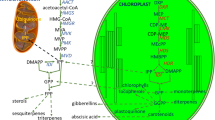
Abstract
The isoprenoid family represents one of the most ancient and widespread classes of structurally and functionally rich biomolecules known to man. Although these natural products are synthesized in all organisms, the plant kingdom exhibits tremendous variation in their chemistry and roles, ranging from primary metabolism to secondary metabolism and specialized ecological interactions with the environment. Despite enormous diversity in structure and function, all isoprenoids are derived from the universal C5 precursor isoprene. The isoprenoid biosynthetic pathway has three major stages, viz., (1) synthesis of the isoprene building blocks, followed by their (2) assembly into flexible linear and branched hydrocarbon substrates, which then undergo (3) multistep reaction cascades to generate the vast assortment of isoprenoid end products. One of the most interesting aspects of isoprenoid biosynthesis is its being finely tuned by a multilayered and complex regulatory network, which excellently controls the machinery producing one of the most heterogeneous groups of molecules in plants. Terpene synthases, enzymes of the final stage, are key players in the generation of isoprenoid diversity, catalyzing one of the most complex reactions known to chemistry and biology. Unraveling the mechanism by which a minimal pool of substrates is thus converted into tens of thousands of regiospecific and stereospecific products, is a promising research avenue: This knowledge may be practically used for rational design of novel compounds by metabolic engineering, in order to yield plants with improved nutritional efficacy, stress resistance, bio-pharmaceutical properties etc. This review is an attempt to summarize the biochemical, molecular, physiological, structural, genomic and evolutionary aspects of isoprenoid biosynthesis, providing new insights into how these enzymes utilize various innovative strategies for creation of the so-called final terpenome.









Similar content being viewed by others
Abbreviations
- 3-D:
-
Three-dimensional
- AACT:
-
Acetoacetyl-coenzyme A thiolase
- Ac-CoA:
-
Acetyl-coenzyme A
- AcAc-CoA:
-
Acetoacetyl-coenzyme A
- ABA:
-
Abscisic acid
- ADP:
-
Adenosine diphosphate
- ATP:
-
Adenosine triphosphate
- CCN:
-
Cloud condensation nuclei
- CDP-ME:
-
4-Diphosphocytidyl-2C-methyl-d-erythritol
- CDP-MEP:
-
4-Diphosphocytidyl-2C-methyl-d-erythritol 2-phosphate
- CMK:
-
4-Diphosphocytidyl-2C-methyl-d-erythritol kinase
- CDP:
-
Cytidine diphosphate
- CLR:
-
Cavity lining residues
- CMC:
-
Cambial meristematic cells
- CMP:
-
Cytidine monophosphate
- CPS:
-
ent-copalyl diphosphate synthase
- CTP:
-
Cytidine triphosphate
- DDC:
-
Dedifferentiated cells
- DMAPP:
-
Dimethylallyl diphosphate
- DOXP:
-
Deoxyxylulose phosphate
- DX:
-
1-Deoxy-d-xylulose
- DXP:
-
1-Deoxy-d-xylulose 5-phosphate
- DXR:
-
1-Deoxy-d-xylulose 5-phosphate reducto-isomerase
- DXS:
-
1-Deoxy-d-xylulose 5-phosphate synthase
- EAS:
-
epi-aristolochene synthase
- ER:
-
Endoplasmic reticulum
- FPPS:
-
Farnesyl pyrophosphate synthase
- FPP:
-
Farnesyl diphosphate
- G6P:
-
Glucose 6-phosphate
- GA:
-
Gibberellic acids
- GAP:
-
D-glyceraldehyde 3-phosphate
- GGPPS:
-
Geranylgeranyl pyrophosphate synthase
- GHMP:
-
Galacto-homoserine-mevalonate-phosphomevalonate
- GPP:
-
Geranyl diphosphate
- GGPP:
-
Geranylgeranyl diphosphate
- GUS:
-
β-Glucuronidase
- HDR:
-
1-Hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate reductase
- HDS:
-
1-Hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate synthase
- HGT:
-
Horizontal gene transfer
- HMBPP:
-
1-Hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate
- HMG-CoA:
-
3-Hydroxy-3-methylglutaryl coenzyme A
- HMGR:
-
3-Hydroxy-3-methylglutaryl coenzyme A reductase
- HMGS:
-
3-Hydroxy-3-methylglutaryl coenzyme A synthase
- HR:
-
Hypersensitive response
- IDI:
-
Isopentenyl-diphosphate isomerase
- IPP:
-
Isopentenyl diphosphate
- KO:
-
Knock-out
- LDL:
-
Low density lipoproteins
- LGT:
-
Lateral gene transfer
- MDS:
-
2C-Methyl-d-erythritol 2,4-cyclodiphosphate synthase
- LSU:
-
Large subunit
- MCT:
-
2C-Methyl-d-erythritol 4-phosphate cytidyltransferase
- ME:
-
2C-Methyl-d-erythritol
- ME-cPP:
-
2C-Methyl-d-erythritol 2,4-cyclodiphosphate
- MEP:
-
2C-Methyl-d-erythritol 4-phosphate
- MVA:
-
Mevalonic acid/mevalonate
- MK:
-
Mevalonate kinase
- MVADP:
-
Mevalonate 5-diphosphate
- NADPH:
-
Reduced nicotinamide adenine dinucleotide phosphate
- NGF:
-
Nerve growth factor
- NMP:
-
Nucleoside monophosphate
- PDB:
-
Protein data bank
- PDP:
-
Prenyl diphosphates
- PPMD:
-
Diphospho-mevalonate decarboxylase
- PMK:
-
Phospho-mevalonate kinase
- PPi:
-
Pyrophosphate leaving group
- PT:
-
Prenyl transferases
- RCSB:
-
Research collaboratory for structural bioinformatics
- RMSD:
-
Root mean square deviation
- SSU:
-
Small subunit
- THC:
-
Tetra hydro cannabinol
- TPS:
-
Terpene synthase
- UDS:
-
Undecaprenyl pyrophosphate synthase
References
Ahn C, Pai HS (2008) Physiological function of IspE, a plastid MEP pathway gene for isoprenoid biosynthesis, in organelle biogenesis and cell morphogenesis in Nicotianabenthamiana. Plant Mol Biol 66:503–517
Ait Said S, Fernandez C, Greff S, Derridj A, Gauquelin T, Mevy JP (2011) Inter-population variability of leaf morpho-anatomical and terpenoid patterns of Pistaciaatlantica Desf. ssp. atlantica growing along an aridity gradient in Algeria. Flora Morph Distr Funct Ecol Plants 206:397–405
Ajikumar PK, Xiao WH, Tyo KEJ, Wang Y, Simeon F, Leonard E, Mucha O, Phon TH, Pfeifer B, Stephanopoulos G (2010) Isoprenoid pathway optimization for taxol precursor overproduction in Escherichia coli. Science 330:70–74
Albermann S, Linnemannstöns P, Tudzynski B (2012) Strategies for strain improvement in Fusarium fujikuroi: overexpression and localization of key enzymes of the isoprenoid pathway and their impact on gibberellin biosynthesis. Appl Microbiol Biotech 97:1–17
Alex D, Bach TJ, Chye M-L (2000) Expression of Brassica juncea 3-hydroxy-3-methylglutaryl CoA synthase is developmentally regulated and stress-responsive. Plant J 22:415–426
Andersson SGE, Zomorodipour A, Andersson JO, Sicheritz-Pontön T, Alsmark UCM, Podowski RM, Näslund AK, Eriksson AS, Winkler HH, Kurland CG (1998) The genome sequence of Rickettsia prowazekii and the origin of mitochondria. Nature 396:133–140
Andreassi JL, Dabovic K, Leyh TS (2004) Streptococcus pneumoniae isoprenoid biosynthesis is downregulated by diphosphomevalonate: an antimicrobial target. Biochemistry 43:16461–16466
Andreassi JL, Bilder PW, Vetting MW, Roderick SL, Leyh TS (2009a) Crystal structure of the Streptococcus pneumoniae mevalonate kinase in complex with diphosphomevalonate. Protein Sci 16:983–989
Andreassi JL, Vetting MW, Bilder PW, Roderick SL, Leyh TS (2009b) Structure of the ternary complex of phosphomevalonate kinase: the enzyme and its family. Biochemistry 48:6461–6468
