Abstract
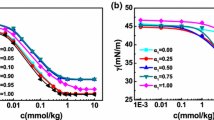
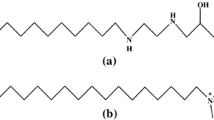
The volumetric properties of the mixed surfactants bis(2-ethylhexyl) sodium sulfosuccinate and ethoxylated-2,5,8,11-tetramethyl-6-dodecyne-5,8-diol (Dynol-604) at various compositions in isooctane have been studied by density measurements at 303.15 K. The apparent specific volumes and the critical reverse micelle concentrations of the binary mixed surfactants were determined. The results were used to calculate the compositions, excess properties of mixing, activity coefficients and the interaction parameter between the two surfactants in the mixed micelles, based on Rubingh’s formalism.




Similar content being viewed by others
References
Xu, J.A., Yin, L., Zhao, J.K., Li, D.X., Hou, W.G.: Surfactant-free microemulsion composed of oleic acid, n-propanol, and H2O. J. Phys. Chem. B 117, 450–456 (2013)
Alexandridis, P., Holzwarth, J.F., Hatton, T.A.: Thermodynamics of droplet clustering in percolating AOT water-in-oil microemulsions. J. Phys. Chem. 99, 8222–8232 (1995)
Zheng, P.Z., Ma, Y.M., Peng, X.H., Yin, T.X., An, X.Q., Shen, W.G.: Determination of the interaction enthalpy between microemulsion droplets by isothermal titration microcalorimetry. Langmuir 27, 12280–12283 (2011)
Assih, T., Larché, F., Delord, P.: Evolution of the radius of the inverse micelles at high dilution in the aerosol-OT/water/n-decane system. J. Colloid Interface Sci. 89, 35–39 (1982)
Eicke, H.F., Arnold, V.: Interactions of proton donors with colloidal electrolytes in apolar solvents. J. Colloid Interface Sci. 46, 101–110 (1974)
Batra, U., Russel, W.B.: Viscosity anomaly and charge fluctuations in dilute AOT microemulsions with X < 20. Langmuir 15, 3718–3725 (1999)
Mehta, S.K., Kaur, K., Bhasin, K.K.: Modification of interface of water/AOT/isooctane reverse micelles by halogenated organodiselenides. Colloids Surf. A 355, 37–44 (2010)
Shaikh, V.R., Dagade, D.H., Hundiwale, D.G., Patil, K.J.: Volumetric studies of aqueous solutions of local anesthetical drug compounds [hydrochlorides of procaine (PC HCl), lidocaine (LC HCl) and tetracaine (TC HCl)] at 298.15 K. J. Mol. Liquids 164, 239–242 (2011)
Wang, J.J., Wang, H.Y., Zhang, S.L., Zhang, H.C., Zhao, Y.: Conductivities, volumes, fluorescence, and aggregation behavior of ionic liquids [C4mim][BF4] and [C n mim]Br (n = 4, 6, 8, 10, 12) in aqueous solutions. J. Phys. Chem. B 111, 6181–6188 (2007)
Peyre, V., Bouguerra, S., Testard, F.: Micellization of dodecyltrimethylammonium bromide in water–dimethylsulfoxide mixtures: A multi-length scale approach in a model system. J. Colloid Interface Sci. 389, 164–174 (2013)
Du, C.F., Wang, H.H., Yin, T.X., Shen, W.G.: The thermodynamic properties of AOT/Dynol-604 mixed surfactant solutions. J. Chem. Thermodyn. 64, 226–230 (2013)
Cai, D.X., Du, C.F., Yin, T.X., Zheng, P.Z., Shen, W.G.: Thermodynamic and microscopic properties of AOT/Dynol-604 mixed micelles in aqueous solutions. Fluid Phase Equilib. 387, 50–58 (2015)
