Abstract
Key message
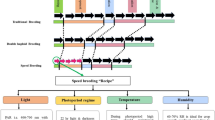
This study demonstrates that an active breeding nursery with rotation can be used to identify marker–trait associations for biomass yield and quality parameters that are important for biorefinery purposes.
Abstract
Wheat straw is a valuable feedstock for bioethanol production, but due to the recalcitrant nature of lignocellulose, its efficient use in biorefineries is limited by its low digestibility and difficult conversion of structural carbohydrates into free sugars. A genome-wide association study (GWAS) was conducted to search for significant SNP markers that could be used in a breeding programme to improve the value of wheat straw in a biorefinery setting. As part of a 3-year breeding programme (2013–2016), 190 winter wheat lines were phenotyped for traits that affect the yield and quality of the harvested biomass. These traits included straw yield, plant height, lodging at three growth stages and Septoria tritici blotch (STB) susceptibility. Release of glucose, xylose and arabinose was determined after hydrothermal pretreatment and enzymatic hydrolysis of the straw. The lines were genotyped using 15 K SNP markers and 5552 SNP markers could be used after filtering. Heritability for all traits ranged from 0.02 to 0.74. GWASs were conducted using CMLM, SUPER and FarmCPU algorithms, to analyse which algorithm could detect the highest number of marker–trait associations (MTAs). Comparable tendencies were obtained from CMLM and FarmCPU, but FarmCPU produced the most significant results. MTAs were obtained for lodging, harvest index, plant height, STB, glucose, xylose and arabinose at a significance level of p < 9.01 × 10−6. MTAs in chromosome 6A were observed for glucose, xylose and arabinose, and could be of importance for increasing sugar release for bioethanol production.






Similar content being viewed by others
References
Adhikari TB, Yang X, Cavaletto JR et al (2004) Molecular mapping of Stb1, a potentially durable gene for resistance to septoria tritici blotch in wheat. Theor Appl Genet 109:944–953. https://doi.org/10.1007/s00122-004-1709-6
Ain Q-U, Rasheed A, Anwar A et al (2015) Genome-wide association for grain yield under rainfed conditions in historical wheat cultivars from Pakistan. Front Plant Sci 6:743. https://doi.org/10.3389/fpls.2015.00743
Akhunov E, Nicolet C, Dvorak J (2009) Single nucleotide polymorphism genotyping in polyploid wheat with the Illumina GoldenGate assay. Theor Appl Genet 119:507–517. https://doi.org/10.1007/s00122-009-1059-5
Arraiano LS, Brown JKM (2017) Sources of resistance and susceptibility to Septoria tritici blotch of wheat. Mol Plant Pathol 18:276–292. https://doi.org/10.1111/mpp.12482
Assanga SO, Fuentealba M, Zhang G et al (2017) Mapping of quantitative trait loci for grain yield and its components in a US popular winter wheat TAM 111 using 90 K SNPs. PLoS ONE 12:e0189669. https://doi.org/10.1371/journal.pone.0189669
Bellucci A, Torp AM, Bruun S et al (2015) Association mapping in scandinavian winter wheat for yield, plant height, and traits important for second-generation bioethanol production. Front Plant Sci 6:1–12. https://doi.org/10.3389/fpls.2015.01046
Berry PM, Berry ST (2015) Understanding the genetic control of lodging-associated plant characters in winter wheat (Triticum aestivum L.). Euphytica. Springer, Berlin, pp 671–689. https://doi.org/10.1007/s10681-015-1387-2
Bordes J, Goudemand E, Duchalais L et al (2014) Genome-wide association mapping of three important traits using bread wheat elite breeding populations. Mol Breed 33:755–768. https://doi.org/10.1007/s11032-013-0004-0
Börner A, Plaschke J, Korzun V, Worland AJ (1996) The relationships between the dwarfing genes of wheat and rye. Euphytica 89:69–75. https://doi.org/10.1007/BF00015721
