Abstract
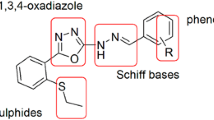
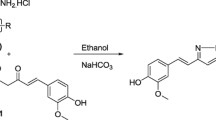
Twenty-four new compounds were prepared, taking curcumin as a lead, in order to explore their antioxidant and antitumor properties. The capacities of these derivatives to scavenge the 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonic acid) radical cation (ABTS.+), and to protect human red blood cells (RBCs) from oxidative haemolysis were investigated. In addition, the percentage viability of different cell lines (Hep G2, WI38, VERO and MCF-7) was tested. The result of the antitumor testing was generally in accordance with those of the antioxidant assays. Compounds which bear o-methoxy substitution to the 4-hydroxy function in the phenyl ring (7g, 5g and 3g) exhibited significantly higher ABTS.+-scavenging, antihaemolysis, and antitumor activities than other derivatives. In addition, molecular modelling studies were carried out for biologically active and inactive compounds, to study the structure–activity relationship, with the aim to elucidate which portions of the molecules are critical for the antioxidant and antitumor activity.











Similar content being viewed by others
References
Aggarwal BB, Kumar A, Bharti AC (2003) Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res 23:363–398
Al-Omary FAM, Abou-zeid LA, Nagi MN, Habib EE, Abdel-Aziz AA-M, El-Azab AS, Abdel-Hamide SG, Al-Omar MA, Al-Obaid AM, El-Subbagh HI (2010) Non-classical antifolates. Part 2. Synthesis, biological evaluation, and molecular modeling study of some new 2,6-substituted quinazolin-4-ones. Bioorg Med Chem 18:2849–2863
Anand P, Kunnumakkara AB, Newman RB, Aggarwal BB (2007) Bioavailability of curcumin: problems and promises. Mol Pharm 4:807–818
Chyong FH, Hui P, Cédric B, Jadranka T-S, Paul A (2011) ABTS.+ scavenging activity of polypyrrole, polyaniline and poly(3,4-ethylenedioxythiophene). Polym Int 60:69–77
Dinkova-Kostova AT, Talalay P (2008) Direct and indirect antioxidant properties of inducers of cytoprotective proteins. Mol Nutr Food Res 52:5128–5138
Djordjevic VB (2004) Free radical in cell biology. Int Rev Cytol 237:57–89
Du Z, Liu R, Shao W, Mao X, Ma L, Gu L, Huang Z, Cham ASC (2006) α-Glucosidase inhibition of natural curcuminoids and curcumin analogs. Eur J Med Chem 41:213–218
El-Sherbeny MA, Abdel-Aziz AA-M, Ahmed MA (2010) Synthesis and antitumor evaluation of novel diarylsulfonylurea derivatives: molecular modeling applications. Eur J Med Chem 45:689–697
Fadda AA, Badria FA, El-Attar KM (2010) Synthesis and evaluation of curcumin analogues as cytotoxic agents. Med Chem Res 19:413–430
Geol A, Kunnumakkara AB, Aggarwal BB (2008) Curcumin as “curecumin”: from kitchen to clinic. Biochem Pharm 75:787–809
Guthrie N, Carroll KK (1998) Inhibition of mammary cancer by citrus flavonoids. In: Manthey JA, Buslig BS (eds) Flavonoids in the living system. Plenum, New York, p 227
Halgren TAJ (1996) Merck molecular force field. I. Basis, form, scope, parameterization, and performance of MMFF94. J Comput Chem 17:490–519
Kálai T, Kuppusamy ML, Balog M, Selvendiran K, Rivera BK, Kuppusamy P, Hideg K (2011) Synthesis of N-substituted 3,5-bis(arylidene)-4-piperidones with high antitumor and antioxidant activity. J Med Chem 54:5414–5421
Kuang ZH, Wang PF, Zheng RL, Liu Z, Liu Y (1994) Making vitamin C liposoluble enhances its protective effect against radical induced hemolysis of erythrocytes. Chem Phys Lipids 91:95–97
Labute P, Williams C, Feher M, Sourial E, Schmidt JM (2001) Flexible alignment of small molecules. J Med Chem 44:1483–1490
