Summary
Since the discovery of Helicobacter pylori our views concerning the pathogenesis and treatment of gastroduodenal disease have changed dramatically. Most individuals infected with this organism remain asymptomatic throughout life. However, some manifest clinical disease, such as gastric and duodenal ulcer. Recently results have also implicated H. pylori infection in the development of gastric lymphoma of mucosa-associated lymphoid tissue and gastric cancer. Variations of an individual’s immune response based on inheritance, age or environmental factors could explain the diverse clinical outcomes of the infection. However, our understanding concerning these mechanisms is still very incomplete.
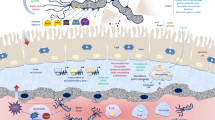
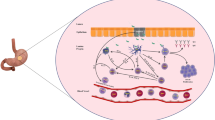
Marked neutrophilic infiltration within the gastric mucosa induced by bacterial components may account for acute mucosal damage. Chronic infection is accompanied by T cell and B cell infiltration into lymphoid follicles, which could contribute to atrophy and malignant transformation of glandular epithelium.
Antimicrobial therapy can eradicate the organism, thus alleviating the host’s immune response. More efficient treatment regimens such as the combination of the proton pump inhibitor omeprazole with amoxicillin are now being used. These combinations have fewer adverse effects and improve patient compliance.
Oral vaccines may be useful for the prophylactic treatment of large populations in whom the incidence of H. pylori infection is especially high. The scarcity of studies on vaccine development may be attributed to 3 factors: (a) lack of knowledge of suitable immunogens for immunisation; (b) lack of appropriate animal models; (c) the poorly characterised cellular immune response towards specific antigens of the organism. Further research into the immunopathogenesis of H. pylori-related disease is necessary.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Krienitz W. Uber das Auftreten von Spirochaeten verschiedener Form im Mageninhalt bei Carcinoma ventriculi. Dtsh Med Wochenschr 1906; 22: 872–82
Doenges JL. Spirochaetes in the gastric glands of Macacus rhesus and humans without definite history of related disease. Pro Soc Exp Biol Med 1938; 38: 536–8
Warren JR, Marshall BJ. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet 1983; 1: 1273–5
Marshall BJ, Warren JR. Unidentified curved bacilli in the stomach of patients with gastritis and peptic ulceration. Lancet 1984; 1: 1311–12
Von Wulffen H, Heesemann J, Butzow GH, et al. Detection of Campylobacter pylori in patients with antrum gastritis and peptic ulcers by culture, complement fixation test and immunoblot. J Clin Microbiol 1986; 24: 716–20
Graham DY, Klein PD, Opekun AR, et al. Effect of age on the frequency of active Campylobacter pylori infection diagnosed by the [13C]urea breath test in normal subjects and patients with peptic ulcer disease. J Infect Dis 1988; 157: 777–80
Blaser MJ. Gastric Campylobacter-like organisms, gastritis and peptic ulcer disease. Gastroenterology 1987; 93: 371–83
Parsonnet J, Friedman GD, Vandersteen DP, et al. Helicobacter pylori infection and the risk of gastric carcinoma. N Engl J Med 1991; 325: 1127–31
Forman D. An international association between Helicobacter pylori infection and gastric cancer. Lancet 1993; 341: 1359–62
Craanen ME, Decker W, Blok P, et al. Intestinal metaplasia and Helicobacter pylori: an endoscopic-bioptic study of the gastric antrum. Gut 1992; 33: 16–20
Marshall B, Armstrong J, McGechie D, et al. Attempt to fulfil Koch’s postulates for pyloric Campylobacter. Med J Aust 1985; 152: 436–9
Morris A, Nicholson G. Ingestion of Campylobacter pylori causes gastritis and raised fasting gastric pH. Am J Gastroenterol 1987; 87: 192–99