Brading PA, Verstappen CP, Kema GHJ, Brown JKM (2002) Genetics and resistance a gene-for-gene relationship between wheat and Mycosphaerella graminicola, the Septoria tritici blotch pathogen. Phytopathology 92:439. https://doi.org/10.1094/PHYTO.2002.92.4.439
Brown JKM, Chartrain L, Lasserre-Zuber P, Saintenac C (2015) Genetics of resistance to Zymoseptoria tritici and applications to wheat breeding. Fungal Genet Biol 79:33–41. https://doi.org/10.1016/j.fgb.2015.04.017
Cowling SG (2006) Identification and mapping of host resistance genes to Septoria tritici blotch of wheat. The University of Manitoba, Winnipeg
Crossa J, Burgueño J, Dreisigacker S et al (2007) Association analysis of historical bread wheat germplasm using additive genetic covariance of relatives and population structure. Genetics 177:1889–1913. https://doi.org/10.1534/genetics.107.078659
Dalgaard T, Hansen B, Hasler B et al (2014) Policies for agricultural nitrogen management-trends, challenges and prospects for improved efficiency in Denmark related content synthesis and review: tackling the nitrogen management challenge: from global to local scales. Environ Res Lett. https://doi.org/10.1088/1748-9326/9/11/115002
Edae EA, Byrne PF, Haley SD et al (2014) Genome-wide association mapping of yield and yield components of spring wheat under contrasting moisture regimes. Theor Appl Genet 127:791–807. https://doi.org/10.1007/s00122-013-2257-8
Edriss V, Cericola F, Orabi J et al (2017) Comparing single trait with multi-trait genome-wide association in winter wheat (Triticum aestivum). In: Buerstmayr H, Lang-Mladek C, Steiner B et al (eds) Proceedings 13th international wheat genetics symposium. BOKU - University of Natural Resources and Life Sciences, Vienna Department of Agrobiotechnology (IFA-Tulln) & Department of Crop Sciences, Tulln Austria, p 419
Gent MPN, Kiyomoto RK (1997) Physiological and agronomic consequences of Rht genes in wheat. J Crop Prod 1:27–46. https://doi.org/10.1300/J144v01n01_02
Giraldo P, Royo C, González M et al (2016) Genetic diversity and association mapping for agromorphological and grain quality traits of a structured collection of durum wheat landraces including subsp. durum, turgidum and diccocon. PLoS ONE 11:e0166577. https://doi.org/10.1371/journal.pone.0166577
Goodwin SB, Thompson I (2011) Development of isogenic lines for resistance to Septoria tritici blotch in wheat. Czech J Genet Plant Breed 47:S98–S101. https://doi.org/10.17221/3262-CJGPB
Griffiths S, Simmonds J, Leverington M et al (2012) Meta-QTL analysis of the genetic control of crop height in elite European winter wheat germplasm. Mol Breed 29:159–171. https://doi.org/10.1007/s11032-010-9534-x
Hao C, Wang L, Ge H et al (2011) Genetic diversity and linkage disequilibrium in chinese bread wheat (Triticum aestivum L.) revealed by SSR markers. PLoS ONE 6:e17279. https://doi.org/10.1371/journal.pone.0017279
Hedden P (2003) The genes of the green revolution. Trends Genet 19:5–9. https://doi.org/10.1016/S0168-9525(02)00009-4
Jørgensen H, van Hecke J, Zhang H et al (2018) Wheat as a dual crop for biorefining: straw quality parameters and their interactions with nitrogen supply in modern elite cultivars. GCB Bioenergy 11:400–411. https://doi.org/10.1111/gcbb.12560
Kaur S, Dhugga KS, Beech R, Singh J (2017) Genome-wide analysis of the cellulose synthase-like (Csl) gene family in bread wheat (Triticum aestivum L.). BMC Plant Biol 17:193. https://doi.org/10.1186/s12870-017-1142-z
Korzun V, Röder MS, Ganal MW et al (1998) Genetic analysis of the dwarfing gene (Rht8) in wheat. Part I. Molecular mapping of Rht8 on the short arm of chromosome 2D of bread wheat (Triticum aestivum L.). Theor Appl Genet 96:1104–1109. https://doi.org/10.1007/s001220050845