Lee K, Ab Aziz FH, Syahida A, Abas F, Shaari K, Israf DA, Lajis NH (2009) Synthesis and biological evaluation of curcumin-like diarylpentanoid analogues for anti-inflammatory, antioxidant and antityrosinase activities. Eur J Med Chem 44:3195–3200
Liang G, Shao L, Wang Y, Zhao C, Chu Y, Xiao J, Zhao Y, Li X, Yang S (2009) Exploration and synthesis of curcumin analogues with improved structural stability both in vitro and in vivo as cytotoxic agents. Bioorg Med Chem 17:2623–2631
Ligeret H, Barthelemy S, Zini R, Tillement J, Labidalle S, Morin D (2004) Effects of curcumin and curcumin derivatives on mitochondrial permeability transition pore. Free Radic Biol Med 36:919–929
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ (1997) Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Del Rev 23:3–25
Lissi E, Modak B, Torres R, Escobar J, Urzua A (1999) Total antioxidant potential of resinous exudates from Heliotropium species, and a comparison of ABTS and DPPH methods. Free Radic Res 30:471–477
Memon VP, Sudheer AR (2007) Antioxidant and anti-inflammatory properties of curcumin. In: Aggarwal BB, Surh Y, Shishodia S (eds) The molecular targets and therapeutic uses of curcumin in health and disease. Springer, New York, pp 106–107
MOE 2007.9 of Chemical Computing Group. Inc.
Oprea TI, Davis AM, Teague SJ, Leeson PD (2001) Is there a difference between leads and drugs? A historical perspective. J Chem Inf Comput Sci 41:1308–1315
Roughley PJ, Whiting DA (1973) Experiments in the biosynthesis of curcumin. J Chem Soc Perkin Trans 1(20):2379–2388
Shang Y, Jin X, Shang X, Tang J, Liu G, Dai F, Qian Y, Fan G, Liu Q, Zhou B (2010) Antioxidant capacity of curcumin-directed analogues: structure–activity relationship and influence of microenvironment. Food Chem 119:1435–1442
Sharma RA, Gescher AJ, Verma WP (2005) Curcumin: the story so far. Eur J Cancer 41:1958–1968
Singh N, Pandey J, Yadav A, Chaturvedi V, Bhatnagar S, Gaikwad AN, Sinha SK, Kumar A, Shukla PK, Tripathi RT (2009) A facile synthesis of α,α’-(EE)-bis(benzylidene)-cycloalkanones and their antitubercular evaluations. Eur J Med Chem 44:1705–1709
Venkatesan P, Rao MN (2000) Structure–activity relationships for the inhibition of lipid peroxidation and the scavenging of free radicals by synthetic symmetrical curcumin analogues. J Pharm Pharmacol 52:1123–1128
Wang L, Sheng J, Tian H, Han J, Fan Z, Qian C (2004) A convenient synthesis of α,α’-bis(substituted benzylidene)cycloalkanones catalyzed by Yb(OTf)3 under solvent-free conditions. Synthesis 18:3060–3064
Weber WM, Hunsaker LA, Abcouwer SF, Deck LM, Vander Jagt DL (2005) Anti-oxidant activities of curcumin and related enones. Bioorg Med Chem 13:3811–3820
Weber WM, Hunsaker LA, Gonzales AM, Heynekamp JJ, Orlando RA, Deck LM, Vander Jagt DL (2006) TPA-induced up-regulation of activator protein-1 can be inhibited or enhanced by analogs of the natural product curcumin. Biochem Pharmacol 72:928–940
Zhou J (2003) One-step synthesis of pyridine and 4H-pyran derivatives from bisarylidenecyclohexanone and malononitrile under microwave irradiation. Syn Commun 33:99–103
Acknowledgments
The authors extend their appreciation to Mansoura University research center for funding this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bayomi, S.M., El-Kashef, H.A., El-Ashmawy, M.B. et al. Synthesis and biological evaluation of new curcumin derivatives as antioxidant and antitumor agents. Med Chem Res 22, 1147–1162 (2013). https://doi.org/10.1007/s00044-012-0116-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-012-0116-9