Summary
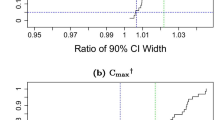
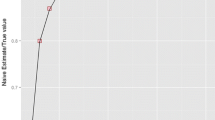
Bioequivalence of two medicinal, or veterinary, products is established by comparing the mean of bioavailability measures, such as AUC and Cmax, following administration of the test (T) and reference (R) products. However, the use of these parameters has several drawbacks, e.g. they do not take into consideration the overall pharmacokinetic profile shape. Therefore, concerns have been raised regarding their appropriateness for assessment of bioequivalence. To overcome the limitations of these bioequivalence parameters, direct curve comparison metrics methods were recently proposed on an average basis. In this paper, an individual based direct curve comparison method for assessing bioequivalence is proposed. The bioequivalence of T and R in each subject is evaluated by a new curve comparison metrics δ. The metrics δ is the absolute sum of the difference between two curves. The significance of the metrics for each subject is assessed by bootstrapping. An overall bioequivalence of T and R may be considered if less than 25% of the subjects show statistically different profiles.
Similar content being viewed by others
References
CPMP/EWP/QWP/1401/98 (2001) Note for Guidance on the Investigation of Bioavailability and Bioequivalence.
CVMP/III/8090/89 (1992) Conduct of Bioequivalence Studies in Animals.
Shuirmann D.J. (1987): A comparison of the two one-sided tests procedure and the power approach for assessing the bioequivalence of average bioavailability. J. Pharmacokinet. Biopharm., 15, 657–680.
Zintzaras E. and Bouka P. (1999): Bioequivalence studies: blometrical concepts of alternative designs and pooled analysis. Eur. J. of Drug. Metab. and Pharmacokinet., 24, 225–232.
Chen M.L., Lesko I., Williams R. (2001): Measures of Exposure versus Measures of Rate and Extent of Absorption. Clin. Pharmacokinet, 40, 565–572.
Rescigno A. (1992): Bioequivalence Pharm. Res., 9, 925–928.
Marston S.A. and Polli J.E. (1997): Evaluation of direct curve comparison metrics applied to pharmacokinetic profiles and relative bioavailability and bioequivalence. Pharm. Res. 14, 1363–1369.
Polli JE., McLean AM. (2001): Novel Direct Curve Comparison Metrics for Bioequivalence. Pharm Res., 18, 734–741.
Tozer T.N., Bois F.Y., Hauck W.W., Chen M.L., Williams R.L. (1996) Absorption rate vs exposure: Which is more useful for bioequivalence testing? Pharm. Res., 13, 453–456.
Langenbucher F. (1999): IVIVC: indices for comparing release and response profiles. Drug. Dev Ind Pharm., 25, 1223–5.
Shah VP., Tsong Y., Sathe P, Liu JP. (1998): In vitro dissolution profile comparison-statistics and analysis of the similarity factor, 12. Pharm. Res. 15, 889–896.
Chow S.C., and Liu J.P. (1992): Design and analysis of bioavailability and bioequivalence studies. New York: Marcel Dekker.
Anderson S. and Hauck W.W. (1990): Consideration of individual bioequivalence. J. Pharmacokinet. Biopharm., 18, 259–273.
Hauck W.W. and Anderson S. (1992): Types of Bioequivalence and related statistical considerations. Int. J. Clin. Pharmacol. Ther. Toxicol., 30, 181–187.
Food and Drug Administration Center for Drug Evaluation and Research (2001) Guidance for Industry: Statistical Approaches to Establishing Bioequivalence.
Chen ML., Lesko LJ. (2001): Individual Bioequivalence revisited Clin. Pharmacokinet., 40, 701–706.
Chen ML, Patnaik R, Hauck WW, Shuirmann DJ, Hyslop T, William R. (2000) An Individual Bioequivalence Criterion: regulatory considerations. Stat. Med., 19, 2821–2842.
Hauck W.W., Hyslop T, Chen ML, Patnaik R, William R. (2000): Subject-by-Formulation Interaction in Bioequivalence: Conseptual and Statistical Issues. Pharm. Research., 17, 375–380.
Efron B. and Tibshirani R. (1986) An Introduction to the Bootstrap. London: Chapman and Hall.
Zintzaras E (2000) The existence of sequence effect in cross-over Bioequivalence trials. Europ. J. Drug Metab. Pharmacokinet., 25, 241–244.
Jones B. and Kenward M.G. (1995) Design and Analysis of Cross-Over Trials. London: Chapman and Hall.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zintzaras, E., Bouka, P. & Kowald, A. Biometrical evaluation of bioequivalence trials using a bootstrap individual direct curve comparison method. Eur. J. Drug Metab. Pharmacokinet. 27, 11–16 (2002). https://doi.org/10.1007/BF03190400
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03190400