Hazell SL, Lee A, Brady L, et al. Campylobacter pylori and gastritis: association with intercellular spaces and adaption to an environment of mucus as important factors in colonization of the gastric epithelium. J Infect Dis 1986; 153: 658–63
Vargas M, Lee A, Fox JG, et al. Inhibition of acid secretion from parietal cells by non-human-infecting Helicobacter species: a factor in colonization of gastric mucosa? Infect Immun 1991; 59: 3694–9
Marshall BJ, Barrett L, Prakash C, et al. Urea protects C. pylori from the bactericidal effect of acid. Gastroenterology 1990; 99: 697–702
Dunn BE. Pathogenic mechanisms of Helicobacter pylori. Gastroenterol Clin North Am 1993; 22: 43–57
Boren T, Falk P, Roth KA, et al. Attachment of Helicobacter pylori to human gastric epithelium mediated by blood group antigens. Science 1993; 262: 1892–5
Cover TL, Blaser MJ. Purification and characterization of the vacuolating toxin from Helicobacter pylori. J Biol Chem 1992; 267: 10570–5
Figura N, Gugliemetti P, Rossolini A, et al. Cytotoxin production by Campylobacter pylori strains isolated from patients with peptic ulcers and from patients with chronic gastritis only. J Clin Microbiol 1989; 27: 225–6
Slomiany BL, Nishikawa H, Piotrowski J, et al. Lipolytic activity of Campylobacter pylori: effect of sofalocone. Digestion 1989; 43: 33–40
Wick MJ. Molecular cross talk between clinical cells and pathogenic microorganisms. Cell 1991; 67: 651–9
Bode G, Malfertheiner P, Ditschuneit H. Pathogenic implications of ultrastructural findings in Campylobacter pylori related gastroduodenal diseases. Scand J Gastroenterol 1988; 23 Suppl. 142: 25–39
Anderson LP, Hoick S. Possible evidence of invasiveness of Helicobacter (Campylobacter) pylori. Eur J Clin Microbiol Infect Dis 1990; 9: 135–8
Thomsen IL, Gavin JB, Tasman-Jones C. Relation of Helicobacter pylori to the human gastric mucosa in chronic gastritis of the antrum. Gut 1990; 31: 1230–6
Bayerdorffer E, Lehn N, Hatz RA, et al. Different expression of Helicobacter pylori gastritis in antrum and body. Gastroenterology 1992; 102: 1575–82
Kozol R, Domanowski A, Jaszewski R, et al. Neutrophil chemotaxis in gastric mucosa — a signal-to-response comparison. Dig Dis Sci 1991; 36: 1277–80
Denizot Y, Raibaud P, Ducluzeau R, et al. Modulation of gut wall Paf-acether and precursors by intestinal microflora. Res Immunol 1990; 141: 235–40
Rosam AC, Wallace JL, Whittle BJR. Potent ulcerogenic actions of platelet-activating factor on the stomach. Nature 1986; 319: 54–6
Wallace JL, Steel G, Whittle BJR, et al. Evidence for platelet-activating factor (PaF) as a mediator of endotoxin-induced gastrointestinal damage in the rat: effects of three Paf antagonists. Gastroenterology 1987; 93: 765–73
Denizot Y, Sobhani I, Rambaud JC, et al. Paf-acether synthesis by Helicobacter pylori. Gut 1990; 31: 1242–5
Wallace N, Beck PL, Morris GP. Is there a role for leukotrienes as mediators of ethanol-induced gastric mucosal damage? Am J Physiol 1988; 254: G117–23
Crabtree JE, Peichl P, Wyatt JI, et al. Gastric interleukin-8 and IgA IL-8 autoantibodies in Helicobacter pylori infection. Scand J Immunol 1993; 37: 65–70
Marasco WA, Phan SH, Krutzsch H, et al. Purification and identification of formyl-methionyl-leucylphenylanlanine as the major peptide neutrophil chemotactic factor produced by Escherichia coli. J Biol Chem 1984; 259: 5430–9
Mooney C, Keenan J, Munster D, et al. Neutrophil activation by Helicobacter pylori. Gut 1991; 32: 853–57
Suzuki M, Soichiro M, Suematsu M, et al. Helicobacter pylori elicits gastric mucosal cell damage associated with neutrophil-derived toxic oxidants. Eur J Gastroenterol Hepatol 1993; 5 Suppl. 1: S35–9
Nielsen H, Andersen LP. Activation of human phagocyte oxidative metabolism by Helicobacter pylori. Gastroenterology 1992; 103: 1747–53
Ferly DM, Butt TJ, Broom MF, et al. Bacterial chemotactic oligopeptides and the intestinal mucosal barrier. Gastroenterology 1989; 97: 61–7
