Abstract
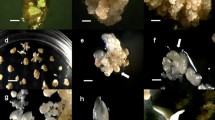
Embryogenic callus which has maintained its embryogenic ability on media without growth regulators for three years has been induced at the base of shoots of a genotype with CMS propagated for a long timein vitro by transferring the shoots onto media richer in inorganic and organic components. The effect of two basal media (MS and PG0) on the intensity and completeness of the proliferation of somatic embryos was examined with different combinations of growth regulators. Pollen fertility was evaluated in 87 plants regenerated from somatic embryos. Cytoplasmic male sterility was conserved in all of them.
Similar content being viewed by others
References
Ammirato, P. V.: Embryogenesis. - In:Evans, D. A., Sharp, W. R., Ammirato, P. V., Yamada, Y. (ed.): Handbook of Plant Cell Culture. Vol. 1. Pp. 82–123. Macmillan, New York 1983.
Atanasov, A. I.: Analysis of organogenetic abilities of continuously cultivated somatic tissues of sugar beet on the basis of obtaining single-celled clones and suspension cultures. - In:Novák, F. J. (ed.): Use of Tissue Cultures in Plant Breeding. Pp. 69–80. Institute of Experimental Botany, Olomouc 1976.
Brears, T., Curtis, G. J., Lonspale, D. M.: A specific rearrangement of mitochondrial DNA induced by tissue culture. - Theor. appl. Genet.77: 620–624, 1989.
de Greef, W., Jacobs, M.:In vitro culture of the sugarbeet: description of a cell line with high regeneration capacity. - Plant Sci. Lett.17: 55–61, 1979.
Earle, E. D., Gracen, V. E., Best, V. M., Batts, L. A., Smith, M. E.: Fertile revertants from S-type male-sterile maizegrown in vitro. -Theor. appl. Genet. 74: 601–609, 1987.
Freytag, A. H., Anand, S. C., Raoarelli, A. P., Owens, L. D.: An improved medium for adventitious shoot formation and callus induction inBeta vulgaris L.in vitro. - Plant Cell Rep.7: 30, 1988.
Kubaláková, M.: [Problems concerningin vitro multiplication of sugar beet (Beta vulgaris L. var.saccharifera)]. In Czech. - Rost. Výroba7: 725–732, 1989.
Kubaláková, M.,Havel, L.: The cytoplasmic male sterility in sugar beet afterin vitro multiplication. 6th Congress of the Federation of European Societies of Plant Physiology (FESPP), Abstract. Split 1988.
Linsmaier, E. M., Skoog, F.: Organic growth factor requirements of tobacco tissue cultures. - Physiol. Plant.18: 100–127, 1965.
Murashige, T., Skoog, F.: A revised medium for rapid growth and bioassay with tobacco tissue cultures. -Physiol. Plant.15: 473–497, 1962.
Nêmec, B.: Botanická Mikrotechmka. [Botanical Microtechnique.] - Nakl. ĈSAV, Praha 1962.
Pedersen, M. G., Andersen, J. M.: Callus Morphology and Endogenous Cytokinins in Regenerating Sugar Beet Callus Cultures. - Eucarpia Meeting Beta, Helsingør 1988.
Powling, A., Ellis, T. H. N.: Studies on the organelle genomes of sugarbeet with male-fertile and male-sterile cytoplasms. - Theor. appl. Genet.65: 323–328, 1983.
Saunders, J. W., Daub, M. E.: Shoot regeneration from hormone autonomous callus from shoot cultures of several sugarbeet (Bete vulgaris L.) genotypes. - Plant Sci. Lett.34: 219–223, 1984.
Saunders, J. W., Doley, W. P.: One step shoot regeneration from callus of whole plant leaf explants of sugarbeet lines and a somaclonal variant forin vitro behaviour. - J. Plant Physiol.124: 479, 1986.
Steen, P., Keimer, B., D’Halluin, K., Pedersen, H. C.: Variability in plants of sugar beet (Beta vulgaris L.) regenerated from callus, cell suspension and protoplasts. - In:Horn, W., Jensen, C. J., Odenbach, W., Schieder, O. (ed.): Genetic Manipulation in Plant Breeding. Pp. 633–635. Walter de Gruyter and Co., Berlin - New York 1986.
Tétu, T., Sangwan, R. S., Sangwan-Norreel, B. S.: Hormonal control of organogenesis and somatic embryogenesis inBete vulgaris callus. - J. exp. Bot.38: 506–517, 1987.
Umbeck, P. F., Gengenbach, B. G.: Reversion of male-sterile T-cytoplasm maize to male fertile in tissue culture. -Crop Sci.23: 584–588, 1983.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KubalÁkovÁ, M. Somatic embryogenesis and cytoplasmic sterility inBeta vulgaris L. var.saccharifera . Biol Plant 32, 414–419 (1990). https://doi.org/10.1007/BF02890886
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02890886