Appendino G, Chianese G, Taglialatela-Scafati O (2011) Cannabinoids: occurrence and medicinal chemistry. Curr Med Chem 18:1085–1099
Ashby M, Kutsunai S, Ackerman S, Tzagoloff A, Edwards PA (1992) COQ2 is a candidate for the structural gene encoding para-hydroxybenzoate: polyprenyltransferase. J Biol Chem 267:4128–4136
Augustyn W, Botha B, Combrinck S, Du Plooy W (2010) Correlation of volatile profiles of twenty mango cultivars with their susceptibilities to mango gall fly infestation. South African J Bot 76:710–716
Bach TJ (1995) Some new aspects of isoprenoid biosynthesis in plants—a review. Lipids 30:191–202
Barrero AF, Herrador M, Arteaga P, Arteaga JF, Arteaga AF (2012) Communic acids: occurrence, properties and use as chirons for the synthesis of bioactive compounds. Molecules 17:1448–1467
Barta ML, Skaff DA, McWhorter WJ, Herdendorf TJ, Miziorko HM, Geisbrecht BV (2011) Crystal structures of Staphylococcus epidermidis mevalonate diphosphate decarboxylase bound to inhibitory analogs reveal new insight into substrate binding and catalysis. J Biol Chem 286:23900–23910
Barta ML, McWhorter WJ, Miziorko HM, Geisbrecht BV (2012) Structural basis for nucleotide binding and reaction catalysis in mevalonate diphosphate decarboxylase. Biochemistry 51:5611–5621
Bartram S, Jux A, Gleixner G, Boland W (2006) Dynamic pathway allocation in early terpenoid biosynthesis of stress-induced lima bean leaves. Phytochemistry 67:1661–1672
Berthelot K, Estevez Y, Deffieux A, Peruch F (2012) Isopentenyl diphosphate isomerase: a checkpoint to isoprenoid biosynthesis. Biochimie 94:1621–1634
Bitok JK, Freel Meyers CL (2012) 2C-Methyl-d-erythritol 4-phosphate enhances and sustains cyclodiphosphate synthase IspF activity. ACS Chem Biol 7:1702–1710
Bloch K (1965) The biological synthesis of cholesterol. Nobel Found 15:119–150
Bohlmann J, Meyer-Gauen G, Croteau R (1998) Plant terpenoid synthases: molecular biology and phylogenetic analysis. Proc Natl Acad Sci 95:4126–4133
Bork P, Sander C, Valencia A (1993) Convergent evolution of similar enzymatic function on different protein folds: the hexokinase, ribokinase, and galactokinase families of sugar kinases. Protein Sci 2:31–40
Botta B, Monache GD, Menendez P, Boffi A (2005) Novel prenyltransferase enzymes as a tool for flavonoid prenylation. Trends Pharm Sci 26:606–608
Bouvier F, Rahier A, Camara B (2005) Biogenesis, molecular regulation and function of plant isoprenoids. Prog Lipid Res 44:357–429
Brandt W, Bräuer L, Günnewich N, Kufka J, Rausch F, Schulze D, Schulze E, Weber R, Zakharova S, Wessjohann L (2009) Molecular and structural basis of metabolic diversity mediated by prenyl diphosphate converting enzymes. Phytochemistry 70:1758–1775
Brüggemann N, Schnitzler JP (2002) Relationship of isopentenyl diphosphate (IDP) isomerase activity to isoprene emission of oak leaves. Tree Physiol 22:1011–1018
Burley SK, Bonanno JB (2002a) Structural genomics of proteins from conserved biochemical pathways and processes. Curr Opin Struct Biol 12:383–391
Burley SK, Bonanno JB (2002b) Structuring the universe of proteins. Ann Rev Genomics Human Genet 3:243–262
Cakmak H, Basar M, Seval-Celik Y, Osteen KG, Duleba AJ, Taylor HS, Lockwood CJ, Arici A (2012) Statins inhibit monocyte chemotactic protein 1 expression in endometriosis. Repr Sci 19:572–579
Calisto BM, Perez-Gil J, Bergua M, Querol-Audi J, Fita I, Imperial S (2007) Biosynthesis of isoprenoids in plants: structure of the 2C-methyl-d-erithrytol 2,4-cyclodiphosphate synthase from Arabidopsis thaliana. Comparison with the bacterial enzymes. Protein Sci 16:2082–2088
Cane DE (2000) Biosynthesis meets bioinformatics. Science 287:818–819
Cane DE, Ikeda H (2011) Exploration and mining of the bacterial terpenome. Acc Chem Res 45:463–472
Cane DE, Kang I (2000) Aristolochene synthase: purification, molecular cloning, high-level expression in Escherichia coli, and characterization of the Aspergillus terreus cyclase. Arch Biochem Biophys 376:354–364
Cao R, Zhang Y, Mann FM, Huang C, Mukkamala D, Hudock MP, Mead ME, Prisic S, Wang K, Lin FY (2010) Diterpene cyclases and the nature of the isoprene fold. Proteins Struc Func Bioinf 78:2417–2432
Cascini F, Aiello C, Di Tanna G (2012) Increasing delta-9-tetrahydrocannabinol (∆-9-THC) content in herbal cannabis over time: systematic review and meta-analysis. Curr Drug Abuse Rev 5:32–40
Casey PJ, Seabra MC (1996) Protein prenyltransferases. J Biol Chem 271:5289–5292
Chang T-H, Hsieh F-L, Ko T-P, Teng K-H, Liang P-H, Wang AH-J (2010) Structure of a heterotetrameric geranyl pyrophosphate synthase from mint (Mentha piperita) reveals intersubunit regulation. Plant Cell 22:454–467
Chappell J (1995a) Biochemistry and molecular biology of isoprenoid biosynthetic pathway in plants. Annu Rev Plant Physiol Plant Mol Biol 46:521–547
Chappell J (1995b) The biochemistry and molecular biology of isoprenoid metabolism. Plant Physiol 107:1–6
Chen F, Tholl D, Bohlmann J, Pichersky E (2011) The family of terpene synthases in plants: a mid-size family of genes for specialized metabolism that is highly diversified throughout the kingdom. Plant J 66:212–229
Christianson DW (2008) Unearthing the roots of the terpenome. Curr Opin Chem Biol 12:141–150
Chu HY, Wegel E, Osbourn A (2011) From hormones to secondary metabolism: the emergence of metabolic gene clusters in plants. Plant J 66:66–79
Clastre M, Goubard A, Prel A, Mincheva Z, Viaud-Massuart MC, Bout D, Rideau M, Velge-Roussel F, Laurent F (2007) The methylerythritol phosphate pathway for isoprenoid biosynthesis in coccidia: presence and sensitivity to fosmidomycin. Exp Parasitol 116:375–384
Clastre M, Papon N, Courdavault V, Giglioli-Guivarc’h N, St-Pierre B, Simkin AJ (2011) Subcellular evidence for the involvement of peroxisomes in plant isoprenoid biosynthesis. Plant Sig Behav 6:2044–2046
Cornish K (1993) The separate roles of plant cis and trans prenyl transferases in cis-1, 4-polyisoprene biosynthesis. Eur J Biochem 218:267–271
Croteau R, Kutchan TM, Lewis NG (2000) Natural products (secondary metabolites). In: Buchanan B, Gruissem W, Jones R (eds) Biochemistry and molecular biology of plants. American Society of Plant Physiologists, Rockville, pp 1250–1318
Dai Z, Cui G, Zhou SF, Zhang X, Huang L (2011) Cloning and characterization of a novel 3-hydroxy-3-methylglutaryl coenzyme A reductase gene from Salvia miltiorrhiza involved in diterpenoid tanshinone accumulation. J Plant Physiol 168:148–157
Darabi M, Masoudi-Nejad A, Nemat-Zadeh G (2012) Bioinformatics study of the 3-hydroxy-3-methylglotaryl-coenzyme A reductase (HMGR) gene in Gramineae. Mol Biol Rep 39:1–11
Degenhardt J, Köllner TG, Gershenzon J (2009) Monoterpene and sesquiterpene synthases and the origin of terpene skeletal diversity in plants. Phytochemistry 70:1621–1637
Dorsey JA, Porter JW (1968) The effect of palmityl coenzyme A on pigeon liver fatty acid synthetase. J Biol Chem 243:3512–3516