Nielson H, Andersen LP. Chemotactic activity of Helicobacter pylori sonicate for human polymorphonuclear leucocytes and monocytes. Gut 1992; 33: 738–42
Yoshida N, Granger DN, Evans Jr DJ, et al. Mechanisms involved in Helicobacter pylori-induced inflammation. Gastroenterology 1993; 105: 1431–40
Hatz RA, Brooks WP, Enders G, et al. Characterization of vascular adhesion molecules in H. pylori infected human gastric mucosa. Acta Gastroenterol Belg 1993; 56: 51
Weiss SJ. Tissue destruction by neutrophils. N Engl J Med 1989; 320: 365–76
Suzuki M, Miura S, Suematsu M, et al. Helicobacter pylori elicits gastric mucosal cell damage associated with neutrophil-derived toxic oxidants. Eur J Gastroenterol Hepatol 1993; 5 Suppl. 1: 35–9
Stolte M, Eidt S. Lymphoid follicles in antral mucosa: immune response to Campylobacter pylori? J Clin Pathol 1989; 42: 1269–71
Brooks WP, Meimarakis G, Hatz RA, et al. Isolation and phenotypical characterisation of T-lymphocytes isolated from the gastric mucosa of patients with and without Helicobacter pylori. In: Gasbanini G, Pretolani S, editors. Basic and clinical aspects of Helicobacter pylori infection. Berlin, Heidelberg: Springer Verlag, 1994: 113–8
Kirchner T, Melber A, Fishbach W, et al. Immunohistological patterns of the local immune response in Helicobacter pylori gastritis. In: Malfartheimer P, Ditchuneit H, editors. Helicobacter pylori: gastritis and peptic ulcer. Heidelberg: Springer Verlag, 1990: 231–22
Wootherspoon AC, Hidalgo CO, Falzon MR, et al. Helicobacter pylori-associated gastritis and primary B-cell gastric lymphoma. Lancet 1991; 338: 1175–6
Zuckerberg LR, Ferry JA, Southern JF, et al. Lymphoid infiltrates of the stomach. Evaluation of histologic criteria for the diagnosis of low-grade gastric lymphoma on endoscopic biopsy specimens. Am J Surg Pathol 1990; 14: 1087–99
Stolte M. Helicobacter pylori gastritis and gastric MALT-lymphoma. Lancet 1992; 335: 745–6
Hussel T, Isaacson PG, Crabbree JE, et al. The response of cells from low-grade B-cell gastric lymphomas of mucosa-associated lymphoid tissue to Helicobacter pylori. Lancet 1993; 342: 571–4
Wootherspoon AC, Doglioni C, Diss TC, et al. Regression of primary low-grade B-cell gastric lymphoma of mucosa-associated lymphoid tissue after eradication of Helicobacter pylori. Lancet 1993; 342: 575–7
Karttunen R, Andersson G, Poikonen K, et al. Helicobacter pylori induces lymphocyte activation in peripheral blood cultures. Clin Exp Immunol 1990; 82: 485–8
Karttunen R. Blood lymphocyte proliferation, cytokine secretion and appearance of T cells with activation surface markers in cultures with Helicobacter pylori: comparison of the responses of subjects with and without antibodies to H. pylori. Clin Exp Immunol 1991; 84: 1–5
Knipp U, Birkholz S, Kaup W, et al. Immune suppressive effects of Helicobacter pylori on human peripheral blood mononuclear cells. Med Microbiol Immunol 1993; 182: 63–76
Mai UEH, Perez-Perez GI, Wahl LM, et al. Soluble surface proteins from Helicobacter pylori activate monocytes/macrophages by lipopolysaccharide-independent mechanism. J Clin Invest 1991; 87: 894–900
Tarkkanen J, Kosunen TU, Saksela E, et al. Contact of lymphocytes with Helicobacter pylori augments natural killer cell activity and induces production of gamma interferon. Infect Immun 1993; 61: 3012–6
Rathbone BJ, Wyatt JI, Worsley BW, et al. Systemic and local antibody responses to gastric Campylobacter pylori in non-ulcer dyspepsia. Gut 1986; 27: 642–7
Kosunen TU, Seppala K, Sarna S, et al. Diagnostic value of decreasing IgG, IgA, and IgM antibody titres after eradication of Helicobacter pylori. Lancet 1992; 339: 893–5
Strober W, Richman LK, Elson CO. The regulation of gastrointestinal immune responses. Immunol Today 1981; 2: 156–62
