Abstract
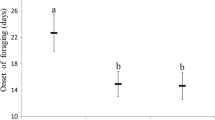
Predictions that precocious foraging in honeybee workers is a result of shortening of their life expectancy were tested in both laboratory and field experiments. In the laboratory experiment, we assessed the impact of anaesthesia with CO2 and infection with Nosema apis on the lifespan of workers. In the field experiment, the age at onset of foraging was observed in groups of workers with different expected lifespans. In both the experiments, workers originating from one queen inseminated with the semen of one drone were divided into five groups. The first group was anaesthetized with CO2 on the first day of life. Workers from the other three groups were individually inoculated with a constant number of N. apis spores on the 1st, 6th and 11th days of life. Workers from the fifth control group were neither treated with CO2 nor inoculated in any way. Both the laboratory and field experiments revealed that anaesthetized and infected workers had shorter expected lifespans compared to control bees. Amongst infected workers, those inoculated earlier in life survived for a significantly shorter period of time in comparison to those infected later in life. In agreement with the expectation, the field experiment showed that anaesthetized and infected workers with shorter expected lifespan start foraging earlier than control workers. Amongst the infected workers, age at inoculation correlated with age at onset of foraging. This means that short-lived workers complete safe nest tasks and begin riskier foraging earlier in life. Our results provide a strong support for the hypothesis that the division of labour in eusocial insects is a consequence of the different expected worker lifespan and different risk associated with their tasks; however, they do not contradict other existing explanations.




Similar content being viewed by others
References
Amdam G.V., Aase A.L.T.O., Seehuus S.C., Fondrk M.K., Norberg K. and Hartfelder K. 2005. Social reversal of immunosenescence in honey bee workers. Exp. Gerontol. 40: 939–947
Amdam G.V. and Omholt S.W. 2003. The hive bee to forager transition in honeybee colonies: the double repressor hypothesis. J. Theor. Biol. 223: 451–464
Amdam G.V. and Page R.P. 2005. Intergenerational transfers may have decoupled physiological and chronological age in a eusocial insect. Ageing Res. Rev. 4: 398–408
Amdam G.V., Simoes Z.L.P., Hagen A., Norberg K., Schroder K., Mikkelsen O., Kirkwood T.B.L. and Omholt S.W. 2004. Hormonal control of the yolk precursor vitellogenin regulates immune function and longevity in honeybees. Exp. Gerontol. 39: 767–773
Austin G.H. 1955. Effect of carbon dioxide anaesthesia on bee behaviour and expectation of life. Bee World 36: 45–47
Becerra-Guzman F., Guzman-Novoa E., Correa-Benitez A. and Zozaya-Rubio A. 2005. Length of life, age at first foraging and foraging life of Africanized and European honey bee (Apis mellifera) workers, during conditions of resource abundance. J. Apicult. Res. 44: 151–156
Behrends A., Scheiner R., Baker N. and Amdam G.V. 2007. Cognitive aging is linked to social role in honey bees (Apis mellifera). Exp. Gerontol. 42: 1146–1153
Beshers S.N. and Fewell J.H. 2001. Models of division of labor in social insects. Annu. Rev. Entomol. 46: 413–440
Breed M.D., Robinson G.E. and Page R.E. 1991. Division of labor during honey bee colony defense. Behav. Ecol. Sociobiol. 27: 395–401
Bühler A., Lanzrein B. and Wille H. 1983. Influence of temperature and carbon dioxide concentration on juvenile hormone titre and dependent parameters of adult worker honey bees (Apis mellifera L.). J. Insect Physiol. 29: 885–893
Calderone N.W. 1998. Proximate mechanisms of age polyethism in the honey bee, Apis mellifera L. Apidology 29: 127–158
Chapuisat M. and Keller L. 2002. Division of labour influences the rate of ageing in weaver ant workers. Proc. R. Soc. B 269: 909–913