Dudareva N, Martin D, Kish CM, Kolosova N, Gorenstein N, Fäldt J, Miller B, Bohlmann J (2003) (E)-β-ocimene and myrcene synthase genes of floral scent biosynthesis in snapdragon: function and expression of three terpene synthase genes of a new terpene synthase subfamily. Plant Cell 15:1227–1241
Dudareva N, Andersson S, Orlova I, Gatto N, Reichelt M, Rhodes D, Boland W, Gershenzon J (2005) The nonmevalonate pathway supports both monoterpene and sesquiterpene formation in snapdragon flowers. Proc Natl Acad Sci USA 102:933–938
Dunford JE (2010) Molecular targets of the nitrogen containing bisphosphonates: the molecular pharmacology of prenyl synthase inhibition. Curr Pharm Des 16:2961–2969
Durbecq V, Sainz G, Oudjama Y, Clantin B, Bompard-Gilles C, Tricot C, Caillet J, Stalon V, Droogmans L, Villeret V (2001) Crystal structure of isopentenyl diphosphate: dimethylallyl diphosphate isomerase. EMBO J 20:1530–1537
Eberl M, Hintz M, Reichenberg A, Kollas AK, Wiesner J, Jomaa H (2003) Microbial isoprenoid biosynthesis and human γδ-T cell activation. FEBS Lett 544:4–10
Elaissi A, Rouis Z, Mabrouk S, Salah KB, Aouni M, Khouja ML, Farhat F, Chemli R, Harzallah-Skhiri F (2012) Correlation between chemical composition and antibacterial activity of essential oils from fifteen Eucalyptus species growing in the Korbous and Jbel Abderrahman arboreta (North East Tunisia). Molecules 17:3044–3057
Ericsson J, Runquist M, Thelin A, Andersson M, Chojnacki T, Dallner G (1993) Distribution of prenyltransferases in rat tissues. Evidence for a cytosolic all-trans-geranylgeranyl diphosphate synthase. J Biol Chem 268:832–838
Fanciullino AL, Dhuique-Mayer C, Luro F, Casanova J, Morillon R, Ollitrault P (2006) Carotenoid diversity in cultivated citrus is highly influenced by genetic factors. J Agric Food Chem 54:4397–4406
Farhi M, Marhevka E, Masci T, Marcos E, Eyal Y, Ovadis M, Abeliovich H, Vainstein A (2011) Harnessing yeast subcellular compartments for the production of plant terpenoids. Metab Eng 13:474–481
Field B, Osbourn AE (2008) Metabolic diversification-independent assembly of operon-like gene clusters in different plants. Science 320:543–547
Field B, Osbourn A (2012) Order in the playground: formation of plant gene clusters in dynamic chromosomal regions. Mob Genet Elem 2:46–50
Field B, Fiston-Lavier AS, Kemen A, Geisler K, Quesneville H, Osbourn AE (2011) Formation of plant metabolic gene clusters within dynamic chromosomal regions. Proc Natl Acad Sci 108:16116–16121
Flores-Pérez U, Pérez-Gil J, Closa M, Wright LP, Botella-Pavía P, Phillips MA, Ferrer A, Gershenzon J, Rodríguez-Concepción M (2010) PLEIOTROPIC REGULATORY LOCUS 1 (PRL1) integrates the regulation of sugar responses with isoprenoid metabolism in Arabidopsis. Mol Plant 3:101–112
Flügge UI, Gao W (2005) Transport of isoprenoid intermediates across chloroplast envelope membranes. Plant Biol 7:91–97
Fraenkel GS (1959) The raison d’etre of secondary plant substances. Science 129:1466–1470
Fraenkel G (1969) Evaluation of our thoughts on secondary plant substances. Entomol Exp Appl 12:473–486
Frey M, Chomet P, Glawischnig E, Stettner C, Grün S, Winklmair A, Eisenreich W, Bacher A, Meeley RB, Briggs SP, Simcox K, Gierl A (1997) Analysis of a chemical plant defense mechanism in grasses. Science 277:696–699
Fu Z, Wang M, Potter D, Miziorko HM, Kim JJP (2002) The structure of a binary complex between a mammalian mevalonate kinase and ATP insights into the reaction mechanism and human inherited disease. J Biol Chem 277:18134–18142
Fu Z, Voynova NE, Herdendorf TJ, Miziorko HM, Kim J-JP (2008) Biochemical and structural basis for feedback inhibition of mevalonate kinase and isoprenoid metabolism. Biochemistry 47:3715–3724
Funck VR, de Oliveira CV, Pereira LM, Rambo LM, Ribeiro LR, Royes LFF, Ferreira J, Guerra GP, Furian AF, Oliveira MS (2011) Differential effects of atorvastatin treatment and withdrawal on pentylenetetrazol induced seizures. Epilepsia 52:2094–2104
Füssel U, Dötterl S, Jürgens A, Aas G (2007) Inter-and intraspecific variation in floral scent in the genus salix and its implication for pollination. J Chem Ecol 33:749–765
Gabrielsen M, Bond CS, Hallyburton I, Hecht S, Bacher A, Eisenreich W, Rohdich F, Hunter WN (2004) Hexameric assembly of the bifunctional methylerythritol 2,4-cyclodiphosphate synthase and protein-protein associations in the deoxy-xylulose-dependent pathway of isoprenoid precursor biosynthesis. J Biol Chem 279:52753–52761
Gabrielsen M, Kaiser J, Rohdich F, Eisenreich W, Laupitz R, Bacher A, Bond CS, Hunter WN (2006) The crystal structure of a plant 2C-methyl-d-erythritol 4-phosphate cytidylyltransferase exhibits a distinct quaternary structure compared to bacterial homologues and a possible role in feedback regulation for cytidine monophosphate. FEBS J 273:1065–1073
Ganjewala D, Kumar S, Luthra R (2009) An account of cloned genes of methyl-erythritol-4-phosphate pathway of isoprenoid biosynthesis in plants. Curr Issues Mol Biol 11:35–45
Gao S, Lin J, Liu X, Deng Z, Li Y, Sun X, Tang K (2006) Molecular cloning, characterization and functional analysis of a 2C-methyl-d-erythritol 2, 4-cyclodiphosphate synthase gene from Ginkgo biloba. J Biochem Mol Biol 39:502–510
Gennadios HA, Gonzalez V, Di Costanzo L, Li A, Yu F, Miller DJ, Allemann RK, Christianson DW (2009) Crystal structure of (+)-δ-Cadinene synthase from Gossypium arboreum and evolutionary divergence of metal binding motifs for catalysis. Biochemistry 48:6175–6183
Gerber E, Hemmerlin AA, Hartmann M, Heintz D, Hartmann M-AE, Mutterer JRM, Rodríguez-Concepción M, Boronat A, Van Dorsselaer A, Rohmer M, Crowell DN, Bach TJ (2009) The plastidial 2-C-methyl-d-erythritol 4-phosphate pathway provides the isoprenyl moiety for protein geranylgeranylation in tobacco BY-2 Cells. Plant Cell 21:285–300
Giménez-Oya V, Villacañas Ó, Fernàndez-Busquets X, Rubio-Martinez J, Imperial S (2009) Mimicking direct protein—protein and solvent-mediated interactions in the CDP-methylerythritol kinase homodimer: a pharmacophore-directed virtual screening approach. J Mol Mod 15:997–1007
Gosztola B, Sárosi S, Németh E (2010) Variability of the essential oil content and composition of chamomile (Matricaria recutita L.) affected by weather conditions. Nat Prod Commun 5:465
Gräwert T, Rohdich F, Span I, Bacher A, Eisenreich W, Eppinger J, Groll M (2009) Structure of active IspH enzyme from Escherichia coli provides mechanistic insights into substrate reduction. Ang Chemie Int Ed 48:5756–5759
Gray J, Kekwick R (1972) The inhibition of plant mevalonate kinase preparations by prenyl pyrophosphates. Biochim Biophys Acta Gen Subj 279:290–296
Green TR, Dennis DT, West CA (1975) Compartmentation of isopentenyl pyrophosphate isomerase and prenyl transferase in developing castor bean endosperm. Biochem Biophys Res Commun 64:976–982
Greenhagen B, Chappell J (2001) Molecular scaffolds for chemical wizardry: learning nature’s rules for terpene cyclases. Proc Natl Acad Sci 98:13479–13481
Grochowski LL, Xu H, White RH (2006) Methanocaldococcus jannaschii uses a modified mevalonate pathway for biosynthesis of isopentenyl diphosphate. J Bact 188:3192–3198