Leplat F, Jensen J, Madsen P (2016) Genomic prediction of manganese efficiency in winter barley. Plant Genome. https://doi.org/10.3835/plantgenome2015.09.0085
Lindedam J, Bruun S, Jørgensen H et al (2014) Evaluation of high throughput screening methods in picking up differences between cultivars of lignocellulosic biomass for ethanol production. Biomass Bioenerg 66:261–267. https://doi.org/10.1016/j.biombioe.2014.03.006
Lipka AE, Tian F, Wang Q et al (2012) GAPIT: genome association and prediction integrated tool. Bioinformatics 28:2397–2399. https://doi.org/10.1093/bioinformatics/bts444
Liu G, Xu SB, Ni Z et al (2011) Molecular dissection of plant height QTLs using recombinant inbred lines from hybrids between common wheat (Triticum aestivum L.) and spelt wheat (Triticum spelta L.). Chin Sci Bull 56:1897–1903. https://doi.org/10.1007/s11434-011-4506-z
Liu X, Huang M, Fan B et al (2016) Iterative usage of fixed and random effect models for powerful and efficient genome-wide association studies. PLoS Genet 12:1–24. https://doi.org/10.1371/journal.pgen.1005767
Oakey H, Shafiei R, Comadran J et al (2013) Identification of crop cultivars with consistently high lignocellulosic sugar release requires the use of appropriate statistical design and modelling. Biotechnol Biofuels 6:185. https://doi.org/10.1186/1754-6834-6-185
Ogbonnaya FC, Rasheed A, Okechukwu EC et al (2017) Genome-wide association study for agronomic and physiological traits in spring wheat evaluated in a range of heat prone environments. Theor Appl Genet 130:1819–1835. https://doi.org/10.1007/s00122-017-2927-z
Pearce S, Saville R, Vaughan SP et al (2011) Molecular characterization of Rht-1 dwarfing genes in hexaploid wheat. Plant Physiol 157:1820–1831. https://doi.org/10.1104/pp.111.183657
Pinthus MJ (1974) Lodging in wheat, barley, and oats: the phenomenon, its causes, and preventive measures. Adv Agron 25:209–263. https://doi.org/10.1016/S0065-2113(08)60782-8
Qaseem MF, Qureshi R, Muqaddasi QH et al (2018) Genome-wide association mapping in bread wheat subjected to independent and combined high temperature and drought stress. PLoS ONE 13:1–22. https://doi.org/10.1371/journal
Selig MJ, Tucker MP, Sykes RW et al (2010) Lignocellulose recalcitrance screening by integrated high-throughput hydrothermal pretreatment and enzymatic saccharification. Ind Biotechnol 6:104–111. https://doi.org/10.1089/ind.2010.0009
Studer MH, DeMartini JD, Brethauer S et al (2010) Engineering of a high-throughput screening system to identify cellulosic biomass, pretreatments, and enzyme formulations that enhance sugar release. Biotechnol Bioeng 105:231–238. https://doi.org/10.1002/bit.22527
Sukumaran S, Dreisigacker S, Lopes M et al (2015) Genome-wide association study for grain yield and related traits in an elite spring wheat population grown in temperate irrigated environments. Theor Appl Genet 128:353–363. https://doi.org/10.1007/s00122-014-2435-3
Thomas SG (2017) Novel Rht-1 dwarfing genes: tools for wheat breeding and dissecting the function of DELLA proteins. J Exp Bot 68:354–358. https://doi.org/10.1093/jxb/erw509
Townsend TJ, Roy J, Wilson P et al (2017) Food and bioenergy: exploring ideotype traits of a dual-purpose wheat cultivar. Field Crop Res 201:210–221. https://doi.org/10.1016/J.FCR.2016.11.007
Troyanskaya O, Cantor M, Sherlock G et al (2001) Missing value estimation methods for DNA microarrays. Bioinformatics 17:520–525. https://doi.org/10.1093/bioinformatics/17.6.520
Turuspekov Y, Baibulatova A, Yermekbayev K et al (2017) GWAS for plant growth stages and yield components in spring wheat (Triticum aestivum L.) harvested in three regions of Kazakhstan. BMC Plant Biol 17:52–61. https://doi.org/10.1186/s12870-017-1131-2