D’Ettorre P. 2007. Evolution of sociality: you are what you learn. Curr. Biol. 17: R766–R768
Downey D.L., Higo T.T. and Winston M.L. 2000. Single and dual parasitic mite infestations on honey bee, Apis mellifera L. Insect. Soc. 47: 171–176
Dukas R. 2008. Mortality rates of honey bees in the wild. Insect. Soc. 55: 252–255
Ebadi R., Gary N.E. and Lorenzen K. 1980. Effects of carbon dioxide and low temperature narcosis on hone bees, Apis mellifera. Environ. Entomol. 9: 144–147
Fergusson L.A. and Winston M.L. 1988. The influence of wax deprivation on temporal polyethism in honey bee (Apis mellifera L.) colonies. Can. J. Zool. 66: 1997–2001
Fox G.A. 2001. Failure-time analysis. In: Design and Analysis of Ecological Experiments (Scheiner S.M. and Gurevitch J., Eds), University Press, New York. pp 235–266
Free J.B. 1961. Hypopharyngeal gland development and division of labor in honey-bee (Apis mellifera L.) colonies. Proc. R. Entomol. Soc. London. Ser. A 36: 5–8
Fries I. 1997. Protozoa. In: Honey Bee Pests, Predators, and Diseases, 3rd ed. (Morse R.A. and Flottum K., Eds), The AI Root Company, Medina. pp 58–76
Gordon D.M. 1987. Group-level dynamics in harvester ants: young colonies and the role of patrolling. Anim. Behav. 35: 833–843
Gordon D.M. 1996. The organisation of work in social insects. Nature 380: 121–124
Guzman-Novoa E., Page R.E. Jr. and Gary N.E. 1994. Behavioral and life-history components of division of labor in honey bees (Apis mellifera L). Behav. Ecol. Sociobiol. 34: 409–417
Hart A.G. and Ratnieks F.L.W. 2001. Task partitioning, division of labour and nest compartmentalisation collectively isolate hazardous waste in the leafcutting ant Atta cephalotes. Behav. Ecol. Sociobiol. 49: 387–392
Huang Z.Y. and Robinson G.E. 1992. Honeybee colony integration: worker–worker interactions mediate hormonally regulated plasticity in division of labor. Proc. Natl. Acad. Sci. U.S.A. 89: 11726–11729
Huang Z.Y. and Robinson G.E. 1996. Regulation of honey bee division of labor by colony age demography. Behav. Ecol. Sociobiol. 39: 147–158
Janmaat A.F. and Winston M.L. 2000. The influence of pollen storage area and Varroa jacobsoni Oudemans parasitism on temporal caste structure in honey bees (Apis mellifera L.). Insect. Soc. 47: 177–182
Jaycox E.R., Skowronek W. and Guynn G. 1974. Behavioral changes in worker honey bees (Apis mellifera L.) induced by injections of juvenile hormone mimic. Ann. Entomol. Soc. Am. 67: 529–534
Jeanne R.L. 1986. The evolution of the organization of work in social insects. Mon. Zool. Ital. NS. 20: 119–133
Jeanne R.L. 1991. Polyethism. In: The Social Biology of Wasps (Ross K.G. and Matthews R.W., Eds), Cornell University Press, New York. pp 389–425
Jemielity S., Chapuisat M., Parker J.D. and Keller L. 2005. Long live the queen: studying aging in social insects. Age 27: 241–248
Keller L. and Genoud M. 1997. Extraordinary lifespans in ants: a test of evolutionary theories of ageing. Nature 389: 958–960
Koywiwattrakul P., Thompson G.J., Sitthipraneed S., Oldroyd B.P. and Maleszka R. 2005. Effects of carbon dioxide narcosis on ovary activation and gene expression in worker honeybees, Apis mellifera. J. Insect Sci. 5: 1–10
Kukuk P.F. 1998. Mutualistic benefits generate an unequal distribution of risky activities among unrelated group members. Naturwissenschaften 85: 445–449
Malone L.A. and Gatehouse H.S. 1998. Effects of Nosema apis infection of honey bee (Apis mellifera) digestive proteolytic enzyme activity. J. Inverterbr. Pathol. 71: 169–174
Malone L.M. and Giacon H.A. 1996. Effects of Nosema apis Zander on inbred New Zealand honey bees (Apis mellifera ligustica L.). Apidologie 27: 479–486
Malone L.A., Giacon H.A. and Newton M.R. 1995. Comparison of the responses of some New Zealand and Australian honey bees (Apis mellifera L.) to Nosema apis Z. Apidologie 26: 495–502