Guda C, Lu S, Scheeff ED, Bourne PE, Shindyalov IN (2004) CE-MC: a multiple protein structure alignment server. Nucl Acids Res 32:W100–W103
Guenther A, Karl T, Harley P, Wiedinmyer C, Palmer P, Geron C (2006) Estimates of global terrestrial isoprene emissions using MEGAN (Model of Emissions of Gases and Aerosols from Nature). Atm Chem Phys 6:3181–3210
Guevara-García A, San Román C, Arroyo A, Cortés ME, de la Luz Gutiérrez-Nava M, León P (2005) Characterization of the Arabidopsis clb6 mutant illustrates the importance of posttranscriptional regulation of the methyl-d-erythritol 4-phosphate pathway. Plant Cell 17:628–643
Guirimand G, Guihur A, Phillips M, Oudin A, Glévarec G, Mahroug S, Melin C, Papon N, Clastre M, Giglioli-Guivarc’h N (2012a) Triple subcellular targeting of isopentenyl diphosphate isomerases encoded by a single gene. Plant Signal Behav 7:0–1
Guirimand GG, Guihur A, Phillips MA, Oudin A, Glévarec GL, Melin CL, Papon N, Clastre M, St-Pierre B, Rodríguez-Concepción M, Burlat V, Courdavault V (2012b) A single gene encodes isopentenyl diphosphate isomerase isoforms targeted to plastids, mitochondria and peroxisomes in Catharanthus roseus. Plant Mol Biol 79:443–459
Hampel D, Mosandl A, Wüst M (2005) Biosynthesis of mono- and sesquiterpenes in carrot roots and leaves (Daucus carota L.): metabolic cross talk of cytosolic mevalonate and plastidial methylerythritol phosphate pathways. Phytochemistry 66:305–311
Hanen F, Riadh K, Samia O, Sylvain G, Christian M, Chedly A (2009) Interspecific variability of antioxidant activities and phenolic composition in Mesembryanthemum genus. Food Chem Toxicol 47:2308–2313
Hemmerlin A, Hoeffler J-F, Meyer O, Tritsch D, Kagan IA, Grosdemange-Billiard C, Rohmer M, Bach TJ (2003a) Cross-talk between the cytosolic mevalonate and the plastidial methylerythritol phosphate pathways in tobacco bright yellow-2 cells. J Biol Chem 278:26666–26676
Hemmerlin A, Rivera SB, Erickson HK, Poulter CD (2003b) Enzymes encoded by the farnesyl diphosphate synthase gene family in the big sagebrush Artemisia tridentata ssp. spiciformis. J Biol Chem 278:32132–32140
Hemmerlin A, Tritsch D, Hartmann M, Pacaud K, Hoeffler J-F, van Dorsselaer A, Rohmer M, Bach TJ (2006) A cytosolic Arabidopsis d-xylulose kinase catalyzes the phosphorylation of 1-deoxy-d-xylulose into a precursor of the plastidial isoprenoid pathway. Plant Physiol 142:441–457
Hemmerlin A, Harwood JL, Bach TJ (2012) A raison d’être for two distinct pathways in the early steps of plant isoprenoid biosynthesis? Prog Lipid Res 51:95–148
Herdendorf TJ, Miziorko HM (2006) Phosphomevalonate kinase: functional investigation of the recombinant human enzyme. Biochemistry 45:3235–3242
Herrero-Jáuregui C, Casado MA, das Graças Bichara Zoghbi M, Célia Martins-da-Silva R (2011) Chemical variability of Copaifera reticulata ducke oleoresin. Chem Biodivers 8:674–685
Hoeffler JF, Hemmerlin A, Grosdemange-Billiard C, Bach TJ, Rohmer M (2002) Isoprenoid biosynthesis in higher plants and in Escherichia coli: on the branching in the methylerythritol phosphate pathway and the independent biosynthesis of isopentenyl diphosphate and dimethylallyl diphosphate. Biochem J 366:573–583
Horbach S, Sahm H, Welle R (1993) Isoprenoid biosynthesis in bacteria: two different pathways? FEMS Microbiol Lett 111:135–140
Hosfield DJ, Zhang Y, Dougan DR, Broun A, Tari LW, Swanson RV, Finn J (2004) Structural basis for bisphosphonate-mediated inhibition of isoprenoid biosynthesis. J Biol Chem 279:8526–8529
Hsieh MH, Chang CY, Hsu SJ, Chen JJ (2008) Chloroplast localization of methylerythritol 4-phosphate pathway enzymes and regulation of mitochondrial genes in ispD and ispE albino mutants in Arabidopsis. Plant Mol Biol 66:663–673
Hsieh FL, Chang TH, Ko TP, Wang AHJ (2010) Enhanced specificity of mint geranyl pyrophosphate synthase by modifying the R-loop interactions. J Mol Biol 404:859–873
Hsieh F-L, Chang T-H, Ko T-P, Wang AH-J (2011) Structure and mechanism of an Arabidopsis medium/long-chain-length prenyl pyrophosphate synthase. Plant Physiol 155:1079–1090
Huang J-Z, Cheng T-C, Wen P-J, Hsieh M-H, Chen F-C (2009) Molecular characterization of the Oncidium orchid HDR gene encoding 1-hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate reductase, the last step of the methylerythritol phosphate pathway. Plant Cell Rep 28:1475–1486
Hunter WN (2007) The non-mevalonate pathway of isoprenoid precursor biosynthesis. J Biol Chem 282:21573–21577
Hyatt DC, Youn B, Zhao Y, Santhamma B, Coates RM, Croteau RB, Kang C (2007) Structure of limonene synthase, a simple model for terpenoid cyclase catalysis. Proc Natl Acad Sci 104:5360–5365
Istvan ES, Deisenhofer J (2001) Structural mechanism for statin inhibition of HMG-CoA reductase. Science 292:1160–1164
Joas J, Vulcain E, Desvignes C, Morales E, Léchaudel M (2012) Physiological age at harvest regulates the variability in postharvest ripening, sensory and nutritional characteristics of mango (Mangifera indica L.) cv. Coghshall due to growing conditions. J Sci Food Agric 92:1282–1290
Jomaa H, Wiesner J, Sanderbrand S, Altincicek B, Weidemeyer C, Hintz M, Tärbachova I, Eberl M, Zeidler J, Lichtenthaler HK (1999) Inhibitors of the nonmevalonate pathway of isoprenoid biosynthesis as antimalarial drugs. Science 285:1573–1576
Jost M, Zocher G, Tarcz S, Matuschek M, Xie X, Li SM, Stehle T (2010) Structure-function analysis of an enzymatic prenyl transfer reaction identifies a reaction chamber with modifiable specificity. J Am Chem Soc 132:17849–17858
Joyard J, Ferro M, Masselon C, Seigneurin-Berny D, Salvi D, Garin J, Rolland N (2009) Chloroplast proteomics and the compartmentation of plastidial isoprenoid biosynthetic pathways. Mol Plant 2:1154–1180
Kalinowska-Tluscik J, Miallau L, Gabrielsen M, Leonard GA, McSweeney SM, Hunter WN (2010) A triclinic crystal form of Escherichia coli 4-diphosphocytidyl-2C-methyl-d-erythritol kinase and reassessment of the quaternary structure. Acta Crystallogr Sect Fb Struct Biol Cryst Commun 66:237–241
Kampranis SC, Ioannidis D, Purvis A, Mahrez W, Ninga E, Katerelos NA, Anssour S, Dunwell JM, Degenhardt JR, Makris AM, Goodenough PW, Johnson CB (2007) Rational conversion of substrate and product specificity in a Salvia monoterpene synthase: structural insights into the evolution of terpene synthase function. Plant Cell 19(6):1994–2005
Kaneda K, Kuzuyama T, Takagi M, Hayakawa Y, Seto H (2001) An unusual isopentenyl diphosphate isomerase found in the mevalonate pathway gene cluster from Streptomyces sp. strain CL190. Proc Natl Acad Sci 98:932–937
Kappers IF, Aharoni A, van Herpen TWJM, Luckerhoff LLP, Dicke M, Bouwmeester HJ (2005) Genetic engineering of terpenoid metabolism attracts bodyguards to Arabidopsis. Science 309:2070–2072
Kasahara H, Hanada A, Kuzuyama T, Takagi M, Kamiya Y, Yamaguchi S (2002) Contribution of the mevalonate and methylerythritol phosphate pathways to the biosynthesis of gibberellins in Arabidopsis. J Biol Chem 277:45188–45194
