Abstract
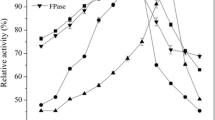
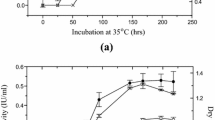
The ability ofT. harzianum to produce the three components of cellulase,viz. filter paper activity, carboxymethylcellulase and β-glucosidase was investigated. The optimum pH and temperature for all the enzymes in the culture nitrate was 5–7 and 27°C, respectively. The shaken culture conditions gave low yields of the enzyme as compared to static cultures. Most of the FP-activity and CM-cellulase were located extracellularly but reasonable amount of β-glucosidase was retained in the cell debris fraction.
Similar content being viewed by others
References
Andreotti R.E., Medeiros J.E., Roche C., Mandels M.: Effect of strain and substrate on production of cellulases byTrichoderma reesei mutants, pp. 353–372 inProceedings of the 2nd International Symposium on Bioconversion and Biochemical Engineering (T.K. Ghose, Ed.), Vol. 1, BERC, IIT, Delhi 1980.
Berg B., Petterson G.: Location and formation of cellulases inTrichoderma viride.J.Appl. Bact.42, 65 (1977).
Bisaria V.S., Ghosh T.K.: Biodegradation of cellulosic materials: substrates, microorganisms, enzymes and products.Enzyme Microb.Technol.3, 90 (1981).
Gallo B.J., Andrbotti R.E., Roche C., Ryu D., Mandels M.: Cellulase production by a new mutant strain ofTrichoderma reesei MCG 77.Biotechnol. Bioeng. Symp.8, 89 (1978).
Halliwell G., Lovelady J.: Utilization of carboxymethyl cellulose and enzyme synthesis byTrichoderma koningii.J. Gen. Microbiol.126, 211 (1981).
Kubicek C.P.: Release of carboxymethyl-cellulase and β-glucosidase from cell walls ofTrichoderma reesei.Eur. J. Appl. Microbiol. Biotechnol.13, 226 (1981).
Kubicek C.P.: β-Glucosidase excretion inTrichoderma strains with different cell wall bound β-1,3-glucanase activities.Can. J. Microbiol.29, 163 (1983).
Mandels M., Andreotti R.E.: Problems and challenges in the cellulose to cellulase fermentation.Proc. Biochem.13, 6 (1978).
Mishra S., Gopalkrishnan K.S., Ghosb T.K.: A constitutive cellulase producing mutant ofTrichoderma reesei.Biotechnol. Bioeng.24, 251 (1982).
Mukhopadhyay S.N., Malik A.K.: Increased production of cellulase ofTrichoderma spp. by pH cyclic and temperature profiling.Biotechnol. Bioeng.22, 2237 (1980).
Peitersen N.: Continuous cultivation ofTrichoderma viride on cellulose.Biotechnol. Bioeng.19, 337 (1977).
Reese E.T.: Polysaccharases and hydrolysis of insoluble substrates, pp. 165–181 inBiological Transformation of Wood (W. Liese, Ed.). Springer-Verlag, Berlin 1975.
Reese E.T., Mandels M.: Stability of cellulase ofT. reesei under use conditions.Biotechnol. Bioeng.22, 323 (1980).
Sandhu D.K., Kalra M.K.: Production of cellulase, xylanase and pectinase byTrichoderma longibrachiatum on different substrates.Trans. Brit. Mycol. Soc.79, 409 (1982).
Sandhu D.K., Sidhu M.S., Kalra M.K., Sandhu R.S.: Regulation of cellulase activity in three species ofTrichoderma. Abstract P-2 14704,7th Internal. Biotechnol. Symp. Indian Institute of Technology, New Delhi 1984.
Sternber G.D., Mandels M.: Regulation of the cellulolytic system inTrichoderma reesei by sophorose. Induction of cellulase and repression of β-glucosidase.J.Bact.144, 1197 (1980).
Updegrapp D.M.: Semimicro determination of cellulose in biological materials.Anal. Biochem.32, 420 (1969).
Vaheri M.P., Vahert M.E.O., Kaupinen V.S.: Formation and release of cellulolytic enzymes during growth ofTrichoderma reesei on cellobiose and glycerol.Eur. J. Appl. Microbiol. Biotechnol.8, 73 (1979).
Wood T.M., McCrae S.I.: The cellulase ofTrichoderma koningii: Purification and properties of some endoglucanase components with special reference to their action on cellulose when acting alone and in synergism with the cellobiohydrolase.Biochem. J.171, 61 (1978).
Author information
Authors and Affiliations
Additional information
The authors acknowledge the receipt of fellowship from theCouncil of Scientific and Industrial Research, New Delhi.
Rights and permissions
About this article
Cite this article
KalRa, M.K., Sandhu, D.K. Cellulase production and its localization inTrichoderma harzianum . Folia Microbiol 31, 303–308 (1986). https://doi.org/10.1007/BF02926955
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02926955