Mardan M. and Rinderer T.E. 1980. Effects of carbon dioxide and cold anaesthesia on the hoaring behaviour of the honeybee. J. Apicult. Res. 19: 149–153
Medawar P.B. 1946. Old age and natural death. Mod. Q. 1: 30–56
Moroń D., Witek M. and Woyciechowski M. 2008. Division of labour among workers with different life expectancy in the ant Myrmica scabrinodis. Anim. Behav. 75: 345–350
Münch D., Amdam G.V. and Wolschin F. 2008. Ageing in a eusocial insect: molecular and physiological characteristics of life span plasticity in the honey bee. Funct. Ecol. 22: 407–421
Nelson C.M., Ihle K., Amdam G.V., Fondrk M.K. and Page R.E. 2007. The gene vitellogenin has multiple coordinating effects on social organization. PLoS Biol. 5: 673–677
Nicolas G. and Sillans D. 1989. Immediate and latent effects of carbon dioxide on insects. Annu. Rev. Entomol. 34: 97–116
O’Donnell S. 1996. Reproductive potential and division of labor in wasps: are queen and worker behavior alternative strategy? Ethol. Ecol. Evol. 8: 305–308
O’Donnell S. and Jeanne R.L. 1995. Implication of senescence patterns for the evolution of age polyethism in eusocial insects. Behav. Ecol. 6: 269–273
Oster G.F. and Wilson E.O. 1978. Caste and Ecology in the Social Insects. Princeton University Press, Princeton, NY. 372 pp
Page R.E., Robinson G.E., Britton D.S. and Fondrk M.K. 1992. Genotypic variability for rates of behavioral development in worker honeybees (Apis mellifera L.). Behav. Ecol. 3: 173–180
Pinto L.Z., Bitondi M.M.G. and Simões Z.L.P. 2000. Inhibition of vitellogenin synthesis in Apis mellifera workers by a juvenile hormone analogue, pyriproxyfen. J. Insect Physiol. 46: 153–160
Quinn G.P. and Keough M.J. 2002. Experimental Design and Data Analysis for Biologists. Cambridge University Press, Cambridge. 537 pp
Ratnieks F.L.W. and Anderson C. 1999. Task partitioning in insect societies. II. Use of queueing delay information in recruitment. Am. Nat. 154: 536–548
Remolina S.C., Hafez D.M., Robinson G.E. and Hughes K.A. 2007. Senescence in the worker honey bee Apis mellifera. J. Insect Physiol. 53: 1027–1033
Ribbands C.R. 1950. Changes in the behaviour of honey-bees following their recovery from anaesthesia. J. Exp. Biol. 27: 302–310
Robinson G.E. 1992. Regulation of division of labor in insect societies. Annu. Rev. Entomol. 37: 637–665
Robinson G.E. and Page R.E. 1995. Genotypic constraints on plasticity for corpse removal in honey bee colonies. Anim. Behav. 49: 867–876
Robinson G.E., Robert P. Jr., Colette S. and Strambi A. 1992. Colony integration in honeybees: mechanisms of behavioral reversion. Ethology 90: 336–348
Rose M. 1991. Evolutionary Biology of Aging. Oxford University Press, New York. 221 pp
Rueppell O., Bachelier C., Fondrk M.K. and Page R.E. 2007a. Regulation of life history determines lifespan of worker honey bees (Apis mellifera L.). Exp. Gerontol. 42: 1020–1032
Rueppell O., Christine S., Mulcrone C. and Groves L. 2007b. Aging without functional senescence in honey bee workers. Curr. Biol. 17: R274–R275
SAS Institute. 2004. SAS User’s Guide. Version 9.1.3. SAS Institute, Cary, NC
Sakagami S.F. and Fukuda H. 1968. Life table for worker honeybees. Res. Popul. Ecol. 10: 127–139
Schmid-Hempel P. 1987. Effect nectar-collecting by honeybees. I. Economic model. J. Anim. Ecol. 56: 209–218
Schmid-Hempel P. 1990. Reproductive competition and the evolution of work load in social insects. Am. Nat. 135: 501–526
Schmid-Hempel P. 1995. Parasite and social insects. Apidologie 26: 255–271
Schmid-Hempel P. 1998. Parasites in Social Insects. Princeton University Press, Princeton, NJ. 409 pp
Schmid-Hempel P. and Schmid-Hempel R. 1984. Life duration and turnover of foragers in the ant Cataglyphis bicolor (Hymenoptera: Formicidae). Insect. Soc. 31: 345–360