Kasahara H, Takei K, Ueda N, Hishiyama S, Yamaya T, Kamiya Y, Yamaguchi S, Sakakibara H (2004) Distinct isoprenoid origins of cis- and trans-zeatin biosyntheses in Arabidopsis. J Biol Chem 279:14049–14054
Kawagishi H, Ishiyama D, Mori H, Sakamoto H, Ishiguro Y, Furukawa S, Li J (1997) Dictyophorines A and B, two stimulators of NGF-synthesis from the mushroom Dictyophora indusiata. Phytochemistry 45:1203–1205
Keasling JD (2008) Synthetic biology for synthetic chemistry. ACS Chem Biol 3:64–76
Keeling CI, Weisshaar S, Lin RPC, Bohlmann J (2008) Functional plasticity of paralogous diterpene synthases involved in conifer defense. Proc Natl Acad Sci 105:1085–1090
Kemp LE, Bond CS, Hunter WN (2002) Structure of 2C-methyl-d-erythritol 2, 4-cyclodiphosphate synthase: an essential enzyme for isoprenoid biosynthesis and target for antimicrobial drug development. Proc Natl Acad Sci 99:6591–6596
Kervizic G, Corcos L (2008) Dynamical modeling of the cholesterol regulatory pathway with Boolean networks. BMC Sys Biol 2:99
Kessler A, Baldwin IT (2001) Defensive function of herbivore-induced plant volatile emissions in nature. Science 291:2141–2144
Kiran U, Ram M, Mather Ali Khan SK, Jha P, Alam A, Abdin MZ (2010) Structural and functional characterization of HMG-CoA reductase from Artemisia annua. Bioinformation 5:146
Kirby J, Keasling JD (2009) Biosynthesis of plant isoprenoids: perspectives for microbial engineering. Annu Rev Plant Biol 60:335–355
Kloer DP, Welsch R, Beyer P, Schulz GE (2006) Structure and reaction geometry of geranylgeranyl diphosphate synthase from Sinapis alba. Biochemistry 45:15197–15204
Kobayashi K, Suzuki M, Tang J, Nagata N, Ohyama K, Seki H, Kiuchi R, Kaneko Y, Nakazawa M, Matsui M, Matsumoto S, Yoshida S, Muranaka T (2007) LOVASTATIN INSENSITIVE 1, a novel pentatricopeptide repeat protein, is a potential regulatory factor of isoprenoid biosynthesis in Arabidopsis. Plant Cell Physiol 48:322–331
Koehn FE, Carter GT (2005) The evolving role of natural products in drug discovery. Nat Rev Drug Discov 4:206–220
Köksal M, Hu H, Coates RM, Peters RJ, Christianson DW (2011a) Structure and mechanism of the diterpene cyclase ent-copalyl diphosphate synthase. Nat Chem Biol 7:431–433
Köksal M, Jin Y, Coates RM, Croteau R, Christianson DW (2011b) Taxadiene synthase structure and evolution of modular architecture in terpene biosynthesis. Nature 469:116–120
Köllner TG, O’Maille PE, Gatto N, Boland W, Gershenzon J, Degenhardt J (2006) Two pockets in the active site of maize sesquiterpene synthase TPS4 carry out sequential parts of the reaction scheme resulting in multiple products. Arch Biochem Biophys 448:83–92
Kuzuyama T, Seto H (2003) Diversity of the biosynthesis of the isoprene units. Nat Prod Rep 20:171–183
Lange BM, Ghassemian M (2003) Genome organization in Arabidopsis thaliana: a survey for genes involved in isoprenoid and chlorophyll metabolism. Plant Mol Biol 51:925–948
Lange BM, Wildung MR, McCaskill D, Croteau R (1998) A family of transketolases that directs isoprenoid biosynthesis via a mevalonate-independent pathway. Proc Natl Acad Sci 95:2100–2104
Lange BM, Rujan T, Martin W, Croteau R (2000) Isoprenoid biosynthesis: the evolution of two ancient and distinct pathways across genomes. Proc Natl Acad Sci 97:13172–13177
Laule O, Fürholz A, Chang H-S, Zhu T, Wang X, Heifetz PB, Gruissem W, Lange M (2003) Crosstalk between cytosolic and plastidial pathways of isoprenoid biosynthesis in Arabidopsis thaliana. Proc Natl Acad Sci 100:6866–6871
Lee EK, Jin YW, Park JH, Yoo YM, Hong SM, Amir R, Yan Z, Kwon E, Elfick A, Tomlinson S (2010a) Cultured cambial meristematic cells as a source of plant natural products. Nat Biotech 28:1213–1217
Lee M, Gräwert T, Quitterer F, Rohdich F, Eppinger J, Eisenreich W, Bacher A, Groll M (2010b) Biosynthesis of isoprenoids: crystal structure of the [4F-4S] cluster protein IspG. J Mol Biol 404:600–610
Leivar P, Antolín-Lloveraa M, Ferrero S, Closa M, Arrób M, Ferrer A, Boronat A, Campos N (2011) Multilevel control of Arabidopsis 3-hydroxy-3-methylglutaryl coenzyme A reductase by protein phosphatase 2A. Plant Cell 23:1494–1511
Lesburg CA, Caruthers JM, Paschall CM, Christianson DW (1998) Managing and manipulating carbocations in biology: terpenoid cyclase structure and mechanism. Curr Opin Struc Biol 8:695–703
Liang PH (2009) Reaction kinetics, catalytic mechanisms, conformational changes, and inhibitor design for prenyl transferases. Biochemistry 48:6562–6570
Liang PH, Ko TP, Wang AHJ (2002) Structure, mechanism and function of prenyl transferases. Eur J Biochem 269:3339–3354
Lichtenthaler HK (1999) The 1-deoxy-d-xylulose-5-phosphate pathway of isoprenoid biosynthesis in plants. Annu Rev Plant Biol 50:47–65
Lichtenthaler HK, Rohmer M, Schwender J (2006) Two independent biochemical pathways for isopentenyl diphosphate and isoprenoid biosynthesis in higher plants. Physiol Plant 101:643–652
Liu YL, Guerra F, Wang K, Wang W, Li J, Huang C, Zhu W, Houlihan K, Li Z, Zhang Y (2012) Structure, function and inhibition of the two-and three-domain 4Fe-4S IspG proteins. Proc Natl Acad Sci 109:8558–8563
Long SB, Casey PJ, Beese LS (2002) Reaction path of protein farnesyl transferase at atomic resolution. Nature 419:645–650
Loreto F, Velikova V (2001) Isoprene produced by leaves protects the photosynthetic apparatus against ozone damage, quenches ozone products, and reduces lipid peroxidation of cellular membranes. Plant Physiol 127:1781–1787
Love A, Naik D, Basak SK, Babu S, Pathak N, Babu CR (2009) Variability in foliar essential oils among different morphotypes of Lantana species complexes, and its taxonomic and ecological significance. Chem Biodivers 6:2263–2274
Lynen F (1959) Participation of acyl-coa in carbon chain biosynthesis. J Cell Comp Physiol 54:33–49
Maimone TJ, Baran PS (2007) Modern synthetic efforts toward biologically active terpenes. Nat Chem Biol 3:396–407
Mansouri H, Asrar Z, Mehrabani M (2009) Effects of gibberellic acid on primary terpenoids and Δ9-tetrahydrocannabinol in Cannabis sativa at flowering stage. J Int Plant Biol 51:553–561
Martin DM, Gershenzon J, Bohlmann J (2003) Induction of volatile terpene biosynthesis and diurnal emission by methyl jasmonate in foliage of Norway Spruce. Plant Physiol 132:1586–1599
McAndrew RP, Peralta-Yahya PP, DeGiovanni A, Pereira JH, Hadi MZ, Keasling JD, Adams PD (2011) Structure of a three-domain sesquiterpene synthase: a prospective target for advanced biofuels production. Structure 19:1876–1884
Mills NS (2006) Chem Draw Ultra 10.0 Cambridge Soft, (Computer Software Review). J Am Chem Soc 128:13649–13650
Miziorko HM (2011) Enzymes of the mevalonate pathway of isoprenoid biosynthesis. Arch Biochem Biophys 505:131–143
Mo H, Yeganehjoo H, Shah A, Mo WK, Soelaiman IN, Shen CL (2012) Mevalonate-suppressive dietary isoprenoids for bone health. J Nutr Biochem 23(12):1543–1551
