Summary
-
1.
The ommatidial structure of a giant water bug (Lethocerus, Belostomatidae) is described, as well as the cell movements that occur on light/dark adaptation.
-
2.
Four cone cells beneath each facet connect the thick cornea to the rhabdomeres of the eight retinula cells.
-
3.
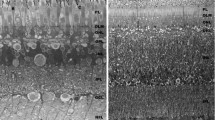
In the light-adapted eye, the four cone cells form a crystalline tract, 5 μm in diameter and 40 (μm long, which passes between heavily pigmented primary pigment cells to the rhabdomeres (Figs. 1, 2).
-
4.
In the dark-adapted eye, however, the rhabdomeres are 40 μm closer to the cornea and indent into the cone cells (Figs. 1, 5). There is no crystalline tract. The primary pigment cells are laterally displaced and the diameter of the light path is about 20 μm.
-
5.
Microtubules are abundant in the cone cells and primary pigment cells (Figs. 6, 8). They are oriented in the direction of the cell movement that occurs on light/dark adaptation.
-
6.
These cell movements may act as a pupil, reducing the amount of light reaching the rhabdom in the light-adapted eye.
Similar content being viewed by others
Literature
Anderson, W. A., Weissman, A., Ellis, R. A.: A comparative study of microtubules in some vertebrate and invertebrate cells. Z. Zellforsch. 71, 1–13 (1966).
Baumann, F.: Slow and spike potentials recorded from retinula cells of the honeybee drone in response to light. J. gen. Physiol. 52, 855–875 (1968).
Bernhard, C. G., Ottoson, D.: On the mechanisms of dark adaptation in day- and night-insects. Acta physiol. scand. 47, 383–384 (1959).
Cummings, C.: The giant water bugs (Belostomatidae, Hemiptera). Univ. Kansas Sci. Bull. 21, 197–219 (1933).
Exner, S.: Die Physiologie der facettirten Augen von Krebsen und Insecten. Vienna: Franz Deuticke 1891.
Höglund, G.: Pigment migration and retinula sensitivity. In: The functional organization of the compound eye, ed. C. G. Bernhard. London: Pergamon Press 1966.
Kirschfeld, K., Franceschini, N.: Ein Mechanismus zur Steuerung des Lichtflusses in den Rhabdomeren des Komplexauges von Musca. Kybernetik 6, 13–22 (1969).
Ledbetter, M. C., Porter, K. R.: A “microtubule” in plant cell fine structure. J. Cell Biol. 19, 239–250 (1963).
Lüdtke, H.: Retinomotorik und Adaptionsvorgänge im Auge des Rückenschwimmers (Notonecta glauca L.). Z. vergl. Physiol. 35, 129–152 (1953).
Naka, K. I., Kishida, K.: Retinal action potentials during dark and light adaptation. In: The functional organization of the compound eye, ed. C. G. Bernhard. London: Pergamon Press 1966.
Parker, G. H.: The movement of the retinal pigment. Ergebn. Biol. 9, 239–291 (1932).
Robison, W. G.: Microtubules in relation to the motility of a sperm syncytium in an armored scale insect. J. Cell Biol. 29, 251–265 (1966).
Sato, S., Kato, M., Toriumi, M.: Structural changes of the compound eye of Culex pipiens var. pallens Coquillet in the process of dark adaptation. Sci. Rep. Res. Inst. Tôhoku Univ. 23, 91–99 (1957).
Silveira, M., Porter, K. R.: The spermatozoids of flatworms and their microtubular systems. Protoplasma (Wien) 59, 240 (1964).
Sjöstrand, J., Frizell, M., Hasseigren, P.-O.: Effects of colchicine on axonal transport in peripheral nerve. J. Neurochem. 17, 1563 (1970).
Slautterback, D. B.: Cytoplasmic microtubules. I. Hydra. J. Cell Biol. 18, 367–388 (1963).
Taylor, A. C.: Microtubules in the microspikes and cortical cytoplasm of isolated cells. J. Cell Biol. 28, 155–168 (1966).
Tilney, L. G., Porter, K. R.: Studies on microtubules in Helizoa. I. The fine structure of Actinosphaerium nucleofilum (Barrett) with particular reference to the axial rod structure. Protoplasma (Wien) 60, 317–344 (1965).
Varela, F. G., Porter, K. R.: Fine structure of the visual system of the honeybee (Apis mellifera). I. The retina. J. Ultrastruct. Res. 29, 236–259 (1969).
Walcott, B.: Movement of retinula cells in insect eyes on light adaptation. Nature (Lond.) 223, 971–972 (1969).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Walcott, B. Cell movement on light adaptation in the retina of Lethocerus (Belostomatidae, Hemiptera). Z. Vergl. Physiol. 74, 1–16 (1971). https://doi.org/10.1007/BF00297785
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00297785