Elson CO, Kagnoff MF, Fiocchi C, et al. Intestinal immunity and inflammation: recent progress. Gastroenterology 1986; 91: 746–68
Engstrand L, Scheynius A, Pahlson C, et al. Association of Campylobacter pylori with induced expression of class II transplantation antigens on gastric epithelial cells. Infect Immun 1989; 57: 827–32
Scheyrdus A, Engstrand L. Gastric epithelial cells in Helicobacter pylori-associated gastritis express HLA-DR but not ICAM-1. Scand J Immunol 1991; 33: 237–41
Wee A, Teh M, Kang JY. Association of Helicobacter pylori with HLA-DR antigen expression in gastritis. J Clin Pathol 1992; 45: 30–3
Vaines K, Huttfeldt HS, Brandtzaeg P. Relation between T cell number and epithelial HLA class II expression quantified by image analysis in normal and inflammed human gastric raucosa. Gut 1990; 31: 647–52
Vaines K, Brandtzaeg P, Elgio K, et al. Specific and non specific humoral defense factors in the epithelium of normal and inflamed gastric mucosa. Gastroenterology 1984; 86: 402–12
Crabtree JE, Taylor JD, Wyatt JI, et al. Mucosal IgA recognition of Helicobacter pylori 120 KD protein, peptic ulceration, and gastric pathology. Lancet 1991; 338: 332–5
Falk P, Roth KA, Boren T, et al. An in vitro adherence assay reveals that Helicobacter pylori exhibits cell lineage-specific tropism in the human gastric epithelium. Proc Natl Acad Sci USA 1993; 90: 2035–9
Thomas JE, Austin S, Dale A, et al. Protection by human milk IgA against Helicobacter pylori infection in infancy. Lancet 1993; 342: 121
Negrini R, Lisato L, Zanella I, et al. Helicobacter pylori infection induces antibodies cross-reacting with human gastric mucosa. Gastroenterology 1991; 101: 437–45
Engstrand L, Scheymus A, Pahlson C. An increased number of gamma/delta T-cells and gastric epithelial cell expression of the groEL stress protein homologue in H. pylori associated chronic gastritis of the antrum. Am J Gastroenterol 1991; 86: 976–80
Dunn BE, Roop RM, Sung CC, et al. Identification and purification of a cpn60 heat shock protein homolog from H. pylori. Infect Immun 1992; 60: 1946–51
Evans DJ, Evans DG, Engstrand LM, et al. Urease-associated heat shock protein of H. pylori. Infect Immun 1992; 60: 2125–7
Trejdosiewicz LK, Calabrese A, Smart CJ, et al. Gamma/delta T cell receptor-positive cells of the human gastrointestinal mucosa: occurrence and V region gene expression in Helicobacter pylori-associated gastritis, coeliac disease and inflammatory bowel disease. Clin Exp Immunol 1991; 84: 440–4
Freston JW. Overview of medical therapy of peptic ulcer disease. Gastroenterol Clin North Am 1990; 19: 121–39
Sloane R, Cohen H. Common-sense management of Helicobacter pylori-associated gastroduodenal disease. Gastroenterol Clin North Am 1993; 22: 199–206
Barr GD, Kang J, Canlese J, et al. A two year prospective controlled study of maintenance cimetidine and gastric ulcer. Gastroenterology 1983; 85: 100–4
Penston JG, Wormsley KG. Review article: maintenance treatment with H2-receptor antagonists for peptic ulcer disease. Aliment Pharmacol Ther 1992; 6: 3–29
Bardhan KD, Bainchi PG, Bose K, et al. A comparison of two different doses of omeprazole versus ranitidine in treatment of duodenal ulcers. J Clin Gastroenterol 1986; 8: 408–12
Glupczynksi Y, Delmee M, Bruck C, et al. Susceptibility of clinical isolates of Campylobacter pylori to 24 microbial and anti-ulcer agents. Eur J Epidemiol 1988; 4: 154–7
Lambert T, Megraud F, Gerbaud G, et al. Susceptibility of Campylobacter pylori to 20 microbial agents. Antimicrob Agents Chemother 1986; 30: 510–1
McNulty CAM. Bacteriological and pharmacological basis for the treatment of Campylobacter pylori infection. Gastroenterol Clin Biol 1989; 13 Suppl. 1: 96B–100B
Marshall BJ, Goodwin CS, Warren JR, et al. Prospective double-blind trial of duodenal ulcer relapse after eradication of Campylobacter pylori. Lancet 1988; 2: 1437–42
