Abstract
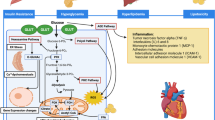
Epidemiologic as well as clinical studies confirm the close link between diabetes mellitus and heart failure. Diabetic cardiomyopathy (DCM) is still a poorly understood “entity”, however, with several contributing pathogenetic factors which lead in different stages of diabetes to characteristic clinical phenotypes. Hyperglycemia with a shift from glucose metabolism to increased beta-oxidation and consecutive free fatty acid damage (lipotoxicity) to the myocardium, insulin resistance, renin-angiotensin-aldosterone system (RAAS) activation, altered calcium homeostasis and structural changes from the natural collagen network to a stiffer matrix due to advanced glycation endproduct (AGE) formation, hypertrophy and fibrosis contribute to the respective clinical phenotypes of DCM.
We propose the following classification of cardiomyopathy in diabetic patients:
-
a)
Diastolic heart failure with normal ejection fraction (HFNEF) in diabetic patients often associated with hypertrophy without relevant hypertension. Relevant coronary artery disease (CAD), valvular disease and uncontrolled hypertension are not present. This is referred to as stage 1 DCM.
-
b)
Systolic and diastolic heart failure with dilatation and reduced ejection (HFREF) in diabetic patients excluding relevant CAD, valvular disease and uncontrolled hypertension as stage 2 DCM.
-
c)
Systolic and/or diastolic heart failure in diabetic patients with small vessel disease (microvascular disease) and/or microbial infection and/or inflammation and/or hypertension but without CAD as stage 3 DCM.
-
d)
If heart failure may also be attributed to infarction or ischemia and remodeling in addition to stage 3 DCM the term should be heart failure in diabetes or stage 4 DCM.
These clinical phenotypes of diabetic cardiomyopathy can be separated by biomarkers, non-invasive (echocardiography, cardiac magnetic resonance imaging) and invasive imaging methods (levocardiography, coronary angiography) and further analysed by endomyocardial biopsy for concomitant viral infection. The role of specific diabetic drivers to the clinical phenotypes, to macro- and microangiopathy as well as accompanying risk factors or confounders, e.g. hypertension, autoimmune factors or inflammation with or without viral persistence, need to be identified in each individual patient separately. Thus hyperglycemia, hyperinsulinemia and insulin resistance as well as lipotoxicity by free fatty acids (FFAs) are the factors responsible for diabetic cardiomyopathy. In stage 1 and 2 DCM diabetic cardiomyopathy is clearly a fact. However, precise determination of to what degree the various underlying pathogenetic processes are responsible for the overall heart failure phenotype remains a fiction.
Zusammenfassung
Diabetes mellitus und Herzinsuffizienz treten häufig gemeinsam auf. Aus der Koinzidenz wird mit dem Begriff diabetische Kardiomyopathie (DCM) ein Zusammenhang beschrieben, der hinsichtlich Ätiologie, Pathogenese und klinischem Phänotyp bislang nur unzureichend präzisiert ist. Hyperglykämie bei gleichzeitiger Insulinresistenz führt zu einer Hochregulation der β-Oxidation. Freie Fettsäuren (FFA) decken bis zu 90% des myokardialen Energiebedarfs, anders als beim Gesunden, bei dem der Anteil der FFA nur 50% ausmacht. Die intramyokardiale FFA-Akkumulation führt zu Lipotoxizität und zu einer gestörten Kalziumhomöostase, die Bindung der FFAs an interstitielles und perivaskulären Kollagen zu „advanced glycation endproducts“ (AGE) und damit zu vermehrter Steifigkeit, Hypertrophie und additiver Fibrose. Der Diabetes mellitus umfasst als weitere Folge mikrovaskuläre Veränderungen („small vessel disease“) sowie eine Prädisposition für Infektionen und autoimmune, inflammatorische Prozesse, die den Phänotyp einer DCM ebenso prägen können wie eine begleitende arterielle Hypertonie oder eine koronare Herzerkrankung.
