Abstract
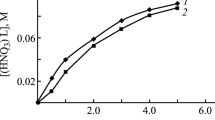
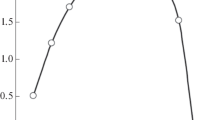
The extraction of actinium with HDEHP from Cl− and NO −3 systems has been investigated. It was found that extraction of actinium from HCl solutions is much better than from HNO3 solutions. Stability constants of the actinium complexes Ac(X−)2+, X−=Cl− or NO −3 , were determined. Our results show that actinium formed less stable complexes with Cl− than with NO −3 ligands.
Similar content being viewed by others
References
D.F. Peppard, G.W. Mason, W.J. Driscoll, R.J. Sironen,J. Inorg. Nucl. Chem., 7 (1958) 276.
Z.K. Karalova, L.M. Radonova, Z.I. Pyzhova, B.F. Myasoedov,Radiokhimiya, 1 (1978) 42.
I.M. White, P. Kelly, N.C. Li,J. Inorg. Nucl. Chem., 16 (1961) 337.
G.R. Choppin, P.J. Unrein,J. Inorg. Nucl. Chem., 25 (1963) 387.
D.F. Peppard, G.W. Mason, I. Hucher,I. Inorg. Nucl. Chem., 24 (1962) 881.
G.R. Choppin, W.F. Strazik,Inorg. Chem., 4, (1965) 1250.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Szeglowski, Z., Kubica, B. Extraction of actinium with di (2-ethylhexyl) phosphoric acid from hydrochloric and nitric acid solutions. Journal of Radioanalytical and Nuclear Chemistry Letters 153, 67–74 (1991). https://doi.org/10.1007/BF02170250
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02170250