Abstract
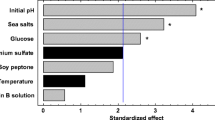
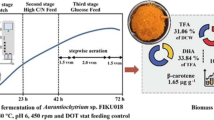
Environmental and medium factors were investigated as basic data for optimizing DHA production when usingThraustochytrium aureum. To study the effect of environmental conditions, the rotation speed and culture temperature were, changed. Plus the trend of the growth characteristics, lipid content in the biomass, and DHA content in lipids were evaluated according to various initial glucose concentrations. The biomass, lipid, and DHA analyses showed that the physiological characteristics ofT. aureum were closely related with the environmental and medium conditions, as in the case of other marine microorganisms. For example, a low rotation speed of 50 rpm lowered the cell growth rate as well as the DHA content in the lipids. A low temperature had a negative effect on the cell growth, yet a positive effect on the lipid content in the biomass. Different initial glucose concentrations had no effect on the lipid content in the biomass or DHA content in the lipids, yet did affect the cell growth. Accordingly, these results show that environmental and medium factors must be synthetically considered in order to optimize DHA production when usingT. aureum.
Similar content being viewed by others
References
Singh, A. and O. P. Ward (1996) Production of high yields of docosahexaenoic acid byThraustochytrium roseum ATCC 28210.J. Ind. Microbiol. 12: 370–373.
Ward, O. P. (1995) Microbial production of long-chain PUFAs.INFORM 6: 683–688.
Bazan, N. G., T. S. Reddy, H. E. P. Bazan, and D. L. Brikle (1986) Metabolism of arachidonic and docosahexaenoic acid in the retina.Prog. Lipid Res. 25: 595–606.
Uauy, R. D., D. G. Brich, E. Z. Brich, J. E. Tyson, and D. R. Hoffman (1990) Effect of dietary omega-3 fatty acids on retinal function of very-low-brith-weight neonates.Pediar. Res. 28: 485–492.
Connor, W. E., N. Martha, and D. S. Lin (1990) Dietary effects on brain fatty acid composition, the reversibility of n-3 fatty acid deficiency and turnover of doecosahexaenoic acid in brain, erythrocyte and plasma of rhesus monkey.J. Lipid Res. 31: 237–247.
Bajpai, P. K., P. Bajpai, and O. P. Ward (1991) Optimization of production of docosahexaenoic acid (DHA) byThraustochytrium aureum ATCC 34304.JAOCS 68: 509–514.
Jseph, J. D. (1975) Identification of 3, 6, 9, 12, 15-octadecapentaenoic acid in laboratory-cultured photosynthetic dinoflagellates.Lipids 10: 395–403.
Patton, S., G. Fuller, A. R. Loehlich, and A. A. Benson (1966) Fatty acids of the ‘red tide’ organismGonyaulax polyedra.Biochem. Biophys. Acta 116: 577–579.
Withers, N. W. and J. C. Nevenzel (1977) Phytyl esters in a marine diflagellate.Lipids 12: 989–993.
Tyrrell, D. (1967) The fatty acid composition of in Entomophthora isolates.Can. J. Microbiol. 13: 755–760.
Shaw, R. (1965) The occurrence of gamma-linolenic acid in fungi.Biochem. Biophys. Acta 98: 230–237.
De Long, E. F. and A. A. Yayanos (1986) Biochemical function and ecological significance of novel bacterial lipid in deep-sea prokaryotes.Appl. Environ. Microbiol. 51: 730–737.
Yano, Y., A. Nakayama H. Saito, and K. Ishihara (1994) Production of docosahexaenoic acid by marine bacteria isolated from deep sea fish.Lipids 29: 527–528.
Yokochi, T., D. Honda, T. Higashihara, and T. Nakahara (1998) Optimization of docosahexaenoic acid production bySchizochytrium limacium SR21.Appl Microbiol. Biotechnol. 49: 72–76.
Wichien, Y. M. and P. W. Owen (1989) Omega-3 fatty acids: Alternative sources of production.Process Biochem. 24: 117–125.
Sajbidor, J., S. Dobronova, and M. Certik (1990) Atachidonic acid production byMortiesella sp. S-17. Influence of C/N ratio.Biotechnol. Lett. 12: 455–456.
Peberdy, J. F. and D. K. Toomer (1975) Effect of nutrient starvation on the utilization of storage lipids inMortiesella ramaruiana.Microbios. 13: 123–131.
Radwan, S. S. and A. H. Soliman (1988) Arachidonic acid from fungi utilizing fatty acids with short chains as sole sources of carbon and energy.J. Gen. Microbiol. 134: 387–393.
Yue, J. and F. Chen (2000) Effects of medium glucose concentration and pH on docosahexaenoic acid content of heterotrophicCrypthecodinium cohnii.Process Biochem. 35: 1205–1209.
Neidelman, S. L. (1987) Effect of temperature on lipid unsaturation.Biotechnol. Eng. Rev. 5: 245–268.
Stinson, I. E., R. Kwoczak, and M. J. Kuruntz (1991) Effect of cultural conditions on production of eicosapentaenoic acid byPythium irregulare.J. Ind. Microbiol. 8: 171–178.
Cohen, Z., A. Vonshek, and A. Richmond (1987) Fatty acid composition of Spirulina strains grown under various environmental conditions.Phytochem. 26: 2255–2258.
Lepage, C. and C. C. Roy (1984) Improved recovers of fatty acid a through direct of transesterification without prior extraction and purification.J. Lipid Res. 25: 1391–1396.
Iida, I., T. Nakahara, T. Yokochi, Y. Kamisaka, H. Yagi, M. Yamaoka, and O. Suzuki (1996) Improvement of docosahexaenoic acid production in a culture ofThraustochytrium aureum by medium optimization.J. Ferment. Bioeng. 81: 76–78.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hur, BK., Cho, DW., Kim, HJ. et al. Effect of culture conditions on growth and production of docosahexaenoic acid (DHA) usingThraustochytrium aureum ATCC 34304. Biotechnol. Bioprocess Eng. 7, 10–15 (2002). https://doi.org/10.1007/BF02935873
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02935873