Abstract
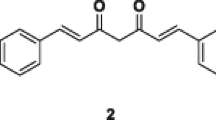
A series of curcumin pyrazole derivatives (3a–e) were synthesized. The chemical structures were determined by 1H and 13C NMR spectroscopic techniques and their purity was confirmed by LC–MS and melting point determination. The compounds were screened for anticancer effects on different cancer cell lines by MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) assay. The analogs demonstrated growth inhibitory effect on MCF-7, HeLa, and K562 cell lines with significant IC50 values. Compound 3b exhibited a high degree of cytotoxicity against cancer cells and minimum growth inhibitory effects against normal cells HEK293T and hence further, cell cycle analysis and mitochondrial membrane potential studies (JC-1 assay) were conducted by utilizing flow cytometry against K562 cells. This compound effectively arrested cell cycle progression at SubG1 phase and cells exhibited decreased membrane potential in a concentration-dependent manner with fluorescence shifting from red to green. Our findings suggest that compound 3b could be a promising anticancer agent since it effectively inhibited cell proliferation and can be selected for further in vitro and in vivo investigations.



Similar content being viewed by others
References
Aggarwal BB, Kumar A, Bharti AC (2003) Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res 23:363–398
Aggarwal BB, Sundaram C, Malani N, Ichikawa H (2007) Curcumin: the Indian solid gold. Adv Exp Med Biol 595:1–75
Aggarwal S, Ichikawa H, Takada Y, Sandur SK, Shidhodia S, Aggarwal BB (2006) Curcumin (diferuloylmethane) down-regulates expression of cell proliferation and antiapoptotic and metastatic gene products through suppression of I-kappa B-alpha kinase and Akt activation. Mol Pharmacol 69:195–206
Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB (2007) Bioavailability of curcumin: Problems and promises. Mol Pharm 4:807–818
Anand P, Sundaram C, Jhurani S, Kunnumakkara AB, Aggarwal BB (2008) Curcumin and cancer: an “old-age” disease with an “age-old” solution. Cancer Lett 267:133–164
Anand P, Thomas SG, Kunnumakkara AB, Sundaram C, Harikumar KB, Sung B, Tharakan ST, Misra K, Priyadarsini IK, Rajasekharan KN, Aggarwal BB (2008) Biological activities of curcumin and its analogues (congeners) made by man and mother nature. Biochem Pharmacol 76:1590–1611
Balbi A, Anzaldi M, Maccio C et al (2011) Synthesis and biological evaluation of novel pyrazole derivatives with anticancer activity. Eur J Med Chem 46(5293):309
Chandru H, Sharada AC, Ananda kumar CS, Rangappa KS (2008) Antiangiogenic and growth inhibitory effects of synthetic novel 1, 5-diphenyl-1,4 pentadiene-3-one-3-yl-ethanone pyridine curcumin analogues on ehrlich ascites tumor in vivo. Med Chem Res 17:515–529
Chandru H, Sharada AC, Bettadaiah BK, Ananda kumar CS, Rangappa KS, Sunila, Jayashree K (2007) In vivo growth inhibitory and anti-angiogenic effects of synthetic novel dienone cyclopropoxy curcumin analogs on mouse ehrlich ascites tumor. Bioorg Med Chem 15:7696–7703
Chandru H, Sharada AC (2007) Antiangiogenic effects of synthetic analogs of curcumin in vivo. Afr J Biomed Res 10:241–248
Davis R, Das U, Mackay H, Brown T, Mooberry SL, Dimmock JR, Lee M, Pati H (2008) Syntheses and cytotoxic properties of the curcumin analogs 2,6-Bis(benzylidene)-4-phenylcyclohexanones. Arch Pharm Chem Life Sci 341:440–445
Devi PU, Solomon FE, Sharada AC (1999) Plumbagin, a plant naphthoquinone with antitumor and radiomodifying properties. Pharma Biol 37:231–236
Duvoix A, Blasius R, Delhalle S, Schnekenburger M, Morceau F, Henry E et al (2005) Chemopreventive and therapeutic effects of curcumin. Cancer Lett 223:181–190
Ferrari M, Fornasiero MC, Isetta AM (1990) MTT colorimetric assay for testing macrophage cytotoxic activity in-vitro. J Immunol Methods 131:165–172
Fuchs JR, Pandit B, Bashin D, Etter JP, Regan N, Abdelhamid D, Li C, Lin J, Li PK (2009) Structure–activity relationship studies of curcumin analogues. Bioorg Med Chem Lett 19:2065–2069
Fujii S, Okinaga T, Ariyoshi W, Takahashi O, Iwanaga K, Nishino N, Tominaga K, Nishihara T (2013) Mechanisms of G1 cell cycle arrest and apoptosis in myeloma cells induced by hybrid-compound histone deacetylase inhibitor. Biochem Biophy Res Commun 434:413–420
Gopal PK, Paul M, Paul S (2014) Curcumin induces caspase mediated apoptosis in JURKAT cells by disrupting the redox balance. Asian Pac J Cancer Prev 15:93–100
Koca I, Ozgur A, Coskun KA, Tutar Y (2013) Synthesis and anticancer activity of acyl thioureas bearing pyrazole moiety. Bio Med Chem 21:3859–3865
Kumar D, Mishra PK, Anand AVK, Agrawal PK, Mohapatra R (2012) Isolation, synthesis and pharmacological evaluation of some novel curcumin derivatives as anticancer agents. J Med Plants Res 6:2880–2884