Vagndorf N, Nielsen NH, Edriss V et al (2017) Genomewide association study reveals novel quantitative trait loci associated with resistance towards Septoria tritici blotch in North European winter wheat. Plant Breed 136:474–482. https://doi.org/10.1111/pbr.12490
van Grinsven HJM, ten Berge HFM, Dalgaard T et al (2012) Management, regulation and environmental impacts of nitrogen fertilization in northwestern Europe under the Nitrates Directive; a benchmark study. Biogeosciences 9:5143–5160. https://doi.org/10.5194/bg-9-5143-2012
Verma V, Worland AJ, Sayers EJ et al (2005) Identification and characterization of quantitative trait loci related to lodging resistance and associated traits in bread wheat. Plant Breed 124:234–241. https://doi.org/10.1111/j.1439-0523.2005.01070.x
Visscher PM, Hill WG, Wray NR (2008) Heritability in the genomics era—concepts and misconceptions. Nat Rev Genet 9:255–266. https://doi.org/10.1038/nrg2322
Wang Q, Tian F, Pan Y et al (2014) A SUPER powerful method for genome wide association study. PLoS ONE 9:e107684. https://doi.org/10.1371/journal.pone.0107684
Würschum T, Langer SM, Longin CFH (2015) Genetic control of plant height in European winter wheat cultivars. Theor Appl Genet 128:865–874. https://doi.org/10.1007/s00122-015-2476-2
Zanke CD, Ling J, Rg Plieske J et al (2014) Whole genome association mapping of plant height in winter wheat (Triticum aestivum L). PLoS ONE 9:e113287. https://doi.org/10.1371/journal.pone.0113287
Zhang K-P, Zhao L, Hai Y et al (2008) QTL mapping for adult-plant resistance to powdery mildew, lodging resistance, and internode length below spike in wheat. Acta Agron Sin 34:1350–1357. https://doi.org/10.1016/S1875-2780(08)60046-2
Zhang Z, Ersoz E, Lai C-Q et al (2010) Mixed linear model approach adapted for genome-wide association studies. Nat Genet 42:355–360. https://doi.org/10.1038/ng.546
Zhang L, Xu X, Zhao C et al (2011) QTL analysis of plant height based on doubled haploid (DH) population derived from PTSMS wheat. Mol Plant Breed 2:92–97. https://doi.org/10.5376/mpb.2011.02.0013
Zhang H, Fangel JU, Willats WGT et al (2014) Assessment of leaf/stem ratio in wheat straw feedstock and impact on enzymatic conversion. GCB Bioenergy 6:90–96. https://doi.org/10.1111/gcbb.12060
Acknowledgements
We thank Morten Læssøe Stephensen from the University of Copenhagen and Ole Andersen, Erling and Helle Hisselholm, and other employees at Sejet Plant Breeding A/S for their help with plant harvests and the recording of phenotypic data. Britta Skov and Anja Hecht Ivø of the University of Copenhagen are acknowledged for their technical support with the laboratory analyses. The project was funded by Sejet Plant Breeding A/S and Innovation Fund Denmark, Grant 12-132625 (biovalue: value-added products from biomass to J.K.S.).
Author information
Authors and Affiliations
Contributions
PLM, JKS, BE and SKR conceived and designed the experiments; LKN, FB and BE designed and carried out the field experiments, conducted phenotyping and provided SNP marker data; PLM performed all the experiments; PLM analysed the data with the contribution of LJ; PLM drafted and finalized the article with writing contributions from all the authors; the final article was approved by all the authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Not applicable.
Additional information
Communicated by Mark E. Sorrells.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Malik, P.L., Janss, L., Nielsen, L.K. et al. Breeding for dual-purpose wheat varieties using marker–trait associations for biomass yield and quality traits. Theor Appl Genet 132, 3375–3398 (2019). https://doi.org/10.1007/s00122-019-03431-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-019-03431-z