Marshall BJ, Armstrong JA, Francis GJ, et al. Antibacterial action of bismuth in relation to Campylobacter pylori colonization and gastritis. Digestion 1987; 37: 16–30
Weil J, Bell GD, Powell K, et al. Helicobacter pylori infection treated with a tripotassium dicitrato bismuthate and metronidazole combination. Aliment Pharmacol Ther 1990; 4: 651–7
Xia H, Daw MA, Sant S, et al. Clinical efficacy of triple therapy in Helicobacter pylori-associated duodenal ulcer. Eur J Gastroenterol Hepatol 1993; 5: 141–4
Bell GD, Powell K, Burridge SM, et al. Experience with ‘triple’ anti-Helicobacter pylori eradication therapy: side effects and the importance of testing the pre-treatment bacterial isolate for metronidazole resistance. Aliment Pharmacol Ther 1992; 6: 427–35
Burette A, Glupczynski Y, De Prez C. Evaluation of various multi drug eradication regimens for Helicobacter pylori. Eur J Gastroenterol Hepatol 1992; 4: 817–23
Hentschel E, Brandstatter G, Dragosics B, et al. Effect of ranitidine and amoxicillin plus metronidazole on the eradication of Helicobacter pylori and the recurrence of duodenal ulcer. N Engl J Med 1993; 328: 308–12
Goodwin CS, Marshall BJ, Blincow ED, et al. Prevention of nitroimidazole resistance in Campylobacter pylori by coadministration of colloidal bismuth subcitrate: clinical and in vitro studies. J Clin Pathol 1988; 41: 207–10
Banatvala N, Davies GR, Abdi Y, et al. 95% metronidazole resistance in H. pylori infection in a UK Asian community [abstract]. Gastroenterology 1993; 104: A37
Malfertheiner P. Compliance, adverse events and antibiotic resistance in Helicobacter pylori treatment. Scand J Gastroenterol 1993; 28 Suppl. 196: 34–7
Graham DY, Lew GM, Malaty HM, et al. Factors influencing the eradication of Helicobacter pylori with triple therapy. Gastroenterology 1992; 102: 493–6
Labenz J, Gyenes E, Ruhl GH, et al. Orale Tripeltherapie zur Helicobacter-pylori-Eradkation bei duodenaler Ulkuskrankheit. Med Klin 1993; 88: 297–9
Logan RPH, Gummett PA, Misiewicz JJ, et al. Two-week eradication regimen for metronidazole-resistant Helicobacter pylori. Aliment Pharmacol Ther 1993; 7: 149–53
Labenz L, Gyenes E, Ruhl GH, et al. Amoxicillin plus omeprazole versus triple therapy for eradication of Helicobacter pylori in duodenal ulcer disease: a prospective, randomized, and controlled study. Gut 1993; 34: 1167–70
Collins R, Beattle S, Xia HA, et al. Short report: high-dose omeprazole and amoxycillin in the treatment of Helicobacter pylori-associated duodenal ulcer. Aliment Pharmacol Ther 1993; 7: 313–5
Unge P, Ekstrom P. Effects of combination therapy with omeprazole and an antibiotic on Helicobacter pylori and duodenal ulcer disease. Scand J Gastroenterol 1993; 28 Suppl 196: 17–8
Bayerdorffer E, Mannes GA, Sommer A, et al. Long-term follow-up after eradication of Helicobacter pylori with a combination of omeprazole and amoxycillin. Scand J Gastroenterol 1993; 28 Suppl. 196: 19–25
Bayerdorffer E, Miehlke S, Mannes GA, et al. Double blind trial of 120mg omeprazole and amoxycillin for eradication of Helicobacter pylori in duodenal ulcer patients. J Clin Gastroenterol Hepatol 1994; 9: A11
Graham DY, Lew GM, Ramirez FC, et al. Short report: a non-metronidazole triple therapy for eradication of Helicobacter pylori infection — tetracycline, amoxicillin, bismuth. Aliment Pharmacol Ther 1993; 7: 111–4
McCarthy CJ, Collins R, Beattle S, et al. Short report: treatment of Helicobacter pylori-associated duodenal ulcer with omeprazole plus antibiotics. Aliment Pharmacol Ther 1993; 7: 463–66
Grayson ML, Eliopoulos GM, Ferraro MJ, et al. Effect of varying pH on the susceptibility of Campylobacter pylori to antimicrobial agents. Eur J Clin Microbiol Infect 1989; 8: 888–9