Lee DS, Lee MK, Kim JH (2009) Curcumin induces cell cycle arrest and apoptosis in human osteosarcoma (HOS) cells. Anticancer Res 29:5039–5044
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ (2001) Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev 46:3–26
Liu E, Wu J, Cao W, Zhang J, Liu W, Jiang X, Zhang X (2007) Curcumin induces G2/M cell cycle arrest in a p53-dependent manner and upregulates ING4 expression in human glioma. J neuro oncol 85:263–270
Liu T, Tan Z, Jiang L, Gu JF, Wu XS, Cao Y, Li M, Wu K, Liu Y (2013) Curcumin induces apoptosis in gallbladder carcinoma cell line GBC-SD cells. Cancer cell Int 13:64–72
Ly JD, Grubb R, Lawen A (2003) The mitochondrial membrane potential (∆ψm) in apoptosis; an update. Apoptosis 8:115–128
Mayadevi M, Sherin DR, Keerthi VS, Rajashekaran KN, Omkumar RV (2012) Curcumin is an inhibitor of calcium/calmodulin dependent protein kinase II. Bioorg Med Chem 20:6040–6047
Mishra S, Karmodiya K, Surolia N, Surolia A (2008) Synthesis and exploration of novel curcumin analogues as anti-malarial agents. Bioorg Med Chem 16:2894–2902
Newman DJ, Cragg GM, Snadder KM (2003) Natural products as sources of new drugs over the period 1981-2002. J Nat Prod 66:1022–1037
Ohori H, Yamakoshi H, Tomizawa M, Shibuya M, Kakudo Y, Takahashi A, Takahashi S, Kato S, Suzuki T, Ishioka C, Iwabuchi Y, Shibata H (2006) Synthesis and biological analysis of new curcumin analogues bearing an enhanced potential for the medicinal treatment of cancer. Mol Cancer Ther 5:2563–2571
Pan MH, Huang TM, Lin J (1999) Biotransformation of curcumin through reduction and glucuronidation in mice. Drug Metab Dispos 27:486–494
Pramod KS, Praveen KS, Gupta SK, Thavaselvam D, Agarwal DD (2012) Synthesis and evaluation of antimicrobial activity of 4H-pyrimido[2,1-b]benzothiazole, pyrazole and benzylidene derivatives of curcumin. Eur J Med Chem 54:366–378
Rocha AB, Lopes RM, Schwartsmann G (2001) Natural products in anticancer therapy. Curr Opin Pharmacol 1:364–369
Senft C, Polacin M, Priester M, Seifert V, Kogel D, Weissenberger J (2010) The non toxic natural compound Curcumin exerts anti-proliferative, anti-migratory and anti-invasive properties against malignant gliomas. BMC Cancer 10:491–498
Shamroukh AH, Rashad AE, Adbel-Megeid RE, Ali HS, Ali MM (2014) Some pyrazole and pyrazolo(3,4-d)pyrimidine derivatives: Synthesis and anticancer evaluation. Arch Pharm Chem Life Sciences 347:559–565
Sharath Kumar KS, Hanumappa A, Hegde M, Narasimhamurthy KH, Raghavan SC, Rangappa KS (2014) Synthesis and antiproliferative effect of novel 4-thiazolidinone-,pyridine- and piperazine-based conjugates on human leukemic cells. Eur J Med Chem 81:341–349
Sharath Kumar KS, Hanumappa A, Vetrivel M, Hegde M, Girish YR, Byregowda TR, Rao S, Raghavan SC, Rangappa KS (2015) Antiproliferative and tumor inhibitory studies of 2,3 disubstituted 4-thiazolidinone derivatives. Bioorg Med Chem Lett 25:3616–3620
Steward WP, Brown K (2013) Cancer chemoprevention: a rapidly evolving field. Br J Cancer 109:1–7
Thangapazham RL, Sharma A, Maheshwari RK (2006) Multiple molecular targets in cancer chemoprevention by curcumin. AAPS J 8:443–449
Tsao AS, Kim ES, Hong WK (2004) Chemoprevention of cancer. Cancer J Clin 54:150–180
Vinaya K, Kavitha CV, Chandrappa S, Prasanna DS, Raghavan SC, Rangappa KS (2011) Synthesis and antileukemic activity of novel 4-(3-(piperidin-4-yl) propyl)piperidine derivatives. Chem Biol Drug Des 78:622–630
Wang J, QI L, Zheng S, Wu T (2013) Curcumin induces apoptosis through the mitochondria-mediated apoptotic pathway in HT-29 cells. J Zhejiang Univ Sci B 10:93–102
Wang YJ, Pan MH, Cheng AL, Lin LI, Ho YS, Hsieh CY, Lin JK (1997) Stability of curcumin in buffer solutions and characterization of its degradation products. J Pharm Biomed Anal 15:1867–1876
Yin HT, Tian QZ, Guan L, Zhou Y, Huang XE, Zhang H (2013) In vitro and in vivo evaluation of the antitumor efficiency of resveratrol against lung cancer. Asian Pac J Cancer Prev 14:1703–1706
Youssef KM, Magda A, El-Sherbeny (2005) Synthesis and antitumor activity of some curcumin analogs. Arch Pharm Chem Life Sci 338:181–189
Acknowledgments
The authors express sincere gratitude to Dr. S Nagendra, Managing Director and Chief executive officer, Scintilla BIO-MARC pvt. Ltd, Bangalore, India for providing technical assistance regarding cell culture facilities for the work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementory Information
Rights and permissions
About this article
Cite this article
Puneeth, H.R., Ananda, H., Kumar, K.S.S. et al. Synthesis and antiproliferative studies of curcumin pyrazole derivatives. Med Chem Res 25, 1842–1851 (2016). https://doi.org/10.1007/s00044-016-1628-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-016-1628-5