Abstract
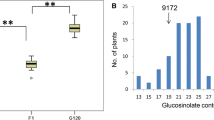
Seed glucosinolate content in Brassica juncea is a complex quantitative trait. A recurrent selection backcross (RSB) method with a doubled haploid (DH) generation interspersing backcross generations was used for the introgression of low glucosinolate alleles from an east European gene pool B. juncea line, Heera into an Indian gene pool variety, Varuna. Phenotypic comparisons among the DH populations derived from early to advanced backcrosses revealed a shift in the mean values for various glucosinolates with the advancement of backcrossing, indicating a change in the selective values of the alleles with change in the genetic background due to the existence of epistasis and context dependencies. QTL mapping for various seed glucosinolates from early (F1DH) and advanced generation (BC4DH) populations confirmed the presence of epistasis and context dependency. The common QTL detected in both F1DH and BC4DH changed their R 2 values from the former to the later generation. Some of the QTL detected in the F1DH became irrelevant in the BC4DH population. Further, new QTL were detected in the BC4DH population for various glucosinolates. A validation study on a population of low glucosinolate DH lines derived from all the backcross generations of the RSB breeding programme revealed that the QTL detected in BC4DH were the ‘true’ QTL. Using glucosinolate as an example, the study provides strong evidence for the importance of the RSB method for the identification of the ‘true’ QTL which would be significant for marker assisted introgression of a complex quantitative trait whose expression is influenced by epistatic interactions.


Similar content being viewed by others
References
Boer MP, ter Braak CJF, Jansen RC (2002) A penalized likelihood method for mapping epistatic quantitative trait loci with one-dimensional genome searches. Genetics 162:951–960
Cheung WY, Landry BS, Raney P, Rakow GFW (1998) Molecular mapping of seed quality traits in Brassica juncea L. Czern and Cross. Acta Hortic 459:139–147
Churchill GA, Doerge RW (1994) Empirical threshold values for quantitative trait mapping. Genetics 138:963–971
Doerge RW, Churchill GA (1996) Permutation test for multiple loci affecting a quantitative character. Genetics 142:285–294
Gao M, Li G, Yang B, Qiu D, Farnham M, Quiros C (2007) High-density Brassica oleracea linkage map: identification of useful new linkages. Theor Appl Genet 115:277–287
Gland A, Röbbelen G, Thies W (1981) Variation of alkenyl glucosinolates in seeds of Brassica species. Z Pflanzenzüchtg 87:96–110
Halkier BA, Gershenzon J (2006) Biology and biochemistry of glucosinolates. Annu Rev Plant Biol 57:303–333
Hill WG (1998) Selection and recurrent backcrossing to develop congenic lines for quantitative trait loci analysis. Genetics 148:1341–1352
Hospital F (2005) Selection in backcross programmes. Phil Trans R Soc B 360:1503–1511
Jansen R, Yu H, Greenbaum D, Kluger Y, Krogan NJ, Chung S, Emili A, Snyder M, Greenblatt JF, Gerstein M (2003) A Bayesian networks approach for predicting protein–protein interactions from genomic data. Science 302:449–453
Kraling K, Robbelen G, Thies W, Herrmann M, Ahmadi MR (1990) Variation in seed glucosinolates in lines of Brassica napus. Plant Breed 105:33–39
Lecomte L, Duffe P, Buret M, Servin B, Hospital F, Causse M (2004) Marker-assisted introgression of five QTLs controlling fruit quality traits into three tomato lines revealed interactions between QTLs and genetic backgrounds. Theor Appl Genet 109:658–668
Lionneton E, Aubert G, Ochatt S, Merah O (2004) Genetic analysis of agronomic and quality traits in mustard (Brassica juncea). Theor Appl Genet 109:792–799
Love HR, Rakow G, Raney JP, Downey RK (1990a) Development of low glucosinolate mustard. Can J Plant Sci 70:419–424
Love HR, Rakow G, Raney JP, Downey RK (1990b) Genetic control of 2-propenyl and 3-butenyl glucosinolate synthesis in mustard. Can J Plant Sci 70:425–429
Luo ZW, Ma L (2004) An improved formulation of marker heterozygosity in recurrent selection and backcross schemes. Genet Res Camb 83:49–53
Luo ZW, Wu CI, Kearsey MJ (2002) Precision and high-resolution mapping of quantitative trait loci by use of recurrent selection, backcross or intercross schemes. Genetics 161:915–929
Magrath R, Herron C, Giamoustaris A, Mithen R (1993) The inheritance of aliphatic glucosinolates in Brassica napus. Plant Breed 111:55–72
