Abstract
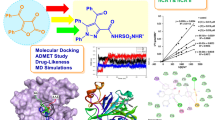
Malaria infection continues to pose a substantial threat to human health in the twenty-first century. The parasites’ resistance against conventional antimalarial drugs creates an emerging demand to develop new and efficient therapeutics against this mosquito-borne infection. Artemisinin, a sesquiterpene lactone, is a potent antimalarial drug found in nature. However, the lack of desirable physicochemical properties actuates researchers to develop new therapeutics based upon this compound. The 1,2,4-trioxane ring system of this natural product was identified as a crucial moiety for exhibiting antimalarial property. A related scientific investigation demonstrates that 1,2,4-trioxane derivatives bind to Plasmodium falciparum dihydrofolate reductase enzymes. The present work reports the synthesis of novel epoxide derivatives of 1,2,4-trioxanes and assesses their receptor binding profile towards Pf-DHFR employing molecular docking and dynamics experiments. The epoxides of 1,2,4-trioxane were synthesized using m-chloroperbenzoic acid and characterized by 1H & 13C NMR spectroscopy and mass spectrometry methods. The Pf-DHFR binding profile of epoxide derivatives of 1,2,4-trioxanes was evaluated utilizing in silico methods like Swiss ADME software. Various parameters calculated from 100 ns atomistic molecular dynamics trajectory, including MM-GBSA binding energy calculation, depict a good binding profile of synthesized compounds against this protein.










Similar content being viewed by others
Code availability
Not applicable.
Availability of data and material
Experimental details and spectral data of all the compounds are available in the attached electronic supplementary information (ESI).
References
Heller LE, Roepe PD (2019) Artemisinin-based antimalarial drug therapy: molecular pharmacology and evolving resistance. Trop Med Infect Dis 4:89. https://doi.org/10.3390/tropicalmed4020089
Fröhlich T, Çapcı Karagöz A, Reiter C, Tsogoeva SB (2016) Artemisinin-derived dimers: potent antimalarial and anticancer agents. J Med Chem 59:7360–7388. https://doi.org/10.1021/acs.jmedchem.5b01380
Slack RD, Jacobine AM, Posner GH (2012) Antimalarial peroxides: advances in drug discovery and design. Medchemcomm 3:281. https://doi.org/10.1039/c2md00277a
Cumming JN, Ploypradith P, Posner GH (1996) Antimalarial activity of artemisinin (Qinghaosu) and related trioxanes: mechanism (s) of action. 253–297
Klayman DL (1985) Qinghaosu (Artemisinin): an antimalarial drug from China. Science (80)228:1049–1055. https://doi.org/10.1126/science.3887571
Ploypradith P (2004) Development of artemisinin and its structurally simplified trioxane derivatives as antimalarial drugs. Acta Trop 89:329–342. https://doi.org/10.1016/j.actatropica.2003.10.006
O’Neill PM, Posner GH (2004) A medicinal chemistry perspective on artemisinin and related endoperoxides. J Med Chem 47:2945–2964. https://doi.org/10.1021/jm030571c
Tiwari MK, Coghi P, Agrawal P et al (2020) Design, synthesis, structure-activity relationship and docking studies of novel functionalized Arylvinyl-1,2,4-trioxanes as potent antiplasmodial as well as anticancer agents. ChemMedChem 15:1216–1228. https://doi.org/10.1002/cmdc.202000045
Debnath P, Debnath B, Bhaumik S, Debnath S (2020) In silico identification of potential inhibitors of ADP-ribose phosphatase of SARS-CoV-2 nsP3 by combining E-Pharmacophore- and receptor-based virtual screening of database. ChemistrySelect 5:9388–9398. https://doi.org/10.1002/slct.202001419
Sen D, Bhaumik S, Debnath P, Debnath S (2021) Potentiality of Moringa oleifera against SARS-CoV-2: identified by a rational computer aided drug design method J Biomol Struct Dyn 1–18. https://doi.org/10.1080/07391102.2021.1898475
Chatterjee S, Maity A, Chowdhury S et al (2021) In silico analysis and identification of promising hits against 2019 novel coronavirus 3C-like main protease enzyme. J Biomol Struct Dyn 39:5290–5303. https://doi.org/10.1080/07391102.2020.1787228
Singh C, Verma VP, Naikade NK et al (2010) 6-(4′-Aryloxy-phenyl)vinyl-1,2,4-trioxanes: a new series of orally active peroxides effective against multidrug-resistant Plasmodium yoelii in Swiss mice. Bioorg Med Chem Lett 20:4459–4463. https://doi.org/10.1016/j.bmcl.2010.06.045
Valdés-Tresanco MS, Valdés-Tresanco ME, Valiente PA, Moreno E (2020) AMDock: a versatile graphical tool for assisting molecular docking with Autodock Vina and Autodock4. Biol Direct 15:12. https://doi.org/10.1186/s13062-020-00267-2
Morris GM, Huey R, Lindstrom W et al (2009) AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. J Comput Chem 30:2785–2791. https://doi.org/10.1002/jcc.21256
Trott O, Olson AJ (2009) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem NA-NA. https://doi.org/10.1002/jcc.21334
Quiroga R, Villarreal MA (2016) Vinardo: a scoring function based on autodock vina improves scoring, docking, and virtual screening. PLoS ONE 11:e0155183. https://doi.org/10.1371/journal.pone.0155183
Masters L, Eagon S, Heying M (2020) Evaluation of consensus scoring methods for AutoDock Vina, smina and idock. J Mol Graph Model 96:107532. https://doi.org/10.1016/j.jmgm.2020.107532
Bowers KJ, Chow DE, Xu H et al (2006) Scalable algorithms for molecular dynamics simulations on commodity clusters. In: ACM/IEEE SC 2006 Conference (SC’06). IEEE 43–43
Laskowski RA, Jabłońska J, Pravda L et al (2018) PDBsum: structural summaries of PDB entries. Protein Sci 27:129–134. https://doi.org/10.1002/pro.3289
O’Boyle NM, Banck M, James CA et al (2011) Open Babel: an open chemical toolbox. J Cheminform 3:33. https://doi.org/10.1186/1758-2946-3-33
Rao PS, Apparao A, Professor A (2014) Energy minimization and conformation analysis of molecules using steepest descent method.