Sharma BK, Santana IA, Wood EC, et al. Intragastric bacterial activity and nitrosation before, during, and after treatment with omeprazole. BMJ 1984; 289: 717–9
Bhatia SJ, Kochar N, Abraham P, et al. Lactobacillus acidophilus inhibits growth of Campylobacter pylori in vitro. J Clin Microbiol 1989; 27: 2328–30
O’Connor HJ, Axon AIB, Dixon MI. Campylobacter like organisms unusual in type A (pernicious anaemia) gastritis. Lancet 1984; 2: 1091
Rauws EAJ, Langenberg W, Houthoff HJ, et al. Campylobacter pylori-associated chronic active antral gastritis: a prospective study on its prevalence and the effects of antibacterial and anti-ulcer treatment. Gastroenterology 1988; 94: 33–40
Valle J, Seppala K, Sipponen P, et al. Disappearance of gastritis after eradication of Helicobacter pylori. Scand J Gastroenterol 1991; 26: 1057–65
Di Napoli A, Petrino R, Boero M, et al. Quantitative assessment of histological changes in chronic gastritis after eradication of Helicobacter pylori. J Clin Pathol 1992; 45: 796–98
Bayerdffrffer E, Mannes GA, Sommer A, et al. High dose omeprazole treatment combined with amoxicillin eradicates Helicobacter pylori. Eur J Gastroenterol Hepatol 1992; 4: 697–702
Bayerdorffer E, Miehlke S, Lehn N, et al. Chronic type B-gastritis as an important denominator of peptic ulcer healing. Eur J Gastroenterol Hepatol 1993; 5 Suppl. 3: 99–105
Genta RM, Hamner HW, Graham DY. Gastric lymphoid follicles in Helicobacter pylori infection: frequency, distribution, and response to triple therapy. Hum Pathol 1993; 24: 577–83
Borody TJ, Andrews P, Jankiewicz E, et al. Apparent reversal of early gastric mucosal atrophy after triple therapy for Helicobacter pylori. Am J Gastroenterol 1993; 88: 1266–8
Correa P, Fox J, Fontham E, et al. Helicobacter pylori and gastric carcinoma: serum antibody prevalence in populations with contrasting cancer risks. Cancer 1990; 66: 2569–74
Kneller RW, Guo WD, Hwing AW, et al. Risk factors for stomach cancer in sixty-five Chinese counties. Cancer Epidemiol Biomarkers Prev 1992; 1: 113–5
De Magistris MT, Di Tommaso A, Bugnoli M, et al. T-lymphocyte responses to Helicobacter pylori in peripheral blood and gastric mucosa of patients with gastritis. Eur J Gastroenterol Hepatol 1993; 5 Suppl. 2: S25–6
Pallen MJ, Clayton CL. Vaccination against Helicobacter pylori urease. Lancet 1990; 336: 186–7
Leying H, Suerbaum S, Geis G, et al. Cloning and genetic characterization of a Helicobacter pylori flagellin gene. Mol Microbiol 1992; 6: 2863–74
Doig P, Austin JW, Kostrzynska M, et al. Production of a conserved adhesin by the human gastroduodenal pathogen Helicobacter pylori. J Bacteriol 1992; 174: 2539–47
Evans DG, Karjalainen TK, Evans DJ, et al. Cloning, nucleotide sequence and expression of a gene encoding an adhesion sub-unit protein of Helicobacter pylori. J Bacteriol 1993; 175: 674–83
Lingwood CL, Wasfy G, Han H, et al. Receptor affinity purification of a lipid-binding adhesin from Helicobacter pylori. Infect Immun 1993; 61: 2474–8
Xiang Z, Bugnoli M, Rappuoli R, et al. Helicobacter pylori: host response in peptic ulceration. Lancet 1993; 341: 900–1
Fox JG, Correa P, Taylor NS, et al. Helicobacter mustelae-associated gastritis in ferrets. Gastroenterology 1990; 99: 352–61
Czinn SJ, Nedrud JG. Oral immunization against Helicobacter pylori. Infect Immun 1991; 59: 2359–63
Chen M, Lee A, Hazell S. Immunisation against gastric helicobacter infection in a mouse/Helicobacter felis model. Lancet 1992; 339: 1120–1
Eaton KA, Krakowka S. Chronic active gastritis due to Helicobacter pylori in immunized gnotobiotic piglets. Gastroenterology 1992; 103: 1580–6
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hatz, R., Bayerdörffer, E., Lehn, N. et al. Immune Response in Helicobacter pylori Infection. Clin. Immunother. 2, 295–306 (1994). https://doi.org/10.1007/BF03258529
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03258529