Schmid-Hempel P. and Wolf T. 1988. Foraging effort and life span of workers in a social insect. J. Anim. Ecol. 57: 509–521
Schneider P. and Drescher W. 1987 Influence of Varroa jacobsoni infection during pupal development of flight activity of the worker honey bee. Apidologie 17: 366–368
Seeley T.D. 1985. Honeybee Ecology. Princeton University Press, NJ. 201 pp
Seeley T.D. 1989. Social foraging in honey bees: how nectar foragers assess their colony’s nutritional status. Behav. Ecol. Sociobiol. 24: 181–199
Sullivan J.P., Fahrbach S.E., Harrison J.F., Capaldi E.A., Fewell J.H. and Robinson G.E. 2003. Juvenile hormone and division of labor in honey bee colonies: effects of allatectomy on flight behavior and metabolism. J. Exp. Biol. 206: 2287–2296
Tofilski A. 2002. Influence of age polyethism on longevity of workers in social insects. Behav. Ecol. Sociobiol. 51: 234–237
Tofts C. and Franks N.R. 1992. Doing the right thing: ants, honeybees and naked mole-rats. Trends Ecol. Evol. 7: 346–349
Toth A.L., Kantarovich S., Meisel A.F. and Robinson G.E. 2005. Nutritional status influences socially regulated foraging ontogeny in honey bees. J. Exp. Biol. 208: 4641–4649
Toth A.L. and Robinson G.E. 2005. Worker nutrition and division of labour in honeybees. Anim. Behav. 69: 427–435
Traniello J.F.A. and Rosengaus R.B. 1997. Ecology, evolution and division of labour in social insects. Anim. Behav. 53: 209–213
Tsuji K. 1990 Reproductive division of labour related to age in the Japanese queenless ant, Pristomyrmex pungens. Anim. Behav. 39: 843–849
Tustain R.C.R. and Faulke J. 1979. Effect of carbon dioxide anaesthesia on the longevity of honey bees in the laboratory. N. Zl. J. Exp. Agr. 7: 327–329
Waibel M., Floreano D., Magnenat S. and Keller L. 2006. Division of labour and colony efficiency in social insects: effects of interactions between genetic architecture, colony kin structure and rate of perturbations. Proc. R. Soc. B 273: 1815–1823
Wang D.I. and Moeller F.E. 1970. The division of labor and queen attendance behavior of nosema-infected workers honey bees. J. Econ. Entomol. 63: 1539–1541
West-Eberhard M.J. 1981. Intragroup selection and the evolution of insect societies. In: Natural Selection and Social Behavior (Alexander R.D. and Tinkle D.W., Eds), New York Chiron Press, New York. pp 3–17
Williams G.C. 1957. Pleitropy, natural selection, and the evolution of senescence. Evolution 11: 398–441
Winston M.L. and Fergusson L.A. 1985. The effect of worker loss on temporal caste structure in colonies of the honey bee (Apis mellifera L.). Can. J. Zool. 63: 777–780
Winston M.L. and Katz J. 1982 Foraging differences between cross-fostered honeybee workers (Apis mellifera) of European and Africanized races. Behav. Ecol. Sociobiol. 10: 125–129
Woyciechowski M. 2007. Risk of water collecting in honeybee (Apis mellifera) workers (Hymenoptera: Apidae). Sociobiology 50: 1059–1068
Woyciechowski M. and Kozlowski J. 1998. Division of labour by division of risk according to worker life expectancy in the honey bee (Apis mellifera L.). Apidologie 29: 191–205
Woyciechowski M. and Lomnicki A. 1995. Honey bee workers parasitized by Nosema apis choose riskier foraging strategies. Pszczel. Zesz. Nauk. 39: 209–210
Acknowledgments
We thank Bozena Pawlas, Katarzyna Piskorz and Maria Sklodowska from the Agricultural University in Krakow for technical assistance. We are grateful to Raghavendra Gadagkar, Juergen Heinze and Adam Tofilski for helpful comments on the manuscript. The study has been partly funded by the Polish Ministry of Science and Higher Education within grant SPUB-3024 and DS-761.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Woyciechowski, M., Moroń, D. Life expectancy and onset of foraging in the honeybee (Apis mellifera). Insect. Soc. 56, 193–201 (2009). https://doi.org/10.1007/s00040-009-0012-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-009-0012-6