit Genet Coop Rpt 4:19–20
Rockwell NC, Lagarias JC (2017) Ferredoxin-dependent bilin reductases in eukaryotic algae: Ubiquity and diversity. J Plant Physiol 217:57–67
Saito H, Okumoto Y, Yoshitake Y, Inoue H, Yuan Q, Teraishi M, Tsukiyama T, Nishida H, Tanisaka T (2011) Complete loss of photoperiodic response in the rice mutant line X61 is caused by deficiency of phytochrome chromophore biosynthesis gene. Theor Appl Genet 122:109–118
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sawers RJ, Linley PJ, Farmer PR, Hanley NP, Costich DE, Terry MJ, Brutnell TP (2002) Elongated mesocotyl1, a phytochrome-defect mutant of maize. Plant Physiol 130:155–163
Sawers RJ, Linley PJ, Gutierrez-Marcos JF, Delli-Bovi T, Farmer PR, Kohchi T, Terry M, Brutnell TP (2004) The Elm1 (ZmHy2) gene of maize encodes a phytochromobilin synthase. Plant Physiol 136:2771–2781
Serrani JC, Sanjuán R, Ruiz-Rivero O, Fos M, García-Martínez JL (2007) Gibberellin regulation of fruit set and growth in tomato. Plant Physiol 145:246–257
Seo DH, Yoon GM (2019) Light-induced stabilization of ACS contributes to hypocotyl elongation during the dark-to-light transition in Arabidopsis seedlings. Plant J 98:898–911
Song JL, Cao K, Hao YW, Song SW, Wei S, Liu HC (2019) Hypocotyl elongation is regulated by supplemental blue and red light in cucumber seedling. Gene 707:117–125
Song K, Choi G (2019) Phytochrome regulation of seed germination. Methods Mol Biol 2026:149–156
Sztatelman O, Łabuz J, Hermanowicz P, Banaś AK, Banaś A, Zgłobicki P, Aggarwal C, Nadzieja M, Krzeszowiec W, Strzałka W, Gabryś H (2016) Fine tuning chloroplast movements through physical interactions between phototropins. J Exp Bot 67:4963–4978
Tanaka R, Kobayashi K, Masuda T (2011) Tetrapyrrole metabolism in Arabidopsis thaliana. Arabidopsis Book 9:e0145
Tavridou E, Pireyre M, Ulm R (2020) Degradation of the transcription factors PIF4 and PIF5 under UV-B promotes UVR8-mediated inhibition of hypocotyl growth in Arabidopsis. Plant J 101:507–517
Terry MJ, Kendrick RE (1996) The aurea and yellow-green-2 mutants of tomato are defect in phytochrome chromophore synthesis. J Biol Chem 271:21681–21686
Wan H, Zhao Z, Qian C, Sui Y, Malik AA, Chen J (2010) Selection of appropriate reference genes for gene expression studies by quantitative real-time polymerase chain reaction in cucumber. Anal Biochem 399:257–261
Wang H, Li WQ, Qin YG, Pan YP, Wang XF, Weng YQ, Chen P, Li YH (2017) The cytochrome P450 gene CsCYP85A1 is a putative candidate for super compact-1 (Scp- 1) plant architecture mutation in cucumber Cucumis sativus L. Front Plant Sci 8:266
Weller JL, Terry MJ, Reid JB, Kendrick RE (1997) The phytochrome-defect pcd2 mutant of pea is unable to convert biliverdin IXα to 3 (Z)-phytochromobilin. Plant J 11:1177–1186
Wu JQ, Cheng J, Xu CM, Qi SL, Sun WR, Wu S (2020) AUREA maintains the balance between chlorophyll synthesis and adventitious root formation in tomato. Hortic Res 7:166
Yang Y, Zhang LB, Chen P, Liang T, Li X, Liu HT (2020) UV-B photoreceptor UVR8 interacts with MYB73/MYB77 to regulate auxin responses and lateral root development. EMBO J 39:e101928
Zhao C, Mao K, You CX, Zhao XY, Wang SH, Li YY, Hao YJ (2016) Molecular cloning and functional analysis of a UV-B photoreceptor gene, MdUVR8 (UV Resistance Locus 8), from apple. Plant Sci 247:115–126
Zhang WQ, Zhong H, Lu H, Zhang YX, Deng X et al (2019) Characterization of Ferredoxin-dependent biliverdin reductase PCYA1 reveals the dual function in retrograde bilin biosynthesis and interaction with light-dependent protochlorophyllide oxidoreductase LPOR in Chlamydomonas reinhardtii. Front Plant Sci 9:676
Zhou Y, Zhang DZ, An JX, Yin HJ, Fang S et al (2018) TCP transcription factors regulate shade avoidance via directly mediating the expression of both phytochrome interacting factors and auxin biosynthetic genes. Plant Physio 176:1850–1861
Zhu LX, Yang ZH, Zeng XH, Gao J, Liu J, Yi B et al (2017) Heme oxygenase 1 defects lead to reduced chlorophyll in Brassica napus. Plant Mol Biol 93:579–592
Acknowledgements
Work in AW’s laboratory was supported by the National Key Research and Development Program of China (2016YFD0101900).Work in PC’s and YL’s laboratory was supported by the National Natural Science Foundation of China under Project #31860557 and 31772300, respectively. YW’s laboratory was supported by the Agriculture and Food Research Initiative competitive grant no. 2017-67013-26195 of the USDA National Institute of Food and Agriculture.
Author information
Authors and Affiliations
Contributions
LH performed the research and prepared a draft of the manuscript. PL, ZJ and JS participated in the research. PC participated in data analysis and provided technical help. YW participated in data analysis and manuscript writing. AW and YL designed the experiments, supervised this study and wrote the manuscript. All authors have read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Communicated by Sanwen Huang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hu, L., Liu, P., Jin, Z. et al. A mutation in CsHY2 encoding a phytochromobilin (PΦB) synthase leads to an elongated hypocotyl 1(elh1) phenotype in cucumber (Cucumis sativus L.). Theor Appl Genet 134, 2639–2652 (2021). https://doi.org/10.1007/s00122-021-03849-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-021-03849-4