Huang J, Rauscher S, Nawrocki G et al (2017) CHARMM36m: an improved force field for folded and intrinsically disordered proteins. Nat Methods 14:71–73. https://doi.org/10.1038/nmeth.4067
Debnath P, Bhaumik S, Sen D et al (2021) Identification of SARS-CoV-2 main protease inhibitors using structure based virtual screening and molecular dynamics simulation of DrugBank database. ChemistrySelect 6:4991–5013. https://doi.org/10.1002/slct.202100854
Martyna GJ, Tobias DJ, Klein ML (1994) Constant pressure molecular dynamics algorithms. J Chem Phys 101:4177–4189. https://doi.org/10.1063/1.467468
Darden T, York D, Pedersen L (1993) Particle mesh Ewald: An N ⋅log( N ) method for Ewald sums in large systems. J Chem Phys 98:10089–10092. https://doi.org/10.1063/1.464397
Gohlke H, Kiel C, Case DA (2003) Insights into protein–protein binding by binding free energy calculation and free energy decomposition for the Ras-Raf and Ras–RalGDS complexes. J Mol Biol 330:891–913. https://doi.org/10.1016/S0022-2836(03)00610-7
Daina A, Michielin O, Zoete V (2017) SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep 7:42717. https://doi.org/10.1038/srep42717
Rudrapal M, Chetia D, Singh V (2017) Novel series of 1,2,4-trioxane derivatives as antimalarial agents. J Enzyme Inhib Med Chem 32:1159–1173. https://doi.org/10.1080/14756366.2017.1363742
Rawat V, Dey S, Sudalai A (2012) Synthesis of the anti-influenza agent (−)-oseltamivir free base and (−)-methyl 3-epi-shikimate. Org Biomol Chem 10:3988. https://doi.org/10.1039/c2ob25635e
Nighot D, Jain AK, Singh M, Rawat V (2020) A convenient 5-exo-dig cyclization route to diastereomerically pure methyl (2S)-2-(1-benzyl-3-oxo-1,3-dihydro-2H-isoindol-2-yl)-3-methylbutanoate. Chem Heterocycl Compd 56:1370–1374. https://doi.org/10.1007/s10593-020-02825-y
Karnatak M, Hassam M, Vanangamudi M et al (2021) Novel naphthyl based 1,2,4-trioxanes: Synthesis and in vivo efficacy in the Plasmodium yoelii nigeriensis in Swiss mice. Bioorg Med Chem Lett 51:128372. https://doi.org/10.1016/j.bmcl.2021.128372
Cross JB, Thompson DC, Rai BK et al (2009) Comparison of several molecular docking programs: pose prediction and virtual screening accuracy. J Chem Inf Model 49:1455–1474. https://doi.org/10.1021/ci900056c
Wang Z, Sun H, Yao X et al (2016) Comprehensive evaluation of ten docking programs on a diverse set of protein–ligand complexes: the prediction accuracy of sampling power and scoring power. Phys Chem Chem Phys 18:12964–12975. https://doi.org/10.1039/C6CP01555G
Islam MA, Pillay TS (2019) Identification of promising anti-DNA gyrase antibacterial compounds using de novo design, molecular docking and molecular dynamics studies. J Biomol Struct Dyn 1–12. https://doi.org/10.1080/07391102.2019.1617785
Kufareva I, Abagyan R (2012) Methods of protein structure comparison. Methods Mol Biol 857:231–257. https://doi.org/10.1007/978-1-61779-588-6_10
Acknowledgements
Authors acknowledge Dr. Ravi Kumar Muttineni, Founder & CSO of Immunocure Discovery Solutions Pvt Ltd, Hyderabad, Telangana, India, 500101, for conducting MM/GBSA experiments by providing software facilities in his establishment.
Funding
Ved Prakash Verma thanks the Department of Science and Technology, New Delhi, India, for the research grant. This research work was financially supported by DST-SERB File number: EEQ /2019/000078.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception & design. The manuscript was written through contributions of all authors. Akriti Kumari and Manvika Karnatak, conducted experiments related to chemical synthesis, purification, and characterization of organic compounds. Varun Rawat and Ved Prakash Verma conceptualized the synthetic work and contributed in manuscript preparation. Debanjan Sen conducted molecular dynamics simulation and contributed in manuscript preparation. Shahnawaz Khan participated in the interpretation of data.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kumari, A., Karnatak, M., Sen, D. et al. Synthesis, molecular docking and dynamics study of novel epoxide derivatives of 1,2,4-trioxanes as antimalarial agents. Struct Chem 33, 907–919 (2022). https://doi.org/10.1007/s11224-022-01885-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-022-01885-4