Morris WL, Ducreux LJM, Hedden P, Millam S, Taylor MA (2006) Overexpression of a bacterial 1-deoxy- d -xylulose 5-phosphate synthase gene in potato tubers perturbs the isoprenoid metabolic network: implications for the control of the tuber life cycle. J Exp Bot 57:3007–3018
Munos JW, Pu X, Mansoorabadi SO, Kim HJ, Liu H (2009) A secondary kinetic isotope effect study of the 1-deoxy-d-xylulose-5-phosphate reductoisomerase-catalyzed reaction: evidence for a retroaldol-aldol rearrangement. J Am Chem Soc 131:2048–2049
Muñoz-Bertomeu J, Arrillaga I, Ros R, Segura J (2006) Up-regulation of 1-deoxy-d-xylulose-5-phosphate synthase enhances production of essential oils in transgenic spike lavender. Plant Physiol 142:890–900
Muntendam R, Melillo E, Ryden A, Kayser O (2009) Perspectives and limits of engineering the isoprenoid metabolism in heterologous hosts. App Microbiol Biotech 84:1003–1019
Ôba K, Uritani I (1979) Biosynthesis of furano-terpenes by sweet potato cell culture. Plant Cell Physiol 20:819–826
Ôba K, Kondo K, Doke N, Uritani I (1985) Induction of 3-hydroxy-3-methylglutaryl CoA reductase in potato tubers after slicing, fungal infection or chemical treatment, and some properties of the enzyme. Plant Cell Physiol 26:873–880
Ogura K, Koyama T, Sagami H (1997) Polyprenyl diphosphate synthases. Subcell Biochem 28:57–87
Okada K, Hase T (2005) Cyanobacterial non-mevalonate pathway. J Biol Chem 280:20672–20679
Okada K, Kasahara H, Yamaguchi S, Kawaide H, Kamiya Y, Nojiri H, Yamane H (2008) Genetic evidence for the role of isopentenyl diphosphate isomerases in the mevalonate pathway and plant development in Arabidopsis. Plant Cell Physiol 49:604–616
Okada A, Okada K, Miyamoto K, Koga J, Shibuya N, Nojiri H, Yamane H (2009) OsTGAP1, a bZIP transcription factor, coordinately regulates the inductive production of diterpenoid phytoalexins in rice. J Biol Chem 284(39):26510–26518
Oldfield E, Lin FY (2012) Terpene biosynthesis: modularity rules. Ang Chem Int Ed 51:1124–1137
Osbourn A (2010) Gene clusters for secondary metabolic pathways: an emerging theme in plant biology. Plant Physiol 154:531–535
Paetzold H, Garms S, Bartram S, Wieczorek J, Urós-Gracia E-M, Rodríguez-Concepción M, Boland W, Strack D, Hause B, Walter MH (2010) The isogene 1-deoxy-d-xylulose 5-phosphate synthase 2 controls isoprenoid profiles, precursor pathway allocation, and density of tomato trichomes. Mol Plant 3:904–916
Page JE, Hause G, Raschke M, Gao W, Schmidt JR, Zenk MH, Kutchan TM (2004) Functional analysis of the final steps of the 1-Deoxy-d-xylulose 5-phosphate (DXP) pathway to isoprenoids in plants using virus-induced gene silencing. Plant Physiol 134:1401–1413
Pandian S, Saengchjan S, Raman T (1981) An alternative pathway for the biosynthesis of isoprenoid compounds in bacteria. Biochem J 196:675
Patil RA, Kolewe ME, Normanly J, Walker EL, Roberts SC (2012) Contribution of taxane biosynthetic pathway gene expression to observed variability in paclitaxel accumulation in taxus suspension cultures. Biotechnol J 7:418–427
Pecetti L, Tava A, Romani M, Cecotti R, Mella M (2012) Variation in terpene and linear-chain hydrocarbon content in Yarrow (Achillea millefolium L.) germplasm from the Rhaetian Alps, Italy. Chem Biodivers 9:2282–2294
Peters RJ, Ravn MM, Coates RM, Croteau RB (2001) Bifunctional abietadiene synthase: free diffusive transfer of the (+)-copalyl diphosphate intermediate between two distinct active sites. J Am Chem Soc 123:8974–8978
Peters RJ, Carter OA, Zhang Y, Matthews BW, Croteau RB (2003) Bifunctional abietadiene synthase: mutual structural dependence of the active sites for protonation-initiated and ionization-initiated cyclizations. Biochemistry 42:2700–2707
Phillips MA, D’Auria JC, Gershenzon J, Pichersky E (2008a) The Arabidopsis thaliana type I isopentenyl diphosphate isomerases are targeted to multiple subcellular compartments and have overlapping functions in isoprenoid biosynthesis. Plant Cell 20:677–696
Phillips MA, León P, Boronat A, Rodríguez-Concepción M (2008b) The plastidial MEP pathway: unified nomenclature and resources. Trends Plant Sci 13:619–623
Plotto A, Baldwin E, McCollum G, Manthey J, Narciso J, Irey M (2010) Effect of liberibacter infection (Huanglongbing or “Greening” disease) of citrus on orange juice flavor quality by sensory evaluation. J Food Sci 75:S220–S230
Pojer F, Ferrer J-L, Richard SB, Nagegowda DA, Chye M-L, Bach TJ, Noel JP (2006) Structural basis for the design of potent and species-specific inhibitors of 3-hydroxy-3-methylglutaryl CoA synthases. Proc Natl Acad Sci 103:11491–11496
Porter JW, Spurgeon SL (1981) Biosynthesis of isoprenoid compounds. Wiley, New York
Poulter CD (1990) Biosynthesis of non-head-to-tail terpenes. Formation of 1′-1 and 1′-3 linkages. Acc Chem Res 23:70–77
Prlić A, Bliven S, Rose PW, Bluhm WF, Bizon C, Godzik A, Bourne PE (2010) Pre-calculated protein structure alignments at the RCSB PDB website. Bioinformatics 26:2983–2985
Proteau PJ (2004) 1-Deoxy-d-xylulose 5-phosphate reductoisomerase: an overview. Bioorg Chem 32:483–493
Qi X, Bakht S, Leggett M, Maxwell C, Melton R, Osbourn A (2004) A gene cluster for secondary metabolism in oat: implications for the evolution of metabolic diversity in plants. Proc Natl Acad Sci USA 101(21):8233–8238
Rekittke I, Wiesner J, Röhrich R, Demmer U, Warkentin E, Xu W, Troschke K, Hintz M, No JH, Duin EC (2008) Structure of (E)-4-hydroxy-3-methyl-but-2-enyl diphosphate reductase, the terminal enzyme of the non-mevalonate pathway. J Am Chem Soc 130:17206–17207
Rekittke I, Nonaka T, Wiesner J, Demmer U, Warkentin E, Jomaa H, Ermler U (2011) Structure of the E-1-hydroxy-2-methyl-but-2-enyl-4-diphosphate synthase (GcpE) from Thermus thermophilus. FEBS Lett 585:447–451
Reumann S, Babujee L, Ma C, Wienkoop S, Siemsen T, Antonicelli GE, Rasche N, Lüder F, Weckwerth W, Jahn O (2007) Proteome analysis of Arabidopsis leaf peroxisomes reveals novel targeting peptides, metabolic pathways, and defense mechanisms. Plant Cell 19:3170–3193
Ricagno S, Grolle S, Bringer-Meyer S, Sahm H, Lindqvist Y, Schneider G (2004) Crystal structure of 1-deoxy-d-xylulose-5-phosphate reductoisomerase from Zymomonas mobilis at 1.9-Å resolution. Biochem Biophys Acta Proteins Proteomics 1698:37–44
Richard SB, Bowman ME, Kwiatkowski W, Kang I, Chow C, Lillo AM, Cane DE, Noel JP (2001) Structure of 4-diphosphocytidyl-2-C methylerythritol synthetase involved in mevalonate-independent isoprenoid biosynthesis. Nat Struct Mol Biol 8:641–648
Richard SB, Ferrer J-L, Bowman ME, Lillo AM, Tetzlaff CN, Cane DE, Noel JP (2002) Structure and mechanism of 2-C-methyl-d-erythritol 2,4-cyclodiphosphate synthase. J Biol Chem 277:8667–8672
Roberts S, Kolewe M (2010) Plant natural products from cultured multipotent cells. Nat Biotech 28:1175–1176
Rodríguez-Concepción M, Boronat A (2002) Elucidation of the methylerythritol phosphate pathway for isoprenoid biosynthesis in bacteria and plastids. A metabolic milestone achieved through genomics. Plant Physiol 130:1079–1089