Wir schlagen eine Klassifikation vor, die der Pathogenese, der zeitlichen Entwicklung und dem klinischen wie hämodynamischen Phänotyp Rechnung trägt:
DCM-Stufe 1: Diastolische Herzinsuffizienz bei Diabetespatienten mit normaler Ejektionsfraktion („heart failure with normal ejection fraction“, HFNEF). Hier überwiegt häufig eine Hypertrophie ohne Hochdruck.
DCM-Stufe 2: Systolische und diastolische Herzinsuffizienz mit optionaler linksventrikulärer Dilatation, aber reduzierter Ejektionsfraktion (<50%) als HFREF („heart failure with reduced ejection fraction“).
DCM-Stufe 3: Systolische und diastolische Herzinsuffizienz (wie Stufe 2), aber mit mehreren additiven, nur indirekt diabetesvermittelten Begleiterkrankungen, wie Mikrovaskulopathie, myokardialer Infektion/Inflammation oder arterieller Hypertonie.
DCM-Stufe 4: Herzinsuffizienz bei Patienten mit Diabetes mellitus, d. h. DCM-Stufe 3, zusätzlich eine koronare Herzerkrankung mit Infarkt und Remodelling.
Die Charakterisierung der vier DCM-Formen erfolgt mit den serologischen Biomarkern der Herzinsuffizienz (BNP, NTproBNP), der Nekrose (Troponine) und der Fibrose (MMPs und TIMPs), mit nichtinvasiver Bildgebung (Echokardiographie, Doppler/TDI, Kardio-MRT) und invasiver Katheterdiagnostik einschließlich Koronarangiographie und Endomyokardbiopsie. Die DCM ist für die Formen 1 und 2 etablierter Fakt. Doch den jeweiligen Anteil pathogenetischer Faktoren aller 4 DCM-Stufen an der Herzinsuffizienz/Kardiomyopathie des jeweiligen Patienten mit diesen Methoden quantitativ bestimmen zu können, bleibt eine Fiktion.









Similar content being viewed by others
References
Abe T, Ohga Y, Tabayashi N et al (2002) Left ventricular diastolic dysfunction in type 2 diabetes mellitus model rats. Am J Physiol Heart Circ Physiol 282:H138–H148
Achenbach S, Ropers D, Regenfus M et al (2001) Noninvasive coronary angiography by magnetic resonance imaging, electron-beam computed tomography, and multislice computed tomography. Am J Cardiol 88(2A):70E–73E
Alter P, Grimm W, Vollrath A et al (2006) Heart rate variability in patients with cardiac hypertrophy – relation to left ventricular mass and etiology. Am Heart J 151(4):829–836
Alter P, Rupp H, Rominger MB et al (2010) Late gadolinium enhancement is associated with increased left ventricular wall stress and mass in patients with non-ischemic cardiomyopathy. Eur Heart J 31(Supp 1):282
Alter P, Rupp H, Rominger MB et al (2008) B-type natriuretic peptide and wall stress in dilated human heart. Mol Cell Biochem 314(1–2):179–191
Alter P, Rupp H, Rominger MB et al (2008) A new methodological approach to assess cardiac work by pressure-volume and stress-length relations in patients with aortic valve stenosis and dilated cardiomyopathy. Pfluegers Arch 455(4):627–636
Alter P, Rupp H, Rominger MB et al (2007) Relation of B-type natriuretic peptide to left ventricular wall stress as assessed by cardiac magnetic resonance imaging in patients with dilated cardiomyopathy. Can J Physiol Pharmacol 85(8):790–799
Alter P, Rupp H, Maisch B (2008) Assessment and relevance of ventricular wall stress in heart failure. Eur Heart J 29(18):2316
Alter P, Rupp H, Stoll F et al (2010) Left ventricular hypertrophy follows increased enddiastolic wall stress in patients with dilative heart failure. Clin Res Cardiol 99(Suppl 2). DOI: 10.1007/s00392-010-1200-0
Aneja A, Tang WH, Bansilal S et al (2008) Diabetic cardiomyopathy: insights into pathogenesis, diagnostic challenges, and therapeutic options. Am J Med 121(9):748–757
Argirova MD, Ortwerth BJ (2003) Activation of protein-bound copper ions during early glycation: study on two proteins. Arch Biochem Biophys 420:176–184
