Abstract
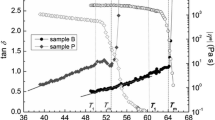
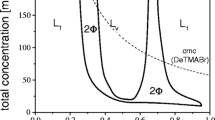
The phase behavior of binary systems composed of water and nonionic surfactants were investigated. The nonionic surfactants studied were the methoxypolyoxyethylene dodecanoates (R11COO(EO) n CH3,n=4.9, 6.1, 7.3, 9.3, and 12.8, wheren is the average number of oxyethylene units).
The phase behaviors of R11COO(EO) n CH3 were compared with those of the polyoxyethylene dodecyl ethers (R12O(EO) p H) and polyoxyethylene methyl dodecyl ethers (R12O(EO) q CH3) which have been previously reported. It was found that the R11COO(EO) n CH3s have lower cloud points and lower upper limit temperatures for the existence of the mesophase as compared to the other two types. A R11COO(EO)12.8CH3/water system had favorable solution properties for practical use, such as a relatively narrow hexagonal liquid crystalline region and a lower melting point than the ordinary alcohol ethoxylate type nonionics.
Similar content being viewed by others
References
Hama I, Nakamura H, Nakano H (1993) INFORM, 4:486, Am Oil Chem Soc Pro of Annual Meeting
Nakagawa T, Kuriyama K, Tori K (1957) Nippon Kagaku Zasshi 78:1568
Nakagawa T, Kuriyama K, Tori K (1957) Nippon Kagaku Zasshi 78:1573
Nakagawa T, Inoue H, Kuriyama K, Oyama T (1958) Nippon Kagaku Zasshi 79:345
Nakagawa T, Inoue H, Kuriyama K, Oyama T (1958) Nippon Kagaku Zasshi 79:348
Nakagawa T, Inoue H, Tori K, Kuriyama K (1958) Nippon Kagaku Zasshi 79:1194
Conroy JP, Hall C, Leng CA, Rendall K, Tiddy GJT, Walsh J, Lindblom G (1990) Progr Colloid Polym Sci 82:253
Rosevear FB (1954) J Am Oil Chem Soc 31:628
Meguro K and Deguchi K (1972) J Colloid Interface Sci 38:596
Mitchell DJ, Tiddy GJT, Waring L, Bostock T, McDonald P (1983) J Chem Soc, Farady Trans 1 79:975
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fujiwara, M., Miyake, M. & Hama, I. Phase behavior of methoxypolyoxyethylene dodecanoate as compared to polyoxyethylene dodecyl ether and polyoxyethylene methyl dodecyl ether. Colloid Polym Sci 272, 797–802 (1994). https://doi.org/10.1007/BF00652420
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00652420