Rodríguez-Concepción M, Querol J, Lois LMA, Imperial S, Boronat A (2003) Bioinformatic and molecular analysis of hydroxymethyl butenyl diphosphate synthase (GCPE) gene expression during carotenoid accumulation in ripening tomato fruit. Planta 217:476–482
Rodríguez-Suárez C, Atienza SG, Pistón F (2011) Allelic variation, alternative splicing and expression analysis of Psy1 gene in Hordeum chilense Roem.et Schult. PLoS ONE 6:e19885
Rohdich F, Hecht S, Gartner K, Adam P, Krieger C, Amslinger S, Arigoni D, Bacher A, Eisenreich W (2002) Studies on the nonmevalonate terpene biosynthetic pathway: metabolic role of IspH (LytB) protein. Proc Natl Acad Sci 99:1158–1163
Rohloff J, Mordal R, Dragland S (2004) Chemotypical variation of Tansy (Tanacetum vulgare L.) from 40 different locations in Norway. J Agric Food Chem 52:1742–1748
Rohmer M (1999) The discovery of a mevalonate-independent pathway for isoprenoid biosynthesis in bacteria, algae and higher Plants. Nat Prod Rep 16:565–574
Rohmer M (2003) Mevalonate-independent methylerythritol phosphate pathway for isoprenoid biosynthesis. Elucidation and distribution. Pure App Chem 75:375–388
Romanowski MJ, Bonanno JB, Burley SK (2002) Crystal structure of the Streptococcus pneumoniae phosphomevalonate kinase, a member of the GHMP kinase superfamily. Proteins 47(4):568–571
Rouis Z, Elaissi A, Abid NB, Lassoued MA, Cioni PL, Flamini G, Aouni M (2012) Chemical composition and intraspecific variability of the essential oils of five populations of Hypericum triquetrifolium Turra growing in North Tunisia. Chem Biodivers 9:806–816
Ruzicka L (1953) The isoprene rule and the biogenesis of terpenic compounds. Cell Mol Life Sci 9:357–367
Sacchettini JC, Poulter CD (1997) Creating isoprenoid diversity. Science 277:1788–1789
Saleh O, Haagen Y, Seeger K, Heide L (2009) Prenyl transfer to aromatic substrates in the biosynthesis of aminocoumarins, meroterpenoids and phenazines: the ABBA prenyltransferase family. Phytochemistry 70:1728–1738
Sapir-Mir M, Mett A, Belausov E, Tal-Meshulam S, Frydman A, Gidoni D, Eyal Y (2008) Peroxisomal localization of Arabidopsis isopentenyl diphosphate isomerases suggests that part of the plant isoprenoid mevalonic acid pathway is compartmentalized to peroxisomes. Plant Physiol 148:1219–1228
Schulte AE, van der Heijden R, Verpoorte R (1999) Purification and characterization of phosphomevalonate kinase from Catharanthus roseus. Phytochemistry 52:975–983
Schulze-Siebert D, Schultz G (1987) Full autonomy in isoprenoid synthesis in spinach chloroplasts. Plant Physiol Biochem 25:145–153
Schwarz M, Arigoni D (1999) Ginkgolide biosynthesis. Comp Nat Prod Chem 2:367–399
Seetang-Nun Y, Sharkey TD, Suvachittanont W (2008) Molecular cloning and characterization of two cDNAs encoding 1-deoxy-d-xylulose 5-phosphate reductoisomerase from Hevea brasiliensis. J Plant Physiol 165:991–1002
Seto H, Watanabe H, Furihata K (1996) Simultaneous operation of the mevalonate and non-mevalonate pathways in the biosynthesis of isopentenyl diphosphate in Streptomyces aeriouvifer. Tetrahed Lett 37:7979–7982
Sharkey TD, Wiberley AE, Donohue AR (2008) Isoprene emission from plants: why and how. Ann Bot 101:5–18
Shen G, Pang Y, Wu W, Liao Z, Zhao L, Sun X, Tang K (2006) Cloning and characterization of a root-specific expressing gene encoding 3-hydroxy-3-methylglutaryl coenzyme A reductase from Ginkgo biloba. Mol Biol Rep 33:117–127
Shimura K, Okada A, Okada K, Jikumaru Y, Ko KW, Toyomasu T, Sassa T, Hasegawa M, Kodama O, Shibuya N, Koga J, Nojiri H, Yamane H (2007) Identification of a biosynthetic gene cluster in rice for momilactones. J Biol Chem 282(47):34013–34018
Simkin AJ, Guirimand G, Papon N, Courdavault V, Thabet I, Ginis O, Bouzid S, Giglioli-Guivarc’h N, Clastre M (2011) Peroxisomal localisation of the final steps of the mevalonic acid pathway in planta. Planta 234:903–914
Singsaas EL, Lerdau M, Winter K, Sharkey TD (1997) Isoprene increases thermotolerance of isoprene-emitting species. Plant Physiol 115:1413–1420
Sivy TL, Fall R, Rosenstiel TN (2011) Evidence of isoprenoid precursor toxicity in Bacillus subtilis. Biosci Biotech Biochem 75:2376–2383
Skorupinska-Tudek K, Poznanski J, Wojcik J, Bienkowski T, Szostkiewicz I, Zelman-Femiak M, Bajda A, Chojnacki T, Olszowska O, Grunler J, Meyer O, Rohmer M, Danikiewicz W, Swiezewska E (2008) Contribution of the mevalonate and methylerythritol phosphate pathways to the biosynthesis of dolichols in plants. J Biol Chem 283:21024–21035
Smanski MJ, Peterson RM, Huang SX, Shen B (2012) Bacterial diterpene synthases: new opportunities for mechanistic enzymology and engineered biosynthesis. Curr Opin Chem Biol 16:132–141
Smit A, Mushegian A (2000) Biosynthesis of isoprenoids via mevalonate in Archaea: the lost pathway. Genome Res 10:1468–1484
Solymosi K, Morandi D, Boka K, Boddi B, Schoefs B (2012) High biological variability of plastids, photosynthetic pigments and pigment forms of leaf primordia in buds. Planta 235:1035–1049
Spacklen DV, Bonn B, Carslaw KS (2008) Boreal forests, aerosols and the impacts on clouds and climate. Philos Trans Math Phys Eng Sci 366:4613–4626
Span I, Wang K, Wang W, Zhang Y, Bacher A, Eisenreich W, Li K, Schulz C, Oldfield E, Groll M (2012) Discovery of acetylene hydratase activity of the iron-sulphur protein IspH. Nat Commun 3:1042
Srivastava N, Akhila A (2010) Biosynthesis of andrographolide in Andrographis paniculata. Phytochemistry 71:1298–1304
Starks CM, Back K, Chappell J, Noel JP (1997) Structural basis for cyclic terpene biosynthesis by tobacco 5-epi-aristolochene synthase. Science 277:1815–1820
Steinbacher S, Kaiser J, Wungsintaweekul J, Hecht S, Eisenreich W, Gerhardt S, Bacher A, Rohdich F (2002) Structure of 2C-methyl-d-erythritol-2,4-cyclodiphosphate synthase involved in mevalonate-independent biosynthesis of isoprenoids. J Mol Biol 316:79–88
Sun G, Budde RJA (1997) Requirement for an additional divalent metal cation to activate protein tyrosine kinases. Biochemistry 36:2139–2146
Suzuki M, Nakagawa S, Kamide Y, Kobayashi K, Ohyama K, Hashinokuchi H, Kiuchi R, Saito K, Muranaka T, Nagata N (2009) Complete blockage of the mevalonate pathway results in male gametophyte lethality. J Exp Bot 60:2055–2064
Swaminathan S, Morrone D, Wang Q, Fulton DB, Peters RJ (2009) CYP76M7 is an ent-cassadiene C11α-hydroxylase defining a second multifunctional diterpenoid biosynthetic gene cluster in rice. Plant Cell 21:3315–3325
Takahashi S, Koyama T (2006) Structure and function of cis-prenyl chain elongating enzymes. Chem Rec 6:194–205
Tarshis LC, Yan M, Poulter CD, Sacchettini JC (1994) Crystal structure of recombinant farnesyl diphosphate synthase at 2.6 Ang. resolution. Biochemistry 33:10871–10877
Tello M, Kuzuyama T, Heide L, Noel J, Richard S (2008) The ABBA family of aromatic prenyl transferases: broadening natural product diversity. Cell Mol Life Sci 65:1459–1463