Aronow WS, Ahn C (1999) Incidence of heart failure in 2,737 older persons with and without diabetes mellitus. Chest 115:867–868
Bell DS (2003) Heart failure: the frequent, forgotten, and often fatal complication of diabetes. Diabetes Care 26(8):2433–2441
Bella JN, Devereux RB, Roman MJ et al (2001) Separate and joint effects of systemic hypertension and diabetes mellitus on left ventricular structure and function in American Indians (the Strong Heart Study). Am J Cardiol 87:1260–1265
Bertoni AG, Goff DC Jr, D’Agostino RB Jr et al (2006) Diabetic cardiomyopathy and subclinical cardiovascular disease: the Multi-Ethnic Study of Atherosclerosis (MESA). Diabetes Care 29(3):588–594
Bertoni AG, Tsai A, Kasper EK, Brancati FL (2003) Diabetes and idiopathic cardiomyopathy: a nationwide case-control study. Diabetes Care 26:2791–2795
Boudina S, Abel ED (2006) Mitochondrial uncoupling: a key contributor to reduced cardiac efficiency in diabetes. Physiology 21:250–258
Boudina S, Abel ED (2007) Diabetic cardiomyopathy revisited. Circulation 115:3213–3223
Boudina S, Abel ED (2010) Diabetic cardiomyopathy, causes and effects. Rev Endocr Metab Disord 11:31–39
Boyer JK, Thanigaraj S, Schechtman KB et al (2004) Prevalence of ventricular diastolic dysfunction in asymptomatic, normotensive patients with diabetes mellitus. Am J Cardiol 93:870–875
Brilla CG (2000) Regression of myocardial fibrosis in hypertensive heart disease: diverse effects of various antihypertensive drugs. Cardiovasc Res 46:324–331
Brilla CG, Funck RC, Rupp H (2000) Lisinopril-mediated regression of myocardial fibrosis in patients with hypertensive heart disease. Circulation 102:1388–1393
Cai L, Wang Y, Zhou G et al (2006) Attenuation by metallothionein of early cardiac cell death via suppression of mitochondrial oxidative stress results in a prevention of diabetic cardiomyopathy. J Am Coll Cardiol 48:1688–1697
Charoenpanichkit C, Morgan TM, Hamilton CA et al (2010) Left ventricular hypertrophy influences cardiac prognosis in patients undergoing dobutamine cardiac stress testing. Circ Cardiovasc Imaging 3(4):392–397
Dhalla NS, Liu X, Panagia V, Takeda N (1998) Subcellular remodeling and heart dysfunction in chronic diabetes. Cardiovasc Res 40(2):239–247
Devereux RB, Roman MJ, Paranicas M et al (2000) Impact of diabetes on cardiac structure and function: the strong heart study. Circulation 101(19):2271–2276
Eckel J, Reinauer H (1990) Insulin action on glucose transport in isolated cardiac myocytes: signalling pathways and diabetes-induced alterations. Biochem Soc Trans 18:1125–1127
Elliott P, Andersson B, Arbustini E et al (2008) Classification of the cardiomyopathies: a position statement from the European Society Of Cardiology Working Group on Myocardial and Pericardial Diseases. 29(2):270–276
Fang ZY, Prins JB, Marwick TH (2004) Diabetic cardiomyopathy: evidence, mechanisms, and therapeutic implications. Endocr Rev 25(4):543–567
Fiordaliso F, Leri A, Cesselli D et al (2001) Hyperglycemia activates p53 and p53-regulated genes leading to myocyte cell death. Diabetes 50:2363–2375
Gebker R, Mirelis JG, Jahnke C et al (2010) Influence of left ventricular hypertrophy and geometry on diagnostic accuracy of wall motion and perfusion magnetic resonance during dobutamine stress. Circ Cardiovasc Imaging 3(5):507–514
Ha JW, Lee HC, Kang ES et al (2007) Abnormal left ventricular longitudinal functional reserve in patients with diabetes mellitus: implication for detecting subclinical myocardial dysfunction using exercise tissue Doppler echocardiography. Heart 93(12):1571–1576