Mahmood T, Ekuere U, Yeh F, Good AG, Stringam GR (2003) Molecular mapping of seed aliphatic glucosinolates in Brassica juncea. Genome 46:753–760
Mukhopadhyay A, Arumugam N, Sodhi YS, Gupta V, Pradhan AK, Pental D (2007) High frequency production of microspore derived doubled haploid (DH) and its application for developing low glucosinolate lines in Indian Brassica juncea. In: Proceedings of the 12th International Rapeseed Congr, Wuhan, pp 333–335
Pradhan AK, Sodhi YS, Mukhopadhyay A, Pental D (1993) Heterosis breeding in Indian mustard (Brassica juncea L. Czern & Cross): analysis of component characters contributing to heterosis for yield. Euphytica 69:219–229
Pradhan AK, Gupta V, Mukhopadhyay A, Arumugam N, Sodhi YS, Pental D (2003) A high density linkage map in Brassica juncea (Indian mustard) using AFLP and RFLP markers. Theor Appl Genet 106:607–614
Quijada PA, Udall JA, Lambert B, Osborn TC (2006) Quantitative trait analysis of seed yield and other complex traits in hybrid spring rapeseed (Brassica napus L.): identification of genomic regions from winter germplasm. Theor Appl Genet 113:549–561
Ramchiary N, Padmaja KL, Sharma S, Gupta V, Sodhi YS, Mukhopadhyay A, Arumugam N, Pental D, Pradhan AK (2007) Mapping of yield influencing QTL in Brassica juncea: implications for breeding of major oilseed crop of dryland areas. Theor Appl Genet. doi:10.1007/s00122-007-0610-5
Rogers SO, Bendich AJ (1994) Extraction of total cellular DNA from plants, algae and fungi. In: Gelvin SB, Shilperoot RA (eds) Plant molecular biology manual. Kluwer, Dordrecht, pp 1–8
Sebolt AM, Shoemaker RC, Diers BW (2000) Analysis of a quantitative trait locus allele from wild soybean that increases seed protein concentration in soybean. Crop Sci 40:1438–1444
Shen L, Courtois B, McNally KL, Robin S, Li Z (2001) Evaluation of near isogenic lines rice introgressed with QTLs for root depth through marker-aided selection. Theor Appl Genet 103:75–83
Sodhi YS, Mukhopadhyay A., Arumugam N, Verma JK, Gupta V, Pental D, Pradhan AK (2002) Genetic analysis of total glucosinolate in crosses involving a high glucosinolate Indian variety and a low glucosinolate line of Brassica juncea. Plant Breed 121:508–511
Srivastava A, Gupta V, Pental D, Pradhan AK (2000) AFLP-based genetic diversity assessment amongst agronomically important natural and some newly synthesized lines of Brassica juncea. Theor Appl Genet 102:193–199
Stringam GR, Thiagarajah MR (1995) Inheritance of alkenyl glucosinolates in traditional and microspore-derived doubled haploid populations of Brassica juncea L. Czern & Cross. In: Proceedings of the 9th International Rapeseed Congr, Cambridge, pp 804–806
van Eeuwijk FA, Crossa J, Vargas M, Ribaut J-M (2002) Analyzing QTL by environment interaction by factorial regression, with an application to the CIMMYT drought and low nitrogen stress programme in maize. In: Kang MS (ed) Quantitative genetics, genomics and plant breeding. CAB International, Wallingford, pp 245–256
Wang S, Basten CJ, Gaffney P, Zeng ZB (2001–2005) Windows QTL Cartographer version 2.5. North Carolina State University, Bioinformatics Research Center, Raleigh
Wright S (1952) Quantitative inheritance. Her Majesty’s Stationery Office, London
Zeng ZB (1993) Theoretical basis for separation of multiple linked gene effects in mapping quantitative trait loci. Proc Natl Acad Sci USA 90:10972–10976
Zeng ZB (1994) Precision mapping of quantitative trait loci. Genetics 136:1457–1468
Acknowledgments
This work was supported by the Dhara Vegetable Oil and Food Company Ltd (DOFCO), a fully owned company of the National Dairy Development Board (NDDB) and the Department of Biotechnology (DBT). N. Ramchiary acknowledges the receipt of a fellowship from Council of Scientific and Industrial Research (CSIR), Government of India.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. F. Quiros.
Authors N. Ramchiary, N. C. Bisht, V. Gupta, A. Mukhopadhyay and N. Arumugam have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ramchiary, N., Bisht, N.C., Gupta, V. et al. QTL analysis reveals context-dependent loci for seed glucosinolate trait in the oilseed Brassica juncea: importance of recurrent selection backcross scheme for the identification of ‘true’ QTL. Theor Appl Genet 116, 77–85 (2007). https://doi.org/10.1007/s00122-007-0648-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-007-0648-4