Thoma R, Schulz-Gasch T, D’Arcy B, Benz J, Aebi J, Dehmlow H, Hennig M, Stihle M, Ruf A (2004) Insight into steroid scaffold formation from the structure of human oxidosqualene cyclase. Nature 432:118–122
Thompson A, Cooper J, Ingram LL (2006) Distribution of terpenes in heartwood and sapwood of loblolly pine. For Prod J 56:7–8
Thoss V, O’Reilly-Wapstra J, Iason GR (2007) Assessment and implications of intraspecific and phenological variability in monoterpenes of Scots pine (Pinus sylvestris) foliage. J Chem Ecol 33:477–491
Trainer M, Williams E, Parrish D, Buhr M, Allwine E, Westberg H, Fehsenfeld F, Liu S (1987) Models and observations of the impact of natural hydrocarbons on rural ozone. Nature 329:705–707
Trapp SC, Croteau RB (2001) Genomic organization of plant terpene synthases and molecular evolutionary implications. Genetics 158:811–832
Treharne K, Mercer E, Goodwin T (1966) Incorporation of [14C] carbon dioxide and [2-14C] mevalonic acid into terpenoids of higher plants during chloroplast development. Biochem J 99:239
Tritsch D, Hemmerlin A, Bach TJ, Rohmer M (2010) Plant isoprenoid biosynthesis via the MEP pathway: in vivo IPP/DMAPP ratio produced by (E)-4-hydroxy-3-methylbut-2-enyl diphosphate reductase in tobacco BY-2 cell cultures. FEBS Lett 584:129–134
Tully J (2009) A Victorian ecological disaster: imperialism, the telegraph, and Gutta-Percha. J World Hist 20:559–579
van der Meer JY, Hirsch AKH (2012) The isoprenoid-precursor dependence of Plasmodium spp. Nat Prod Rep 29:721–728
Vandermoten S, Santini S, Haubruge A, Heuze F, Francis F, Brasseur R, Cusson M, Charloteaux B (2009) Structural features conferring dual geranyl/farnesyl diphosphate synthase activity to an aphid prenyltransferase. Insect Biochem Mol Biol 39:707–716
Velikova V, Loreto F (2005) On the relationship between isoprene emission and thermotolerance in Phragmites australis leaves exposed to high temperatures and during the recovery from a heat stress. Plant, Cell Environ 28:318–327
Vollack KU, Dittrich B, Ferrer A, Boronat A, Bach TJ (1994) Two radish genes for 3-hydroxy-3-methylglutaryl-CoA reductase isozymes complement mevalonate auxotrophy in a yeast mutant and yield membrane-bound active enzyme. J Plant Physiol 143:479–487
Voynova NE, Rios SE, Miziorko HM (2004) Staphylococcus aureus mevalonate kinase: isolation and characterization of an enzyme of the isoprenoid biosynthetic pathway. J Bacteriol 186:61–67
Vranova E, Coman D, Gruissem W (2012) Structure and dynamics of the isoprenoid pathway network. Mol Plant 5:318–333
Wagner AM, Krab K (2006) The alternative respiration pathway in plants: role and regulation. Physiol Plant 95:318–325
Wang KC, Ohnuma S (2000) Isoprenyl diphosphate synthases. Biochem Biophys Acta Mol Cell Biol Lipids 1529:33–48
Wanke M, Skorupinska-Tudek K, Swiezewska E (2001) Isoprenoid biosynthesis via 1-deoxy-d-xylulose 5-phosphate/2-C-methyl-d-erythritol 4-phosphate (DOXP/MEP) pathway. Acta Biochim Pol 48:663–672
Wendt KU, Schulz GE (1998) Isoprenoid biosynthesis: manifold chemistry catalyzed by similar enzymes. Structure 6:127–133
Wendt K, Lenhart A, Schulz G (1999) The structure of the membrane protein squalene-hopene cyclase at 2.0 Å resolution. J Mol Biol 286:175–187
Wendt KU, Schulz GE, Corey EJ, Liu DR (2000) Enzyme mechanisms for polycyclic triterpene formation. Ang Chemie Int Ed 39:2812–2833
Whittington DA, Wise ML, Urbansky M, Coates RM, Croteau RB, Christianson DW (2002) Bornyl diphosphate synthase: structure and strategy for carbocation manipulation by a terpenoid cyclase. Proc Natl Acad Sci 99:15375–15380
Wiemer AJ, Hohl RJ, Wiemer DF (2009) The intermediate enzymes of isoprenoid metabolism as anticancer targets. Anticancer Agents Med Chem 9:526–542
Wildermann PR, Xu M, Jin Y, Coates RM, Peters RJ (2004) Identification of syn-pimara-7, 15-diene synthase reveals functional clustering of terpene synthases involved in rice phytoalexin/allelochemical biosynthesis. Plant Physiol 135:2098–2105
Wille A, Zimmermann P, Vranova E, FÃrholz A, Laule O, Bleuler S, Hennig L, Prelic A, Von Rohr P, Thiele L (2004) Sparse graphical Gaussian modeling of the isoprenoid gene network in Arabidopsis thaliana. Genome Biol 5:R92
Williams SL, McCammon JA (2009) Conformational dynamics of the flexible catalytic loop in Mycobacterium tuberculosis 1,5-deoxy-xylulose 5,-phosphate reductoisomerase. Chem Biol Drug Res 73:26–38
Witschel MC, Höffken HW, Seet M, Parra L, Mietzner T, Thater F, Niggeweg R, Röhl F, Illarionov B, Rohdich F, Kaiser J, Fischer M, Bacher A, Diederich F (2011) Inhibitors of the herbicidal target IspD: allosteric site binding. Angew Chem Int Ed Engl 50:7931–7935
Xiang S, Usunow G, Lange G, Busch M, Tong L (2007) Crystal structure of 1-deoxy-d-xylulose 5-phosphate synthase, a crucial enzyme for isoprenoids biosynthesis. J Biol Chem 282:2676–2682
Xie Z, Kapteyn J, Gang DR (2008) A systems biology investigation of the MEP/terpenoid and shikimate/phenylpropanoid pathways points to multiple levels of metabolic control in sweet basil glandular trichomes. Plant J 54:349–361
Yamaguchi S, Saito T, Abe H, Yamane H, Murofushi N, Kamiya Y (1996) Molecular cloning and characterization of a cDNA encoding the gibberellin biosynthetic enzyme ent-kaurene synthase B from pumpkin (Cucurbita maxima L.). Plant J 10:203–213
Ye Y, Godzik A (2003) Flexible structure alignment by chaining aligned fragment pairs allowing twists. Bioinformatics 19:ii246–ii255
Zabin I (1957) The formation of radioactive lycopene in ripening tomatoes. J Biol Chem 226:851–859
Zhang Y (2008) I-TASSER server for protein 3D structure prediction. BMC Bioinformatics 9:40
Zhang C, Liu L, Xu H, Wei Z, Wang Y, Lin Y, Gong W (2007) Crystal structures of human IPP isomerase: new insights into the catalytic mechanism. J Mol Biol 366:1437–1446
Zhou D, White RH (1991) Early steps of isoprenoid biosynthesis in Escherichia coli. Biochem J 273:627
Zhou T, Daugherty M, Grishin NV, Osterman AL, Zhang H (2000) Structure and mechanism of homoserine kinase: prototype for the GHMP kinase superfamily. Structure 8:1247–1257
Acknowledgments
Authors’ thanks are due to Director NIPGR for encouragement, the Innovative Young Biotechnologist Award (IYBA) grant to GY for financial support, the Biotechnology Information System Network (BTISNET) program of Department of Biotechnology (DBT), Govt of India, for computational resources, the Council of Scientific and Industrial Research (CSIR) for Senior Research Fellowship (SRF) to SK and PP, the DBT for SRF to GM, and finally, to the anonymous reviewer for his thorough review, which significantly contributed to improving this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Sangita Kumari and Piyush Priya contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kumari, S., Priya, P., Misra, G. et al. Structural and biochemical perspectives in plant isoprenoid biosynthesis. Phytochem Rev 12, 255–291 (2013). https://doi.org/10.1007/s11101-013-9284-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11101-013-9284-6