Havers J, Haude M, Erbel R, Spiller P (2008) Röntgendensitometische Messung der myokardialen Perfusionsreserve bei symptomatischen Patienten ohne angiographisch detektierbare Koronarstenosen. Herz 33:223–232
Herrero P, Peterson LR, McGill JB et al (2006) Increased myocardial fatty acid metabolism in patients with type 1 diabetes mellitus. J Am Coll Cardiol 47:598–604
Hunt SA, American College of Cardiology, American Heart Association Task Force on Practice Guidelines (Writing Committee to Update the 2001 Guidelines for the Evaluation, Management of Heart Failure) (2005) ACC/AHA 2005 guideline update for the diagnosis and management of chronic heart failure in the adult: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Update the 2001 Guidelines for the Evaluation and Management of Heart Failure). JACC 46(6):e1–e82
Iltis I, Kober F, Desrois M et al (2005) Defective myocardial blood flow and altered function of the left ventricle in type 2 diabetic rats: a noninvasive in vivo study using perfusion and cine magnetic resonance imaging. Invest Radiol 40(1):19–26
Iribarren C, Karter AJ, Go AS et al (2001) Glycemic control and heart failure among adult patients with diabetes. Circulation 103:2668–2673
Islam KN, Takahashi M, Higashiyama S et al (1995) Fragmentation of ceruloplasmin following nonenzymatic glycation reaction. J Biochem 118:1054–1060
Jahnke C, Nagel E, Gebker R et al (2007) Prognostic value of cardiac magnetic resonance stress tests: adenosine stress perfusion and dobutamine stress wall motion imaging. Circulation 115(13):1769–1776
Kannel WB, McGee DL (1979) Diabetes and cardiovascular disease: the Framingham study. JAMA 241:2035–2038
Khavandi K, Khavandi A, Asghar O et al (2009) Diabetic cardiomyopathy-a distinct disease? Best Pract Res Clin Endocrinol Metab 23(3):347–360
Liang Q, Carlson EC, Donthi RV et al (2002) Overexpression of metallothionein reduces diabetic cardiomyopathy. Diabetes 51:174–181
Liedtke AJ, DeMaison L, Eggleston AM et al (1988) Changes in substrate metabolism and effects of excess fatty acids in reperfused myocardium. Circ Res 62:535–542
Liu GX, Hanley PJ, Ray J, Daut J (2001) Long-chain acylcoenzyme A esters and fatty acids directly link metabolism to K (ATP) channels in the heart. Circ Res 88(9):918–924
Lundbaek K (1954) Diabetic angiopathy: a specific vascular disease. Lancet 266:377–379
Ma H, Li AY, Xu P et al (2009) Advanced glycation endproduct (AGE) accumulation and AGE receptor (RAGE) upregulation contribute to the onset of diabetic cardiomyopathy. J Cell Mol Med 13(8B):1751–1764
Maisch B (2006) Obesity, diabetes mellitus and metabolic syndrome: the implications for heart and the vascular system. Herz 31(3):185–188
Maisch B, Hufnagel G, Kölsch S et al (2004) Treatment of inflammatory dilated cardiomyopathy and myocarditis with immunosuppression and i.v. immunoglobulins. Herz 29:624–636
Maisch B, Pankuweit S, Karatolios K, Ristić AD (2006) Invasive techniques – from diagnosis to treatment. Rheumatology (Oxford) 45(Suppl 4):iv32–iv38
Maisch B, Richter A, Koelsch S et al (2006) Management of patients with suspected (peri-)myocarditis and inflammatory dilated cardiomyopathy. Herz 31(9):881–890
Maisch B, Rupp H (2006) Myocardial fibrosis: a cardiopathophysiologic Janus head. Herz 31(3):260–268
Maisch B, Seferovic PM, Ristic AD et al (2004) The Task Force on the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology. Guidelines on the diagnosis and management of pericardial diseases executive summary; The Task force on the diagnosis and management of pericardial diseases of the European Society of Cardiology Eur Heart J 25(7):587–610