Yin, T.X., Wang, S.Z., Shen, W.G.: Volumetric studies on binary mixtures of surfactants N, N′-bis(dimethyldodecyl)-1,2-ethanediammonium dibromide and 1-dodecyl-3-methylimidazolium bromide in aqueous solutions. J. Chem. Thermodyn. 83, 97–103 (2015)
Du, C.F., He, W., Yin, T.X., Shen, W.G.: Volumetric properties of water/AOT/isooctane microemulsions. Langmuir 30, 15135–15142 (2014)
D’Aprano, A., Lizzio, A., Liveri, V.T.: Enthalpies of solution and volumes of water in reversed AOT micelles. J. Phys. Chem. 91, 4749–4751 (1987)
Yoshimura, Y., Abe, I., Ueda, M., Kajiwara, K., Hori, T., Schelly, Z.A.: Apparent molar volume of solubilized water in AOT/isooctane/water reverse micellar aggregates. Langmuir 16, 3633–3635 (2000)
Valero, M., Sanchez, F., Gomez-Herrera, C., Lopez-Cornejo, P.: Study of water solubilized in AOT/n-decane/water microemulsions. Chem. Phys. 345, 65–72 (2008)
Karukstis, K.K., Zieleniuk, C.A., Fox, M.J.: Fluorescence characterization of DDAB-AOT catanionic vesicles. Langmuir 19, 10054–10060 (2003)
Rosen, M.J., Zhou, Q.: Surfactant–surfactant interactions in mixed monolayer and mixed micelle formation. Langmuir 17, 3532–3537 (2001)
Holland, P.M., Rubingh, D.N.: Nonideal multicomponent mixed micelle model. J. Phys. Chem. 87, 1984–1990 (1983)
Gazzillo, D., Giacometti, A., Fantoni, R., Sollich, P.: Multicomponent adhesive hard sphere models and short-ranged attractive interactions in colloidal or micellar solutions. Phys. Rev. E 74, 051407 (2006)
Yu, D., Huang, X., Deng, M.L., Lin, Y.Y., Jiang, L.X., Huang, J.B., Wang, Y.L.: Effects of inorganic and organic salts on aggregation behavior of cationic gemini surfactants. J. Phys. Chem. B 114, 14955–14964 (2010)
Mahajan, R.K., Nandni, D.: Micellization and phase behavior of binary mixtures of anionic and nonionic surfactants in aqueous media. Ind. Eng. Chem. Res. 51, 3338–3349 (2012)
Du, C.F., Cai, D.X., Qin, M., Zheng, P.Z., Hao, Z.G., Yin, T.X., Zhao, J.H., Shen, W.G.: Thermodynamics of mixed surfactant solutions of N, N’-bis(dimethyldodecyl)- 1,2-ethanediammon-iumdibromide with 1-dodecyl-3-methylimidazolium bromide. J. Phys. Chem. B 118, 1168–1179 (2014)
Al-Wardian, A., Glenn, K.M., Palepu, R.M.: Thermodynamic and interfacial properties of binary cationic mixed systems. Colloids Surf. A 247, 115–123 (2004)
Maeda, H.: A thermodynamic analysis of charged mixed micelles in water. J. Phys. Chem. B 109, 15933–15940 (2005)
Aralaguppi, M.I., Jadar, C.V., Aminabhavi, T.M.: Density, refractive index, viscosity, and speed of sound in binary mixtures of cyclohexanone with hexane, heptane, octane, nonane, decane, dodecane, and 2,2,4-trimethylpentane. J. Chem. Eng. Data 44, 435–440 (1999)
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Projects 21173080, 21373085, 21303055, 21403098) and the Fundamental Research Funds for the Central Universities (WJ1516001).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Du, C., Wu, J., Wang, X. et al. The Thermodynamic Properties of Dynol-604/AOT Mixed Reverse Micelles in Isooctane. J Solution Chem 45, 1031–1042 (2016). https://doi.org/10.1007/s10953-016-0485-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-016-0485-7