Abstract
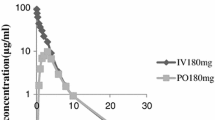
The pharmacokinetics of cefroxadin have been studied after the administration of single oral and intravenous doses to healthy volunteers. Cefroxadin was assayed by HPLC. The kinetics in plasma following i.v. administration were described by using a three-compartment model. An additional disposition phase was observed following oral administration that could not be detected after the low i.v. dose. The terminal half-life was 1.03 h. The apparent volume of distribution at the steady state was consistent with a diffusion of the antibiotic in all extracellular fluids. The AUCafter oral administration was linearly related to the dose. The urinary excretion amounted to 95% of the dose with virtually complete absorption of orally administered drug.
Similar content being viewed by others
References
R. Scartazzini and H. Bickel. New orally active cephalosporins.Heterocycles 7:1165–1188 (1977).
O. Zak, W. A. Vischer, C. Schenk, W. Tosch, W. Zimmermann, J. Regös, E. R. Suter, P. Kradolfer, and J. Geizer. CGP 9000, new orally active, broad spectrum cephalosporin.J. Antibiot. 29:653–655 (1976).
O. Zak, W. A. Vischer, W. Tosch, and F. Kradolfer. Comparative experimental studies on 3-methoxy and 3-methyl-cephems.Drugs Exp. Clin. Res. 1:11–20 (1977).
W. A. Vischer, O. Zak, E. A. Konopka, H. Fehlmann, J. Regös, and W. Tosch. Experimental evaluation of CGP 9000, a new orally active cephalosporin.Curr. Chemother. 2:825–827 (1978).
D. Greenwood. In vitro activity against Escherichia coli of CGP 9000, a new oral cephalosporin.J. Antibiot. 31:697–702 (1978).
H. Wirz, W. A. Vischer, J. Füllhaas, and P. R. Imhof. Pharmacokinetics of CGP 9000, a new orally active cephalosporin, in healthy volunteers.Curr. Chemother. 2:827–829 (1978).
H. Lode, R. Stahlmann, G. Dzwillo, and P. Koeppe.Pharmacokinetische Vergleichsunter-suchungen zur Resorption von drei neuen Cephalosporinen. Verhandlungen der Deutschen Gesellschaft für innere Medizin. J. F. Bergmann Verlag, München, 1978, pp. 964–966.
H. Lode, R. Stahlmann, and P. Koeppe. Comparative pharmacokinetics of cephalexin, cefaclor, cefadroxil and CGP 9000.Antimicrob. Agents Chemother. 16(1): 1–6 (1978).
T. Bergan. Pharmacokinetics of a new cephalosporin, CGP 9000 (cefroxadine), in healthy volunteers.Chemotherapy,26:225–230 (1980).
R. Cadorniga, J. A. Gutierrez, M. C. Saiz Vadillo, I. T. Molina, C. M. Evoka, P. Pastoriza, J. Aluarez, and J. Campo. Etude pharmacocinétique du CGP 9000 chez l'homme par perfusion. 2nd Mediterranean Congress of Chemotherapy, Nice 1980, Abstract 209:181 (1980).
J. B. Lecaillon, J. L. Hirtz, J. P. Schoeller, G. Humbert, and W. Vischer. Pharmacokinetic comparison of cefroxadin (CGP 9000) and cephalexin by simultaneous administration to humans.Antimicrob. Agents Chemother. 18(4):656–660 (1980).
P. W. M. John.Statistical Design and Analysis of Experiments. Macmillan, New York, 1971, p. 115.
C. H. Nightingale, D. S. Greene, and R. Quintiliani. Pharmacokinetics and clinical use of cephalosporin antibiotics.J. Pharm. Sci. 64:1899–1927 (1975).
D. S. Greene, R. Quintiliani, and C. H. Nightingale. Physiological perfusion model for cephalosporin antibiotics I: Model selection based on blood drug concentrations.J. Pharm. Sci. 67:191–196 (1978).
A. P. Gillett and R. Wise. Penetration of four cephalosporins into tissue fluid in man.Lancet 6:962–964 (1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gerardin, A., Lecaillon, J.B., Schoeller, J.P. et al. Pharmacokinetics of cefroxadin (CGP 9000) in man. Journal of Pharmacokinetics and Biopharmaceutics 10, 15–26 (1982). https://doi.org/10.1007/BF01059181
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01059181