Maksimović R, Dill T, Seferović PM et al (2006) Magnetic resonance imaging in pericardial diseases. Indications and diagnostic value. Herz 31(7):708–714
Malhotra A, Sanghi V (1997) Regulation of contractile proteins in diabetic heart. Cardiovasc Res 34:34–40
Matsushima S, Kinugawa S, Ide T et al (2006) Overexpression of glutathione peroxidase attenuates myocardial remodeling and preserves diastolic function in diabetic heart. Am J Physiol Heart Circ Physiol 291:H2237–H2245
Maya L, Villarreal FJ (2010) Diagnostic approaches for diabetic cardiomyopathy and myocardial fibrosis. J Mol Cell Cardiol 48:524–529
McGavock JM, Lingvay I, Zib I et al (2007) Cardiac steatosis in diabetes mellitus: a 1H-magnetic resonance spectroscopy study. Circulation 116:1170–1175
Murray AG, Anderson RE, Watson GC et al (2004) Uncoupling proteins in human heart. Lancet 364:1786–1788
Nagel E, Lorenz C, Baer F et al (2001) Stress cardiovascular magnetic resonance: consensus panel report. J Cardiovasc Magn Reson 3(3):267–281
Neubauer S, Krahe T, Schindler R et al (1992) 31P magnetic resonance spectroscopy in dilated cardiomyopathy and coronary artery disease. Altered cardiac high-energy phosphate metabolism in heart failure. Circulation 86(6):1810–1818
Neubauer S, Horn M, Cramer M et al (1997) Myocardial phosphocreatine-to-ATP ratio is a predictor of mortality in patients with dilated cardiomyopathy. Circulation 96(7):2190–2196
Neubauer S (2007) The failing heart-an engine out of fuel. N Engl J Med 356(11):1140–1151
Ng ACT, Delgado V, Bertini M et al (2009) Findings from left ventricular strain and strain rate imaging in asymptomatic patients with type 2 diabetes mellitus. Am J Cardiol 104:1398–1401
Nichols GA, Hillier TA, Erbey JR, Brown JB (2001) Congestive heart failure in type 2 diabetes: prevalence, incidence, and risk factors. Diabetes Care 24:1614–1619
Palmieri V, Capaldo B, Russo C et al (2008) Uncomplicated type 1 diabetes and preclinical left ventricular myocardial dysfunction: insights from echocardiography and exercise cardiac performance evaluation. Diabetes Res Clin Pract 79(2):262–268
Pankuweit S, Funck R, Grimm W, Maisch B (2006) Diagnosis and treatment of inflammatory heart diseases: role of endomyocardial biopsy. Herz 31(4):361–365
Pankuweit S, Ruppert V, Maisch B (2004) Inflammation in dilated cardiomyopathy. Herz 29(8):788–793
Paulus WJ, Tschöpe C, Sanderson JE et al (2007) How to diagnose diastolic heart failure: a consensus statement on the diagnosis of heart failure with normal left ventricular ejection fraction by the Heart Failure and Echocardiography Associations of the European Society of Cardiology. Eur Heart J 28(20):2539–2550
Peterson LR, Herrero P, Schechtman KB et al (2004) Effect of obesity and insulin resistance on myocardial substrate metabolism and efficiency in young women. Circulation 109:2191–2196
Pogatsa G (2001) Metabolic energy metabolism in diabetes: therapeutic implications. Coron Artery Dis 12(Suppl 1):S29–S33
Portig I, Maisch B (2004) Noninvasive methods in the diagnosis of macro- and microangiopathy of peripherial and carotid arteries. Herz 29(1):17–25
Regan TJ, Lyons MM, Ahmed SS et al (1977) Evidence for cardiomyopathy in familial diabetes mellitus. J Clin Invest 60(4):884–899
Richardson P, McKenna W, Bristow M et al (1996) Report of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the Definition and Classification of cardiomyopathies. Circulation 93(5):841–842
Rijzewijk LJ, Meer RW van der, Lamb HJ et al (2009) Altered myocardial substrate metabolism and decreased diastolic function in nonischemic human diabetic cardiomyopathy: studies with cardiac positron emission tomography and magnetic resonance imaging. J Am Coll Cardiol 54:1524–1532
