Summary
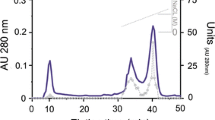
Periostracin is a soluble presclerotin of the periostracum ofMytilus edulis. Periostracin is unusually unstable in vitro. It was extracted with high yield from marginal periostracum with pure formic acid. It is a basic, water-insoluble protein with a molecular weight of 20,000 as determined by gel electrophoresis in sodium dodecyl sulfate. The amino acid composition of periostracin is unique with 55% of the residues being glycine, 10% tyrosine and 2.2% Dopa. The presence of Dopa was further substantiated by reaction with ethylenediamine, by difference spectra at various pH and by UV and visible spectra of the oxidation products. The origin of Dopa in periostracin seems linked to a tyrosine hydroxylase active toward protein tyrosyl residues.
Similar content being viewed by others
Abbreviations
- Dopa:
-
3,4-dihydroxyphenylalanine
- SDS:
-
sodium dodecyl sulfate
References
Abercrombie, M., Hickman, C.J., Johnson, M.L.: A dictionary of biology. Middlexes, Eng.: Penguin 1976
Andersen, S.O.: Sclerotization in insect cuticle. In: Perspectives in experimenal biology, Vol. 1, Zoology, Davies, P.S. (ed), pp. 187–190. Oxford-New York: Pergamon 1976
Arce, C.A., Rodriguez, J.A., Barra, H.S., Caputto, R.: Incorporation ofl-tyrosine,l-phenylalanine andl-3,4 dihydroxyphenylalamine as single repeating units into rat brain tubulin. Eur. J. Biochem.59, 145–149 (1975)
Beedham, G.E.: Observations on the non-calcareous component of the shell of Lamellibranchia. Quart. J. Microsc. Sci.99, 341–357 (1958)
Bornstein, P.: The biosynthesis of collagen. Ann Rev. Biochem.43, 567–603 (1974)
Brown, C.H.: Quinone tanning in the animal kingdom. Nature165, 275 (1950)
Brown, C.H.: Some structural proteins ofMytilus edulis. Quart. J. Microsc. Sci.93, 487–502 (1952)
Brunet, P.C.J.: The sclerotins. Endeavour26, 68–74 (1967)
Bubel, A.: An EM investigation into the distribution of polyphenols in the periostracum and cells of the inner face of the outer fold ofMytilus edulis. Mar. Biol.23, 2–15 (1973)
Davie, E.W., Fujikawa, K.: Basic mechanisms in blood coagulation. Ann. Rev. Biochem.44, 799–829 (1975)
Degens, E.T., Spencer, D.W., Parker, R.H.: Paleobiochemistry of molluscan shell proteins. Comp. Biochem. Physiol.20, 553–579 (1967)
Gaffin, S.L.: The clotting of the lysed white cells ofLimulus induced by endotoxin — I.: Preparation and characterization of clot forming proteins. Biorheol.13, 273–280 (1976)
Högenauer, G., Kreil, G., Bernheimer, H.: Studies on the binding of Dopa (3,4-dihydroxyphenylalanine) to tRNA. FEBS Letters88, 101–104 (1978)
King, J., Laemmli, U.K.: Polypeptides of the tail fibres of bacteriophage T4. J. Mol. Biol.62, 465–477 (1971)
Lissitzky, S., Rolland, M., Reynaud, J., Lasry, S.: Oxydation de la tyrosine et de peptides ou protéines la contenant, par la polyphénoloxydase de champignon — III: Propiétés des protéines oxydées et de certaines DOPA-protéines. Biochim. Biophys. Acta65, 481–494 (1962)
Liu, T.Y., Chang, Y.H.: Hydrolysis of proteins with p-toluene sulfonic aicd. J. Biol. Chem.246, 2842–2848 (1971)
Miller, D.W., Elgin, S.C.R.: Isoelectric focussing of proteins exposed to SDS. Anal. Biochem.60, 142–147 (1974)
Moore, S.: Amino acid analysis: Aqueous dimethyl sulfoxides as solvent for the ninhydrin reaction. J. Biol. Chem.243, 6281–6283 (1968)
Moore, S., Spackman, D.H., Stein, W.H.: Chromatography of amino acids on sulfonated polystyrene resins. Anal. Chem.30, 1185–1190 (1958)
Murphy, J.B., Kies, M.W.: Spectrophotometric determination of proteins in dilute solutions. Biochim. Biophys. Acta45, 382–384 (1960)
Nagatsu, T., Levitt, M., Udenfriend, S.: Tyrosine hydroxylase the initial step in norepinephrine biosynthesis. J. Biol. Chem.239, 2910–2916 (1964)
Pau, R.N., Brunet, P.C.J., Williams, M.J.: The isolation and characterization of proteins from the left colleterial gland of the cockroach,Periplaneta americana (L.). Proc. Roy. Soc. Lond. B177, 565–579 (1971)
Saleuddin, A.S.M.: Ultrastructural studies on the formation of periostracum inHelix aspersa (Mollusca). Calcif. Tiss. Res.22, 49–65 (1976)
Swank, R.T., Munkres, K.D.: Molecular weight analysis of oligopeptides by polyacrylamide gels with sodium dodecyl sulfate. Anal. Biochem.39, 462–477 (1971)
Waite, J.H.: Evidence for the mode of sclerotization in a molluscan periostracum. Comp. Biochem. Physiol.58B, 157–162 (1977)
Waite, J.H., Andersen, S.O.: 3,4-Dihydroxyphenylalanine in an insoluble shell protein ofMytilus edulis. Biochem. Biophys. Acta541, 107–114 (1978)
Waite, J.H., Wilbur, K.M.: Phenoloxidase in the periostracum of the marine bivalve,Modiolus demissus (Dillwyn). J. Exp. Zool.195, 359–368 (1976)
Wetlaufer, D.B.: Ultraviolet spectra of proteins and amino acids. Adv. Protein Chem.17, 303–390 (1962)
Yagi, K., Nagatsu, T., Nagatsu-Ishibashi, I.: Condensation reaction of DOPA with ethylene diamine. J. Biochem.48, 617–620 (1960)
Yasunobu, K.T., Peterson, E.W., Mason, H.S.: The oxidation of tyrosine containing peptides by tyrosinase. J. Biol. Chem.234, 3291–3295 (1958)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Herbert Waite, J., Saleuddin, A.S.M. & Andersen, S.O. Periostracin — A soluble precursor of sclerotized periostracum inMytilus edulis L.. J Comp Physiol B 130, 301–307 (1979). https://doi.org/10.1007/BF00689847
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00689847