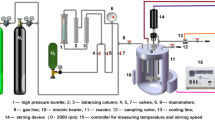
Abstract
Palladium supported on alumina was used to hydrogenate soybean and canola oil. Previous literature reports indicated that palladium forms moretrans isomers than nickel. At 750 psig, 50 ppm palladium, and at 70 C, only 9.4%trans were formed when canola oil was hydrogenated to IV 74. In general, high pressure and low temperature favored lowtrans formation with no appreciable loss in catalyst activity. The effect of pressure, temperature and catalyst concentration on reaction rate,trans formation and selectivity is presented. A survey of various catalyst supports is discussed. Apparent activation energies of 6.3 to 8.9 kcal/mol were obtained; they are in good agreement with values reported in the literature.
Similar content being viewed by others
References
Hsu, N., L.L. Diosady, W.F. Graydon and L.J. Rubin,J. Am. Oil Chem. Soc. 63:1036 (1986).
Ahmad, M.M., T.M. Priestley and J.M. Winterbottom,Ibid. 56:571 (1979).
Riesz, C.H., and H.S. Weber,Ibid. 41:400 (1964).
Coenen, J.W.E., and H. Boerma,Fette, Seifen, Anstrichm. 70:8 (1968).
Horiuti, J., and M. Polanyi,Trans. Farad. Soc. 30:1164 (1934).
Albright, L.F.,J. Am. Oil Chem. Soc. 40:16 (1963).
Susu, A., A.F. Ogunye and C.O. Onyegbado,J. Appl. Chem. Biotechnol. 28:823 (1978).
Zajcew, M.,J. Am. Oil Chem. Soc. 39:301 (1962).
Zajcew, M.,Ibid. 37:130 (1960).
Author information
Authors and Affiliations
About this article
Cite this article
Hsu, N., Dlosady, L.L. & Rubin, L.J. Catalytic behavior of palladium in the hydrogenation of edible oils. J Am Oil Chem Soc 65, 349–356 (1988). https://doi.org/10.1007/BF02663075
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02663075