Rodrigues B, Camm MC, Mcneill GH (1998) Metabolic disturbances in diabetic cardiomyopathy. Mol Cell Biochem 180:53–57
Rubler S, Dlugash J, Yuceoglu YZ et al (1972) New type of cardiomyopathy associated with diabetic glomerulosclerosis. Am J Cardiol 30(6):595–602
Ryden L, Standl E, Bartnik M et al (2007) Guidelines on diabetes, pre-diabetes, and cardiovascular diseases: executive summary. The Task Force on Diabetes and Cardiovascular Diseases of the European Society of Cardiology (ESC) and of the European Association for the Study of Diabetes (EASD). Eur Heart J 28(1):88–136
Schneider CA, Pfister R, Erdmann E (2010) Diabetes and heart failure. Herz 35(3):140–146
Scognamiglio R, Avogaro A, Casara D et al (1998) Myocardial dysfunction and adrenergic cardiac innervations in patients with insulin-dependent diabetes mellitus. J Am Coll Cardiol 31(2):404–412
Shen X, Zheng S, Metreveli NS, Epstein PN (2006) Protection of cardiac mitochondria by overexpression of MnSOD reduces diabetic cardiomyopathy. Diabetes 55:798–805
Shimazu M, Umeda K, Sugihara N et al (1993) Collagen remodelling in myocardia of diabetic patients. J Clin Path 46:32–36
Soufi M, Sattler AM, Herzum M et al (2006) Molecular basis of obesity and the risk for cardiovascular disease. Herz 31(3):200–206
Stratmann B, Gawlowski T, Tschoepe D (2010) Diabetic cardiomyopathy-to take a long story serious. Herz 35(3):161–168
Stratmann B, Stork I, Tschoepe D (2008) Diabetic cardiomyopathy-a type of coronary artery disease? Internist 49(4):436–440
Stratton IM, Adler AI, Neil HA et al (2000) Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ 321:405–412
Swan GW, Anker SD, Walton C et al (1997) Insulin resistance in chronic failure: relation to severity and etiology of heart failure. J Am Coll Cardiol 30:527–532
Taegtmeyer H (2004) Cardiac metabolism as a target for the treatment of heart failure. Circulation 110:894–896
Takeda N, Nakamura I, Hatanaka T et al (1988) Myocardial mechanical and myosin isoenzyme alterations in streptozotocin-diabetic rats. Jpn Heart J 29:455–463
Tarquini R, Lazzeri C, Pala L et al (2010) The diabetic cardiomyopathy. Acta Diabetol. DOI: 10.1007/s00592-010-0180-x
Von Bibra H, Sutton J St M (2010) Diastolic dysfunction in diabetes and the metabolic syndrome: promising potential for diagnosis and prognosis. Diabetologia 53:1033–1045
Wachter R, Lüers C, Leta S et al (2007) Impact of diabetes on left ventricular diastolic function in patients with arterial hypertension. Eur J Heart Fail 9:469–476
Westermann D, Walther T, Savvatis K et al (2009) Gene deletion of the kinin receptor B1 attenuates cardiac inflammation and fibrosis during the development of experimental diabetic cardiomyopathy. Diabetes 58(6):1373–1381
Yim MB, Yim HS, Lee C et al (2001) Protein glycation: creation of catalytic sites for free radical generation. Ann N Y Acad Sci 928:48–53
Yu X, Tesiram YA, Towner RA et al (2007) Early myocardial dysfunction in streptozotocin-induced diabetic mice: a study using in vivo magnetic resonance imaging (MRI). Cardiovasc Diabetol 6:6
Conflict of interest
The corresponding author states that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maisch, B., Alter, P. & Pankuweit, S. Diabetic cardiomyopathy—fact or fiction?. Herz 36, 102–115 (2011). https://doi.org/10.1007/s00059-011-3429-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00059-011-3429-4
Keywords
- Diabetic cardiomyopathy (DCM)
- Heart failure with normal ejection fraction (HFNEF)
- Heart failure with reduced ejection fraction (HFREF)
- Diabetic macro-/microangiopathy
- Diabetic neuropathy